Our 2025 autoimmune review reflects a shift toward quality-driven growth, with stronger R&D momentum, selective but high-value M&A, and broader venture participation, as investment concentrates in scalable platforms, advanced modalities, and differentiated assets, supporting balanced and resilient deal environment. R&D partnerships led activity, with deals rising to 46 from 40 and total value more than doubling from $14.3 billion to $29.2 billion. Upfronts increased from $900 million to $1.2 billion. A lower average upfront of $49 million versus $58 million and a higher median of $36 million versus $20 million indicate improved deal distribution. M&A slowed to 7 deals from 8, with total value declining from $11.2 billion to $4.7 billion and cash excluding contingents falling from $10.3 billion to $3.8 billion. However, stable averages and a sharp rise in median value to $1.5 billion from $250 million highlight focus on sizeable, high-conviction transactions. Venture funding strengthened, with rounds increasing from 34 to 48 and total capital rising from $2.5 billion to $2.9 billion, driven by broader participation despite a decline in average round size from $80 million to $74 million.
Key deals involved Monte Rosa & Novartis (September 2025) AI-enabled degrader partnership with $120 million upfront and up to $5.5 billion+ milestones; RemeGen & Vor Bio (June 2025) telitacicept deal with $45 million upfront, $80 million warrants, and >$4 billion milestones; Syneron Bio & AstraZeneca (March 2025) platform deal with up to $75 million upfront and up to $3.4 billion milestones; AbbVie & Capstan (June 2025, completed August 19, 2025) acquisition for up to $2.1 billion upfront; BMS & Orbital Therapeutics (October 2025, completed December 08, 2025) acquisition for $1.5 billion upfront; Alumis & Acelyrin (February 2025, completed May 21, 2025) all-stock merger with $737 million pro forma value and 0.4814 share exchange; Yarrow Bioscience (December 17, 2025) $200 million private financing tied to VYNE merger; Soufflé Therapeutics (October 7, 2025) $200 million series A; and Abcuro (February 12, 2025) $200 million series C.
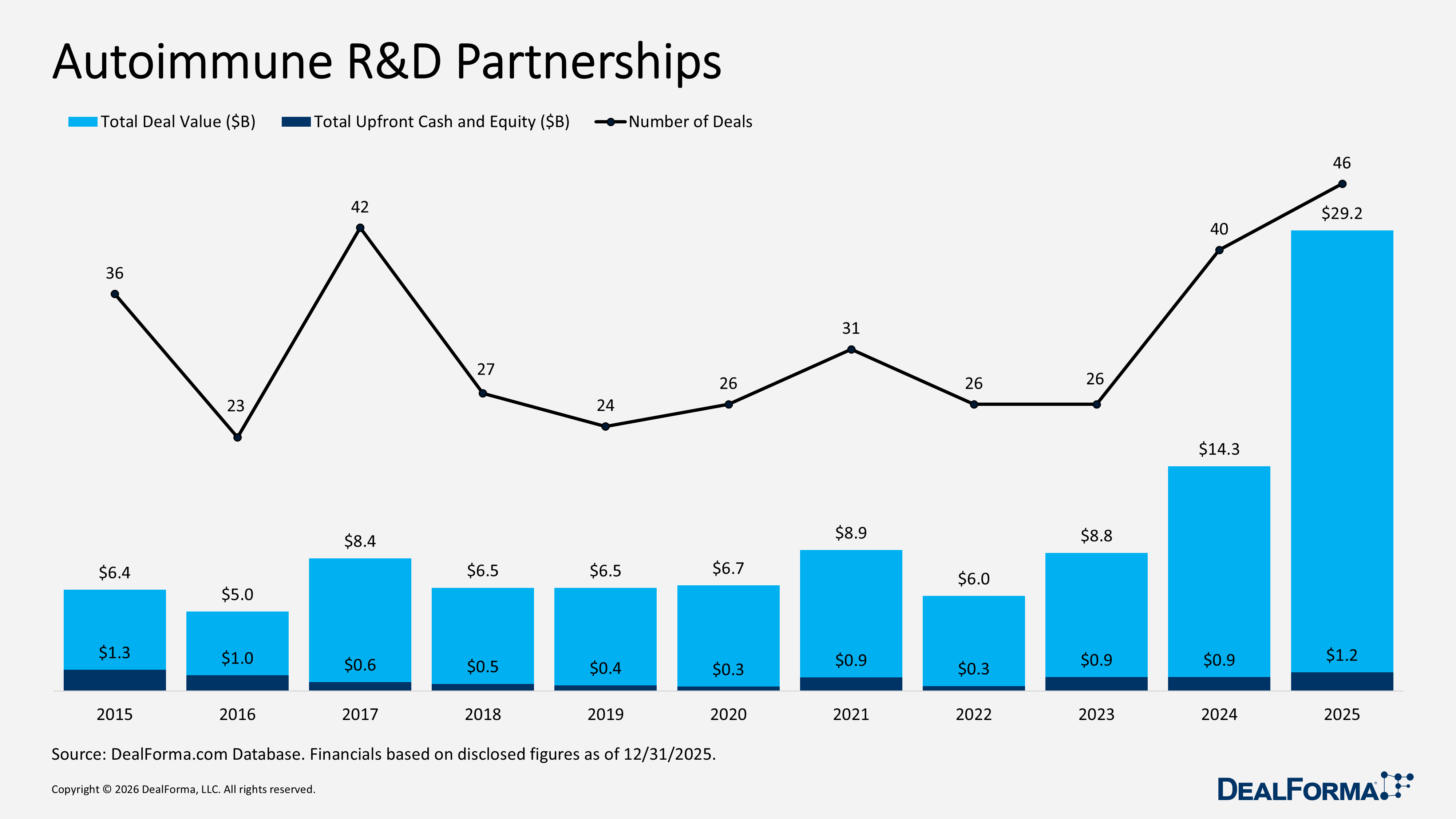
Autoimmune R&D Partnerships
In 2025, autoimmune R&D partnerships rose to 46 deals from 40 in 2024, and total deal value more than doubled year over year, increasing from $14.3 billion to $29.2 billion, indicating a shift toward larger collaborations. Total upfront cash and equity also grew from $900 million to $1.2 billion. However, the average upfront payment declined from $58 million to $49 million, suggesting that while overall deal volume and value increased, individual deal sizes were slightly smaller. Notably, the median upfront payment increased from $20 million to $36 million.
