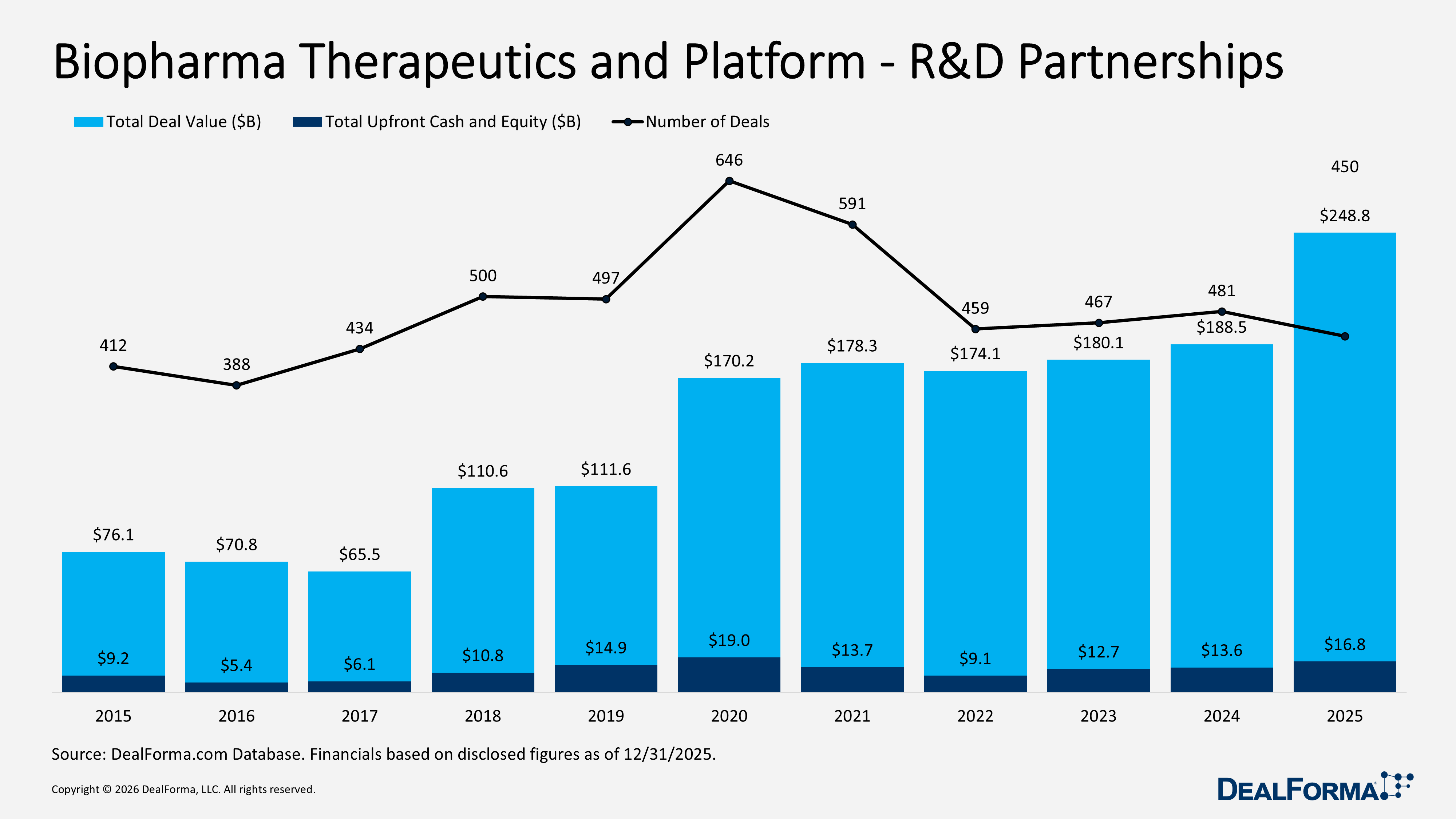
In 2025, Biopharma Therapeutics and Platform R&D partnerships remained highly active with 450 deals worth $248.8B, down 6% from 2024’s 481 deals but up 32% in total value, reflecting fewer but larger transactions with an average deal size of $553M. Upfront cash and equity totalled $16.8B, averaging $107M per deal and a median of $45M, highlighting stronger capital commitment across the market. Therapeutic Platforms and Biopharma drove $212.7B, or 81% of total value, while Diagnostics, Sequencing, Omics, and Tools rose 67% to $44.2B. Deal volume was concentrated in core biopharma/platforms (429 deals, 65% of total), with manufacturing partnerships declining and Medtech/Digital/Wearables modest.
