Biopharma dealmaking in 2024 to mid-2025 remained oncology-led, with neurology commanding premium terms and endocrine/metabolic showing strong upfront economics and top venture averages. Selectivity drove M&A toward late-stage, high-value assets, while venture activity highlighted next-generation platforms, including AI-driven discovery, neurotech, and obesity solutions. From 2024 through Q2 2025, activity was defined by a handful of large-scale transactions across R&D, M&A, and venture: Arrowhead licensed seven RNA programs to Sarepta in Nov 2024 in a deal worth up to $10 billion, BioNTech granted BMS rights to its PD-L1/VEGF-A antibody for up to $11.1 billion in Jun 2025, and Pfizer secured global rights to 3SBio’s PD-1/VEGF bispecific in May 2025 for up to $6.2 billion.
On the M&A side, Johnson & Johnson acquired Intra-Cellular Therapies for $14.6 billion (Apr 2025), Sanofi purchased Blueprint Medicines for $9.5 billion (Jul 2025), and Mallinckrodt merged with Endo in a $6.7 billion stock-and-cash deal (Aug 2025). Venture financings included ARCH and Foresite’s $1 billion launch of AI-driven Xaira Therapeutics (May 2025), Neuralink’s $650 million raise (Jun 2025) to expand brain-computer interfaces, and Verdiva Bio’s $411 million series A (Jun 2025) to advance obesity and cardiometabolic therapies.
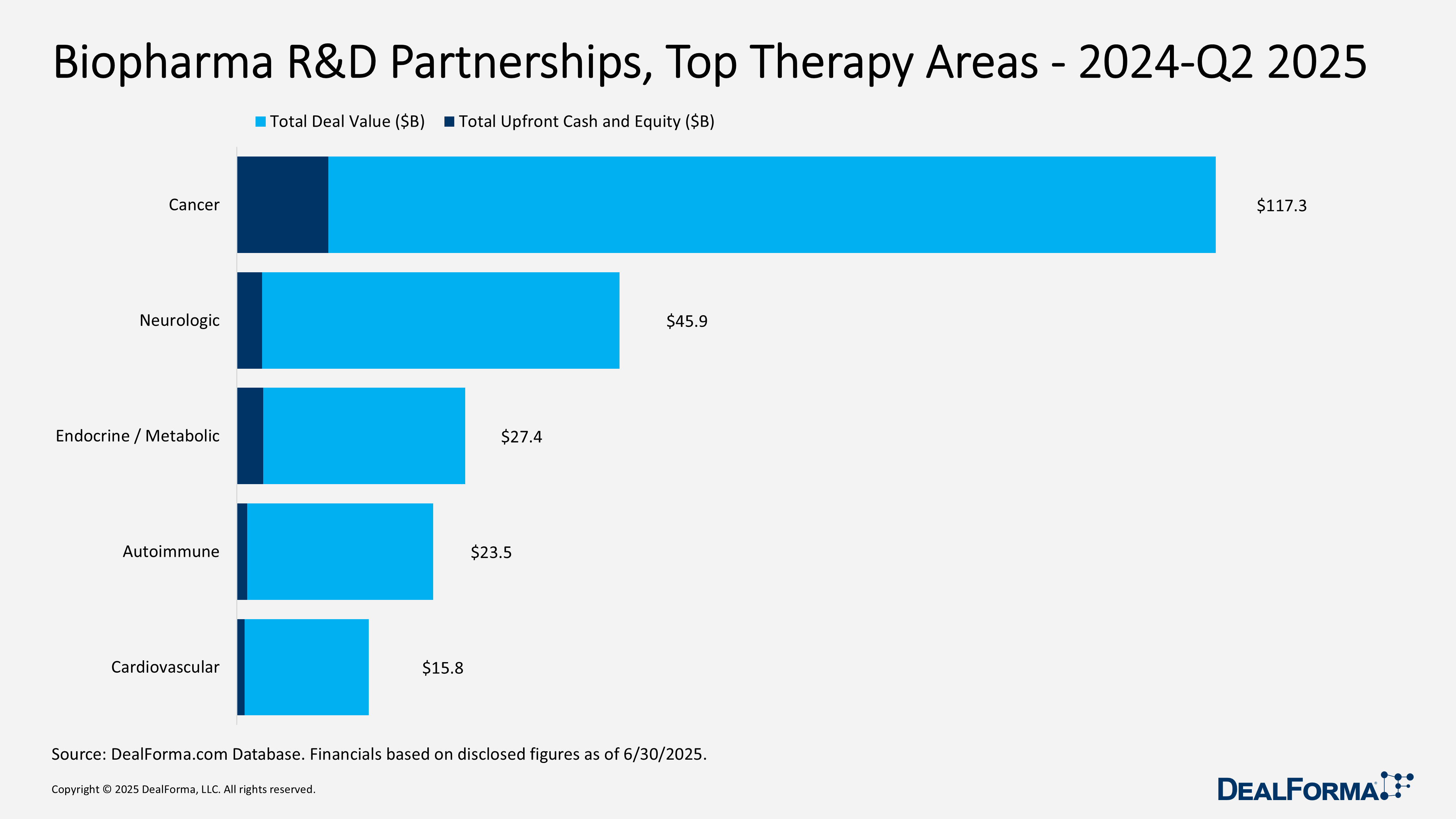
Biopharma R&D Partnerships, Top Therapy Areas – 2024 – Q2 2025
From 2024 through Q2 2025, biopharma R&D partnerships totaled 466 deals worth $229.9 billion, with $19.5 billion in upfront payments. Oncology dominated with 249 agreements ($117.3 billion; $11 billion upfront), where outsized licenses drove the average ($129 million) far above the median ($45 million). Neurology followed with 88 deals ($45.9 billion; $3.1 billion upfront), averaging $85 million but with modest median terms ($28 million). Endocrine/metabolic, although smaller in volume (45 deals), delivered the strongest economics, with a total of $27.4 billion and $3.2 billion upfront (12% of deal value), highlighting concentrated bets on high-value assets. Autoimmune (57 deals; $23.5 billion; $1.3 billion upfront) showed broad but lighter activity, while cardiovascular (27 deals; $15.8 billion; $900 million upfront) rounded out the top five.
Top Biopharma R&D Partnerships – 2024 – Q2 2025