Prominent Autoimmune R&D Partnerships in 2025
Monte Rosa Therapeutics license option deal with Novartis – September 2025
Announced in September 2025, this preclinical/IND-stage autoimmune partnership granted Novartis an exclusive, worldwide option to develop molecular glue degraders using Monte Rosa’s AI/ML-enabled QuEEN platform, building on prior VAV1 work. Novartis can license 1 immunology program and up to 2 additional preclinical programs through IND readiness, with Monte Rosa funding research and Novartis leading development and commercialization. Financial terms include $120 million upfront, up to $60 million in option maintenance, up to $180 million in preclinical and option exercise payments, up to $2.2 billion in development and regulatory milestones, and up to $3.2 billion in commercial milestones, plus high single- to low double-digit royalties on the first program and low double-digit royalties on additional programs.
RemeGen development and commercialization deal with Vor Bio – June 2025
In June 2025, this approved-stage autoimmune deal granted Vor Bio exclusive worldwide rights (excluding China, Hong Kong, Macau, and Taiwan) from RemeGen to develop and commercialize telitacicept, a dual-target protein (BlyS/BAFF and APRIL) for generalized myasthenia gravis. Telitacicept is already approved in China for gMG, systemic lupus erythematosus, and rheumatoid arthritis, with a global Phase III trial in gMG ongoing. Financial terms include $45 million upfront and $80 million in warrants (exercise price $0.0001 per share), with RemeGen eligible for more than $4 billion in regulatory and commercial milestones, plus tiered royalties.
Syneron Bio development and commercialization deal with AstraZeneca – March 2025
In March 2025, at the platform/discovery stage, Syneron Bio granted AstraZeneca rights to develop and commercialize macrocyclic peptides generated via its Synova platform for chronic diseases, including autoimmune, rare, and metabolic conditions. Syneron Bio will receive up to $75 million in upfront and near-term milestone payments (breakout not disclosed), along with an undisclosed equity investment, and is eligible for up to $3.4 billion in development and commercial milestones plus tiered royalties.