The $100M+ mega-deal segment expanded to 36 partnerships totaling $108.7B, with an average deal value of $3B and a total upfront of $12.2B, showing the growing weight of top-tier deals. Prominent 2025 deals include Hengrui Pharma granting GSK Phase I COPD rights in July for $500M up front and up to $12B milestones; Innovent and Takeda October Phase III NSCLC/gastric ADCs for $1.2B up front ($100M equity) and up to $10.2B milestones, 60/40 US profit share; BioNTech and BMS June bispecific PD-L1/VEGF-A for solid tumors, $1.5B up front, $7.6B milestones, shared global P&L; 3SBio and Pfizer May Phase II NSCLC/metastatic/gyn cancers, $1.3B up front, $100M equity, $4.8B milestones, $150M option fees; and Argo Biopharma and Novartis September ANGPTL3 and RNA candidates, $160M up front, equity investment, potential $5.2B milestones, plus tiered royalties.
In 2025, Biopharma Therapeutics and Platform R&D partnerships totaled 450 deals valued at $248.8B, reflecting sustained activity and a meaningful increase in capital deployment. Compared with 2024, which recorded 481 deals totaling $188.5B, deal volume declined by 31 deals, down 6%, while total value increased by $60.3B, up 32%. This shift indicates a move toward fewer but larger transactions, with average deal size rising to approximately $553M in 2025 versus $392M in 2024. Across 2024 and 2025 combined, the sector generated 931 deals worth $437.2B.
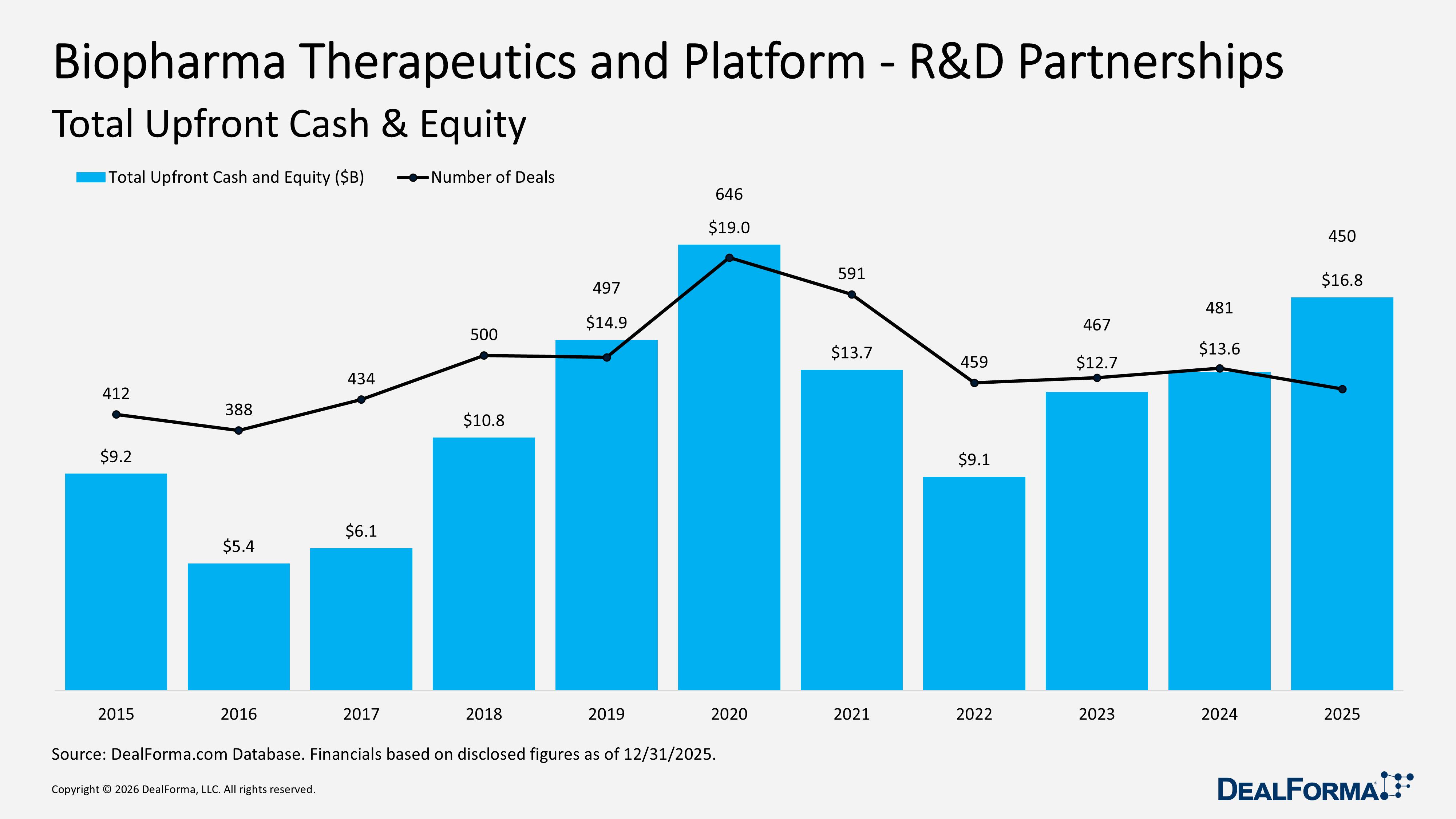
In 2025, Biopharma Therapeutics and Platform R&D partnerships accounted for 450 deals with $16.8B in upfront cash and equity, reflecting stronger capital commitment per transaction despite slightly lower volume. In 2024, the sector recorded 481 deals and $13.6B in upfront value. Year over year, deal count declined by 31 deals, down 6%, while upfront funding increased by $3.2B, up 24%, lifting the average upfront size to approximately $37M per deal in 2025 versus $28M per deal in 2024. Across 2024 and 2025 combined, the market generated 931 deals and $30.3B
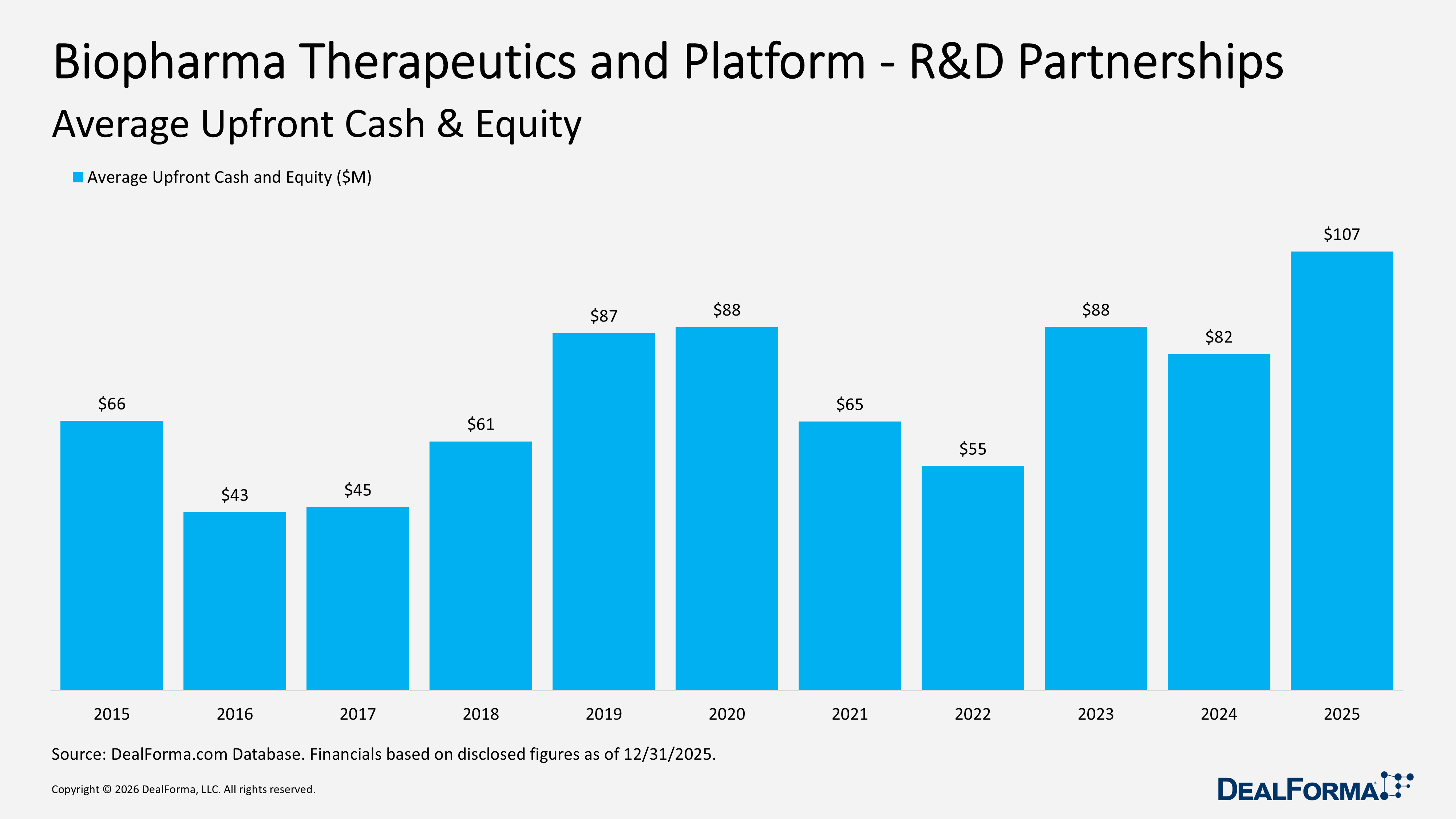
In 2025, the average upfront cash and equity per Biopharma Therapeutics and Platform R&D partnership increased to $107M, indicating greater capital commitment at signing to secure assets. In 2024, the average upfront was $82M, representing a $25M increase, up 30% year over year.