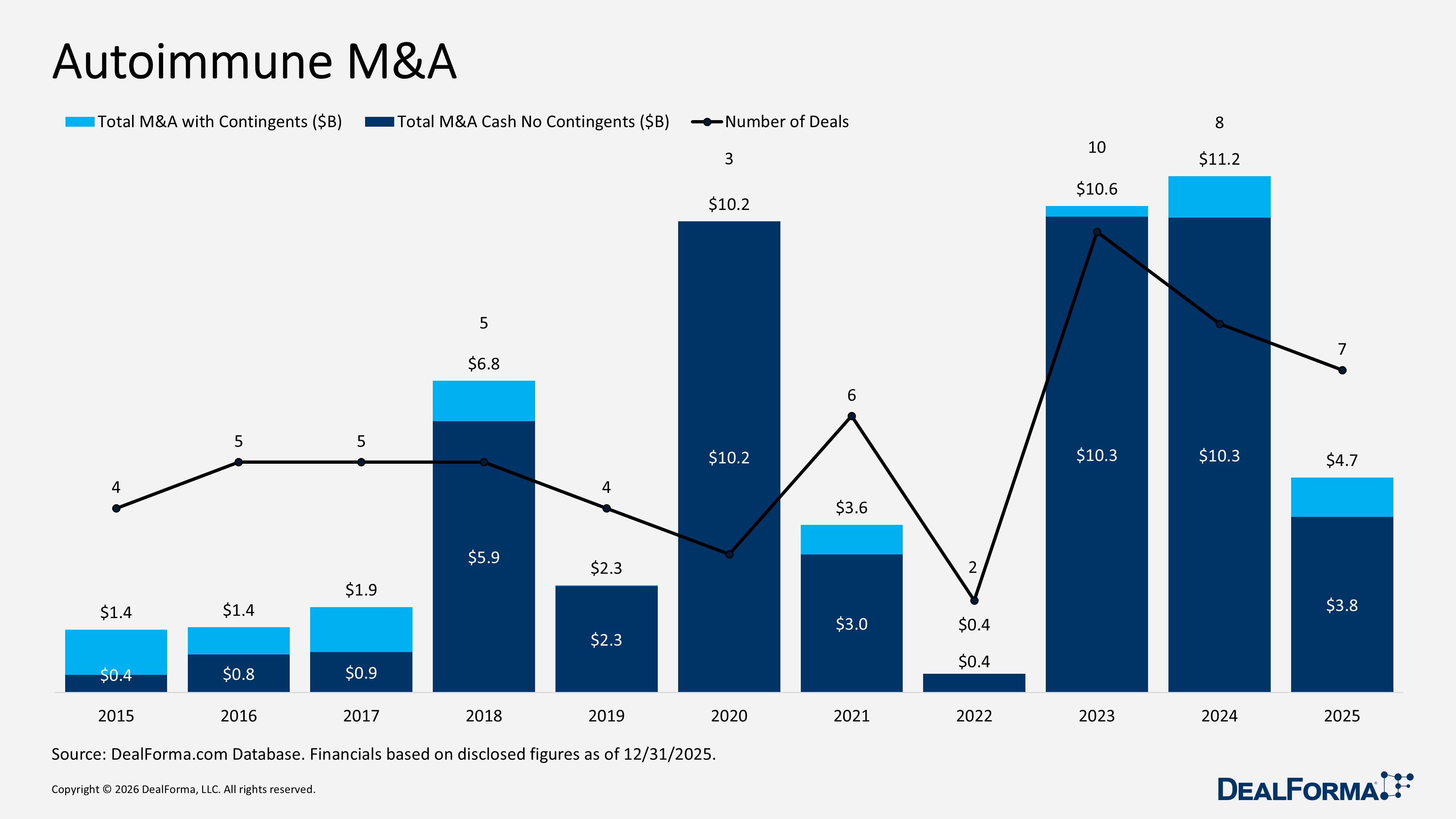
Autoimmune M&A
During 2025, autoimmune M&A activity declined modestly to 7 deals from 8 in 2024, while total deal value dropped significantly from $11.2 billion to $4.7 billion, indicating a slowdown in overall acquisitions. Total cash value excluding contingents also decreased from $10.3 billion to $3.8 billion, reinforcing the reduction in M&A spending. Despite the decline in aggregate value, the average upfront cash and equity value remained broadly stable, with only an approximate $20 million decrease in 2025, suggesting that individual transactions continued to be sizeable. Median upfront value rose from $250 million to $1.5 billion, indicating more consistently large deal sizes, while overall M&A declined in both volume and total value.
Prominent Autoimmune M&A in 2025
AbbVie acquired Capstan – June 2025
Announced in June 2025 at the Phase-I stage, AbbVie acquired Capstan Therapeutics, whose pipeline includes CPTX-2309, an in vivo tLNP anti-CD19 CAR-T therapy for B-cell mediated autoimmune diseases, CPTX-2506, a discovery-stage CAR-T targeting BCMA-expressing plasma cells for cancer and autoimmune indications, along with additional undisclosed programs in immunology, cancer, and fibrotic disorders, supported by its targeted lipid nanoparticle (tLNP) RNA delivery platform. The deal includes up to $2.1 billion in upfront cash (subject to customary adjustments), and the acquisition was completed on August 19, 2025.
BMS acquired Orbital Therapeutics – October 2025
Announced in October 2025 at the preclinical/IND stage, Bristol Myers Squibb acquired Orbital Therapeutics, whose lead asset is OTX-201, an in vivo CAR-T cell therapy for autoimmune diseases, supported by its RNA platform. The transaction includes $1.5 billion in upfront consideration. The acquisition was completed on December 08, 2025.
Alumis acquired Acelyrin in an all-stock Transaction – February 2025
Announced in February 2025 at the Phase II stage, Alumis acquired Acelyrin in an all-stock deal to build a clinical-stage biopharma focused on immune-mediated diseases, led by lonigutamab alongside ESK-001 and A-005, expanding into neurologic and inflammatory indications. Final ownership was approximately 52% Alumis and 48% Acelyrin, with a 0.4814 share exchange ratio and a combined pro forma value of approximately $737 million (cash: $289 million and $448 million). The merger was approved on May 13, 2025, and completed on May 21, 2025, with Acelyrin delisted from Nasdaq.
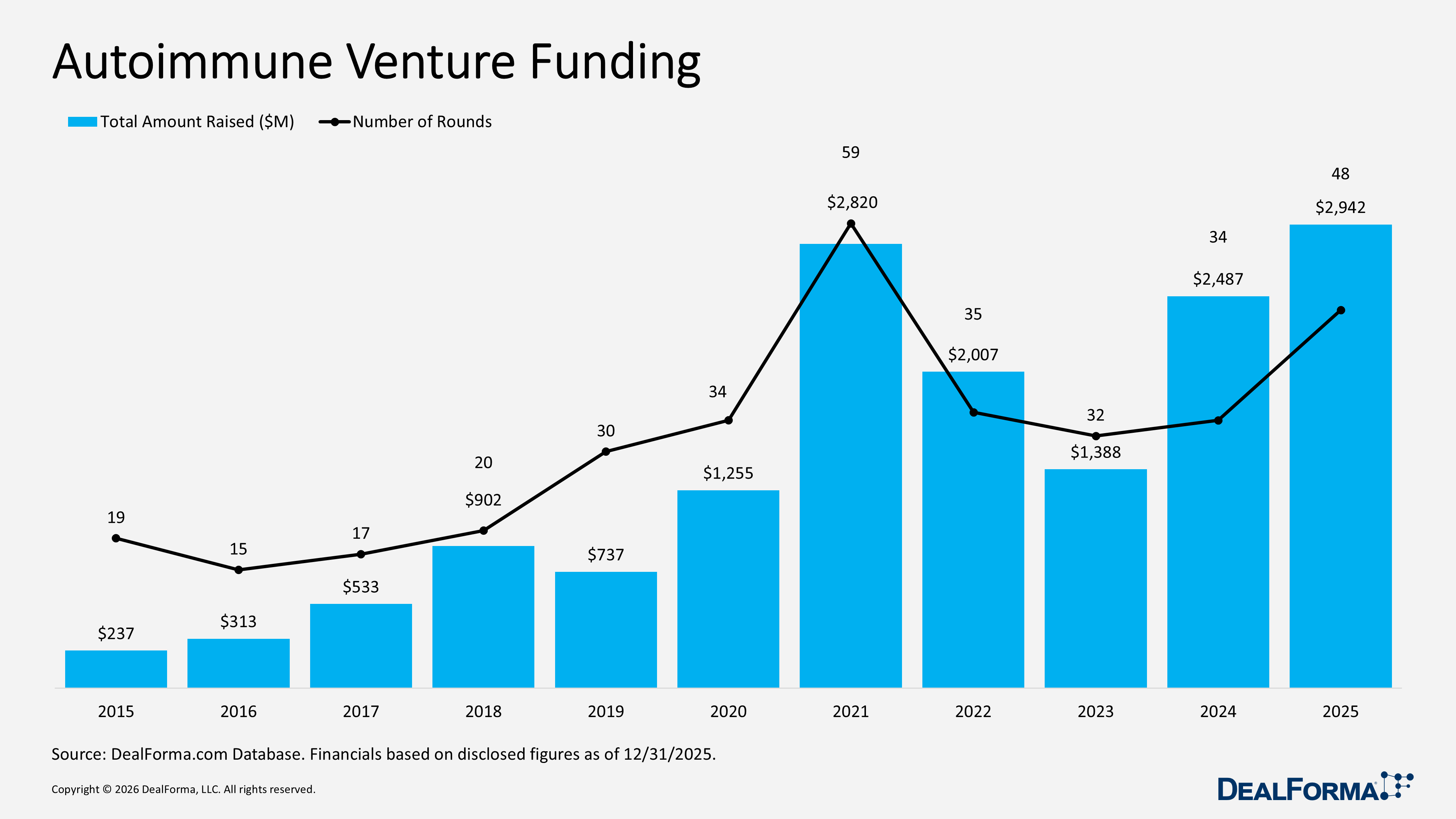
Autoimmune Venture Funding
In 2025, autoimmune venture funding strengthened, with the number of rounds increasing to 48 from 34 in 2024. Total capital rose from $2.5 billion to $2.9 billion, implying improved funding momentum. However, the average round size declined from $80 million to $74 million, indicating growth was driven by higher deal volume rather than larger rounds.