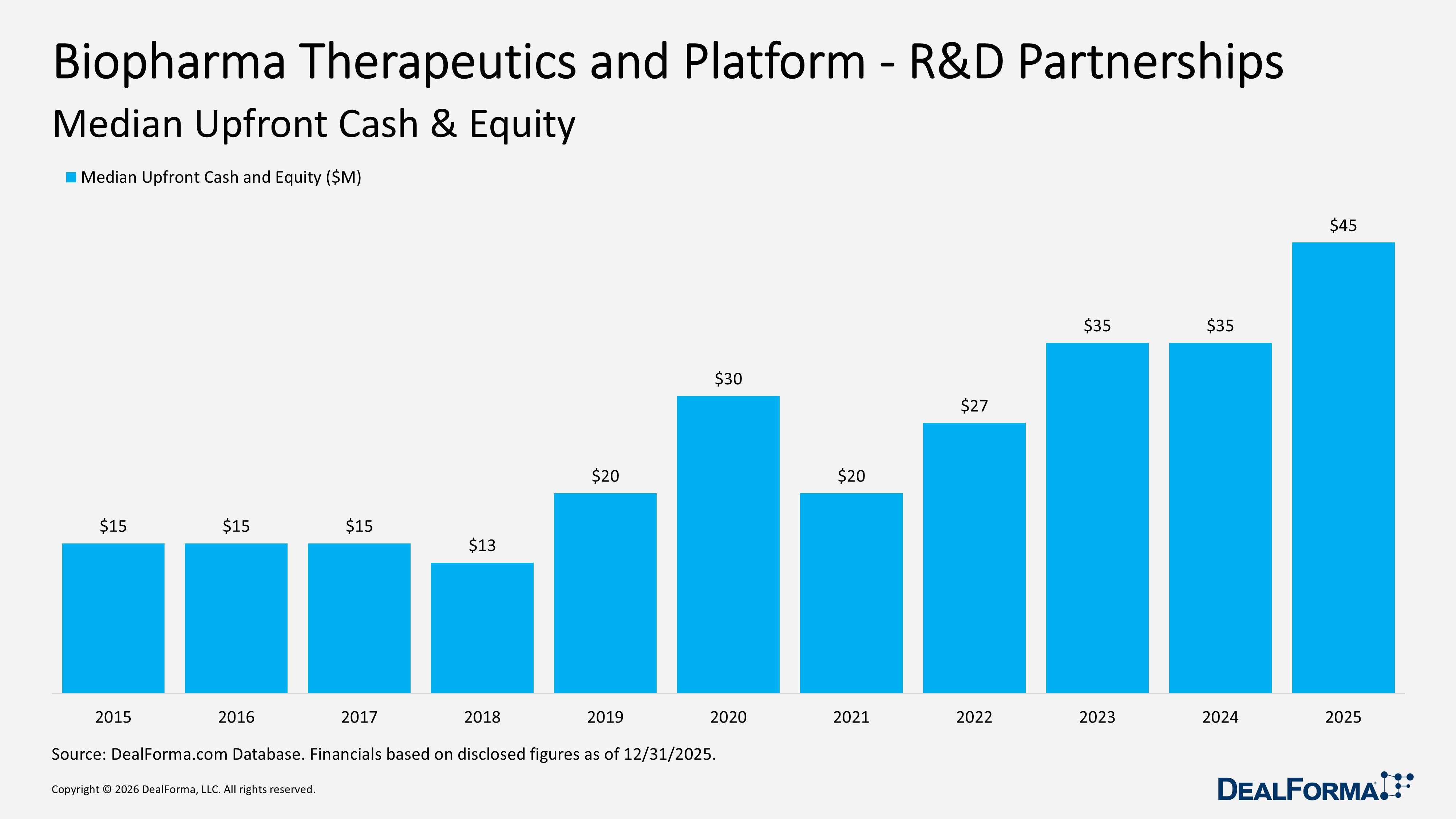
In 2025, the median upfront cash and equity for Biopharma Therapeutics and Platform R&D partnerships increased to $45M, indicating that the typical transaction carried a higher initial payment. In 2024, the median was $35M, representing a $10M increase, up 29% year over year.