Prominent Autoimmune Venture Funding in 2025
Yarrow Bioscience – Private – $200M – December 2025
In December 2025, Yarrow Bioscience raised approximately $200 million in a private pre-closing financing tied to its all-stock merger with VYNE Therapeutics (post-merger ticker: YARW), led by RTW Investments with participation from OrbiMed, Janus Henderson, venBio, Logos Capital, LifeSci Venture Partners, and Perceptive Advisors. The company is focused on autoimmune thyroid diseases, including Graves’ Disease and Thyroid Eye Disease, with lead asset YB-101 (GS-098), a first-in-class TSHR antibody, and plans to initiate a U.S. Phase 1b/2b trial in H1 2026.
Souffle Therapeutics – Series A – $200M – October 2025
In October 2025, Soufflé Therapeutics raised approximately $200 million in a series A (alongside seed) financing for the Robert Langer co-founded Watertown, MA-based biotech, with backing from NEA, Eli Lilly, Atlas Venture, Avidity Partners, F-Prime, GV, Alexandria, and Perceptive Advisors, alongside strategic partners AbbVie, Amgen, Bayer, and Novo Nordisk. The company is developing cell-specific siRNA therapies targeting autoimmune, cardiac, metabolic diseases, and muscular dystrophies, with first-in-human trials planned for 2026.
Abcuro – Series C – $200M – February 2025
In February 2025, Abcuro raised $200 million in a series C financing led by NEA with new participation from Foresite Capital, alongside existing investors including RA Capital, Bain Capital Life Sciences, Redmile, Samsara, Sanofi Ventures, and BlackRock. The Newton, MA-based company is advancing ulviprubart (ABC008), a first-in-class anti-KLRG1 monoclonal antibody for Inclusion Body Myositis and T-LGLL, which has received FDA and EMA Orphan Drug Designation. Phase 2/3 MUSCLE trial data is expected in H1 2026.
Also check out Autoimmune R&D Partnerships and M&A by Tech – H1 2025 Review