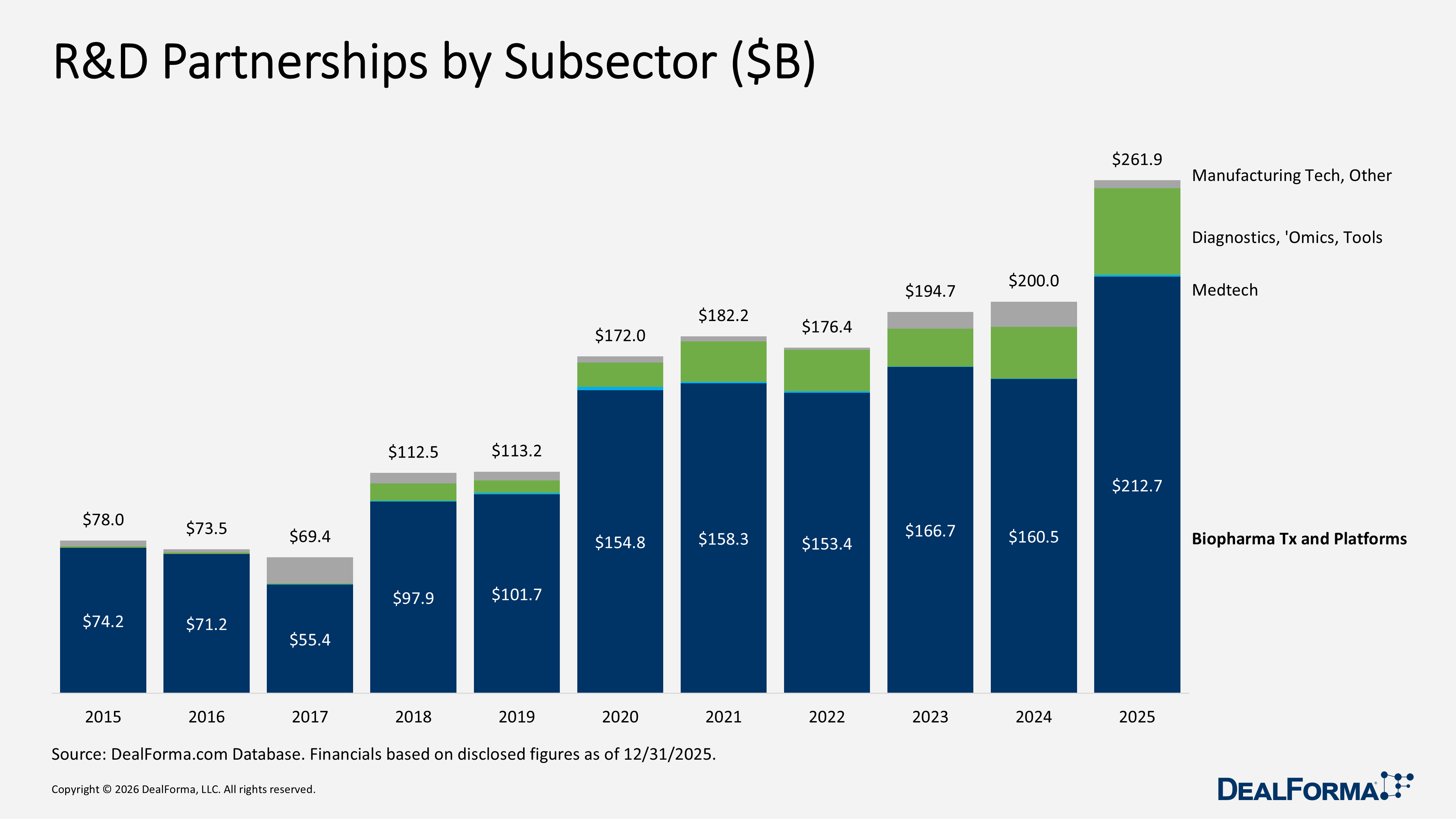
R&D Partnerships by Subsector
In 2025, total R&D partnership value across healthcare and life sciences reached $261.9B, up from $200B in 2024, an increase of $61.9B, 31% growth, reflecting a materially stronger value environment year over year. Therapeutic Platforms and Biopharma remained the primary drivers, rising from $160.5B to $212.7B, up $52.2B, 33%, and accounting for approximately 81% of the 2025 total value. This confirms that core biopharma and platform transactions generated the most incremental capital deployment. Diagnostics, Sequencing, Omics, and Tools also expanded significantly, increasing from $26.4B to $44.2B, up $17.8B, 67%, signaling stronger investment in enabling technologies supporting drug development. In contrast, Manufacturing Tech and Others declined from $12.9B to $4B, down $8.9B, 69%, indicating fewer large-scale manufacturing collaborations. Medtech, Device, Digital Tx, and Wearables remained modest in absolute terms but grew from $100M to $1B.
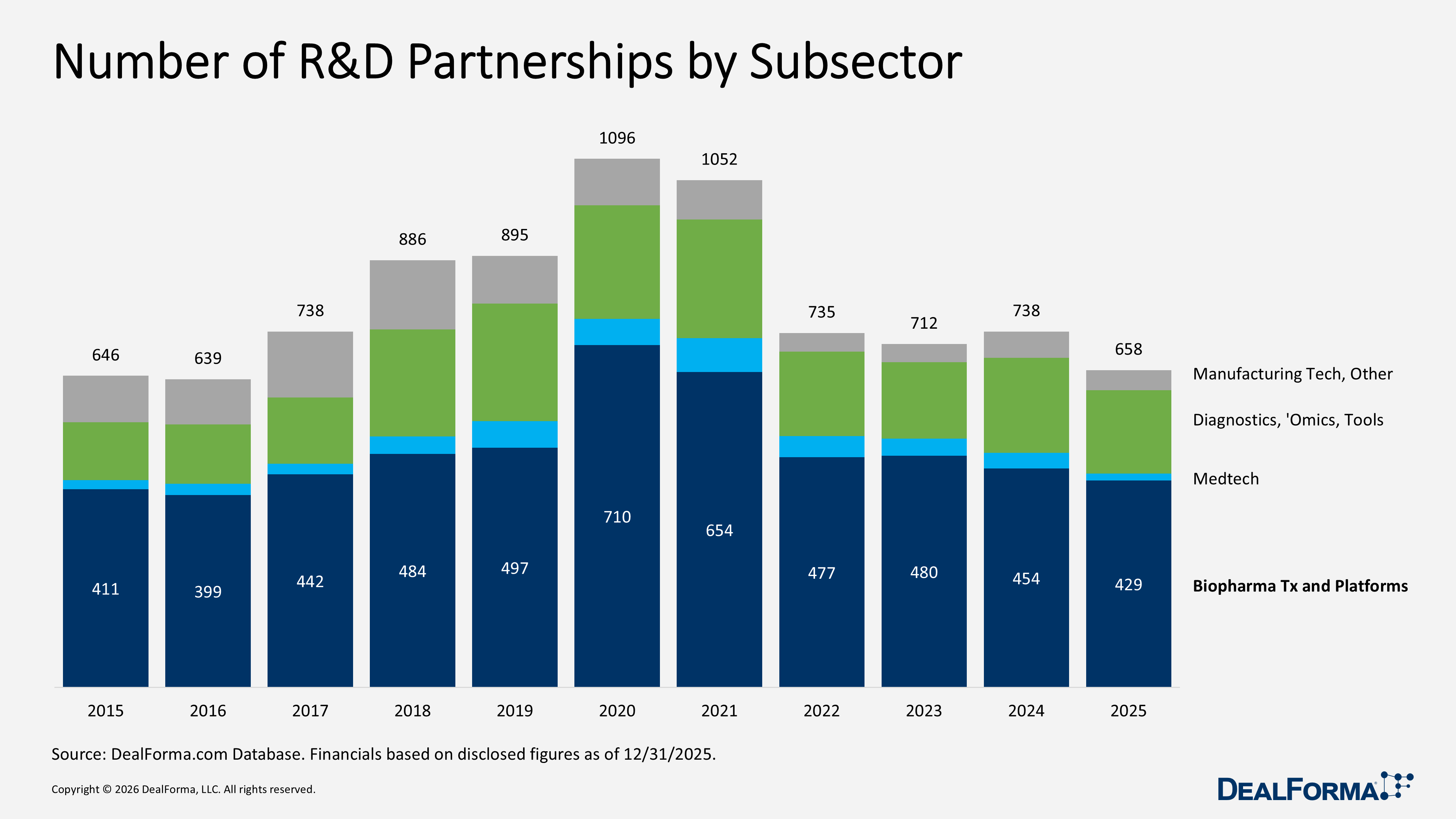
In 2025, total R&D partnerships across subsectors declined to 658 deals, down from 738 in 2024, a reduction of 80 deals, 11%, indicating lower transaction volume despite higher overall deal value. The pullback was broad-based. Therapeutic Platforms and Biopharma decreased from 454 to 429 deals, down 25, 6%, yet still accounted for approximately 65% of all 2025 partnerships, maintaining its position as the market’s primary activity driver. Diagnostics, Sequencing, Omics, and Tools fell from 197 to 173, down 24, 12%. Manufacturing Tech and Others declined from 55 to 42, down 13, 24%, while Medtech, Device, Digital Tx, and Wearables recorded the steepest contraction from 32 to 14, down 18, 56%.
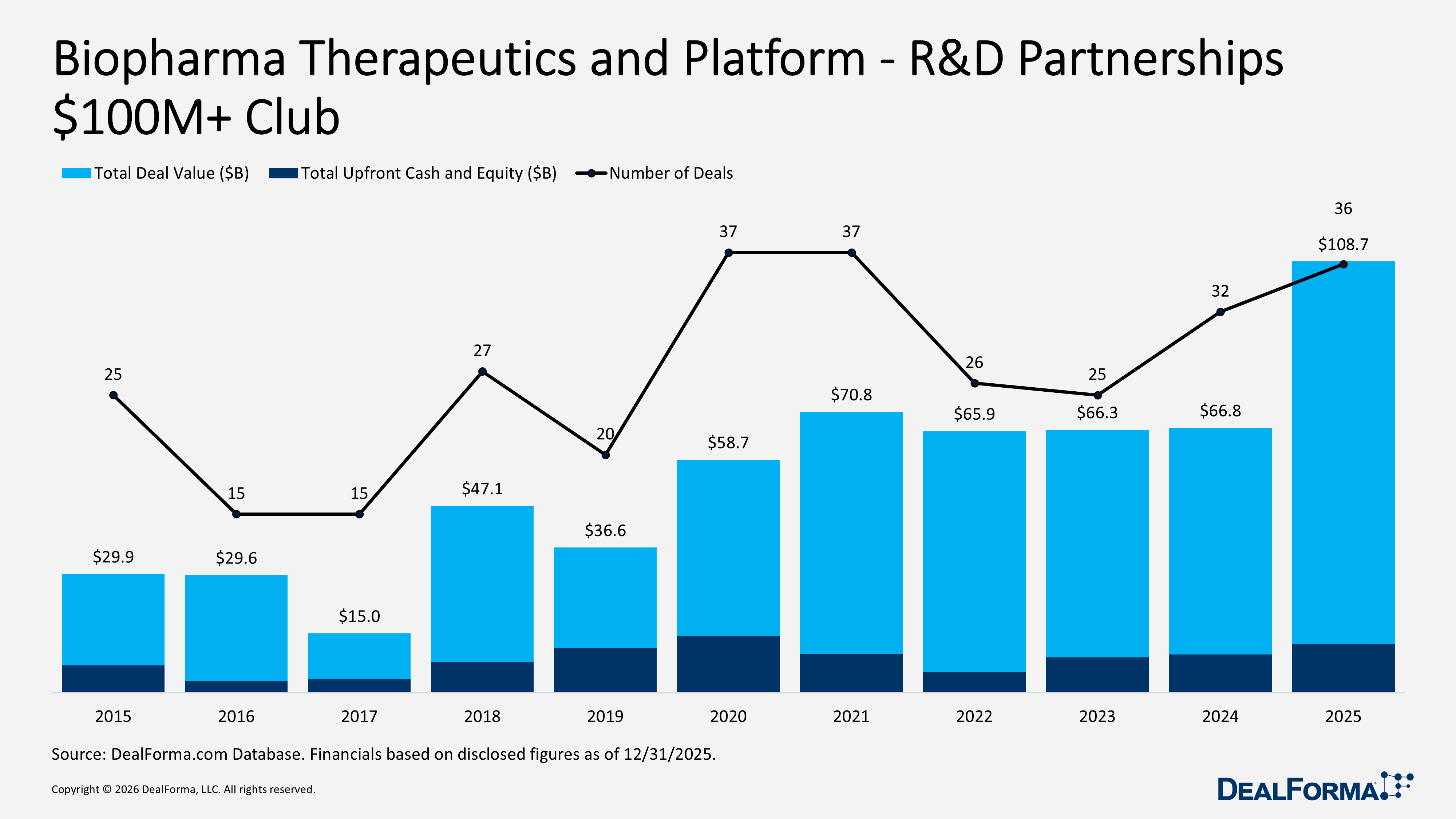
Biopharma Therapeutics and Platform R&D Partnerships – $100M+ Club
In 2025, the Biopharma Therapeutics and Platform $100M+ segment expanded to 36 large partnerships totaling $108.7B, up from 32 deals and $66.8B in 2024. This reflects 4 additional mega-deals, up 13%, and a substantial increase in value of $41.9B, up 63%, indicating not only more top-tier transactions but significantly larger headline collaborations. Deal size metrics confirm this shift. Average total deal value increased from $2.1B to $3B, up approximately 45%, while the median rose from $1.7B to $2B, up 17%, demonstrating that the typical large transaction also scaled higher. Upfront economics strengthened as well, with total upfront cash and equity increasing from $9.7B to $12.2B, up 26%. The average upfront rose from $303M to $339M, and the median upfront increased from $150M to $180M, signaling higher cash commitments at signing even within already sizable partnerships. Across 2024 and 2025 combined, the $100M+ tier accounted for 68 deals worth $175.5B, with 2025 increasingly defined by a larger and more capital-intensive mega-deal segment.
Prominent Biopharma Therapeutics and Platforms R&D Partnerships in 2025
Hengrui Pharma development and commercialization deal with GSK – July 2025
Hengrui Pharma granted GSK exclusive global rights (excluding Greater China) to develop and commercialize HRS-9821, a Phase I PDE3/4 inhibitor for COPD, potentially as a dry-powder inhaler. Hengrui will advance 11 additional early-stage programs through Phase I, after which GSK can opt to develop and commercialize them globally (outside Greater China). The deal includes $500M up front, up to $12B in development, regulatory, and sales milestones, plus tiered royalties.
Innovent Biologics development and commercialization deal with Takeda – October 2025
Innovent Biologics granted Takeda exclusive global rights (excluding China) to develop and commercialize IBI-363 (a Phase III PD-1/IL-2a-bias bispecific antibody for NSCLC and other solid tumors) and IBI-343 (a Claudin 18.2–targeting ADC for gastric and pancreatic cancers), with an option to license IBI-3001, a bispecific ADC in Phase I for advanced solid tumors. The companies will co-develop IBI-363 worldwide (60/40 cost share), co-commercialize in the US with Takeda leading, and Takeda will have exclusive rights outside the US and China. Financial terms include $1.2B up front, including a $100M equity investment, plus up to $10.2B in milestones and royalties, with US profits on IBI-363 shared 60/40. If Takeda licenses IBI-3001, Innovent will receive the option fee, milestones, and royalties.
BioNTech development and commercialization deal with BMS – June 2025
BioNTech granted BMS exclusive worldwide rights to develop and commercialize BNT-327, a bispecific antibody targeting PD-L1 and VEGF-A for multiple solid tumors, including small cell and non-small cell lung cancer, with a Phase III trial in triple-negative breast cancer planned by year-end. BioNTech will supply the drug and retain rights in the US, EU, UK, China, and Turkey, while BMS holds rights elsewhere, with profits booked regionally. The program spans over 10 indications, with both companies retaining flexibility for additional combinations. Financial terms include $1.5B up front, up to $2B in non-contingent payments through 2028, and potential $7.6B in milestones, with development and manufacturing costs, as well as global profits and losses, shared equally.
3SBio development and commercialization deal with Pfizer – May 2025
Shenyang Sunshine Pharmaceutical and 3S Guojian Pharmaceutical (a 3SBio subsidiary) granted Pfizer exclusive global rights (excluding China) to develop, manufacture, and commercialize SSGJ-707, a Phase II bispecific antibody targeting PD-1 and VEGF for NSCLC, metastatic colorectal cancer, and gynecological tumors. Pfizer may commercialize the drug in China and produce the drug substance in Sanford, NC, with drug product manufacturing in McPherson, KS, while 3SBio will initiate the first Phase III trial in China in 2025. Financial terms include $1.3B up front, $100M equity investment, $4.8B in milestones, double-digit tiered royalties, and $150M in option fees payable on July 24, 2025.
Argo Biopharma license option deal with Novartis – September 2025
Argo Biopharma granted Novartis worldwide rights (excluding China) to develop and commercialize its Phase II ANGPTL3 candidate, two additional molecules for severe hypertriglyceridemia and mixed dyslipidemia, and one siRNA candidate in IND-enabling studies. Argo retains first negotiation rights to BW-00112 (ANGPTL3) and has options for profit-and-loss sharing in the US and across multiple geographies, while Novartis holds an ex-China license and a profit-and-loss split option in China. Financial terms include $160M up front, an equity investment, and potential $5.2B in milestones, plus tiered royalties.
Also check out Biopharma Therapeutics and Platforms 2024-Q2 2025