Arrowhead development and commercialization deal with Sarepta Therapeutics – November 2024
Arrowhead granted Sarepta exclusive global rights to seven RNA programs across neurology, pulmonary, and musculoskeletal indications, including Huntington’s disease, myotonic dystrophy type 1, pulmonary fibrosis, and multiple ataxias in a deal valued at up to $10 billion. Terms included $500 million upfront, $325 million equity (11.9 million shares at a 35% premium), $250 million in annual fees, and milestones spanning $300 million near-term, $110 million-$180 million development, and $500 million-$700 million sales per program, plus royalties. Arrowhead leads discovery and manufacturing via its TRiM platform, with Sarepta funding defined clinical work. In 2025, Arrowhead secured a $100 million milestone for DM1 trial enrolment, while Sarepta restructured its finances and sold $224 million of Arrowhead stock, partly in exchange for milestone obligations.
BioNTech development and commercialization deal with BMS – June 2025
In June 2025, BioNTech licensed BMS exclusive global rights to BNT-327, a bispecific antibody targeting PD-L1 and VEGF-A, in a deal worth up to $11.1 billion. BNT-327, acquired via BioNTech’s Biotheus buyout, is in global Phase III trials for small cell and non-small cell lung cancer, with plans for triple-negative breast cancer by year-end 2025 and over 20 additional studies across solid tumors. BioNTech will receive $1.5 billion upfront, up to $2 billion in fixed anniversary payments through 2028, and up to $7.6 billion in milestones, while both companies will split development, manufacturing, and worldwide profits equally.
3SBio development and commercialization deal with Pfizer – May 2025
In May 2025, Pfizer secured exclusive global rights (ex-China) to SSGJ-707, a PD-1/VEGF bispecific antibody from Shenyang Sunshine and 3SBio’s 3S Guojian, in a deal worth up to $6.2 billion. The asset is advancing toward a first Phase III trial in China in 2025 for NSCLC, with potential in colorectal and gynecological cancers. Deal terms include $1.2 billion upfront, a $100 million equity investment, up to $4.8 billion in milestones, and tiered royalties, with Pfizer also gaining an option for China commercialization and US manufacturing rights. On July 24, 2025, 3SBio received an additional $150 million option fee.
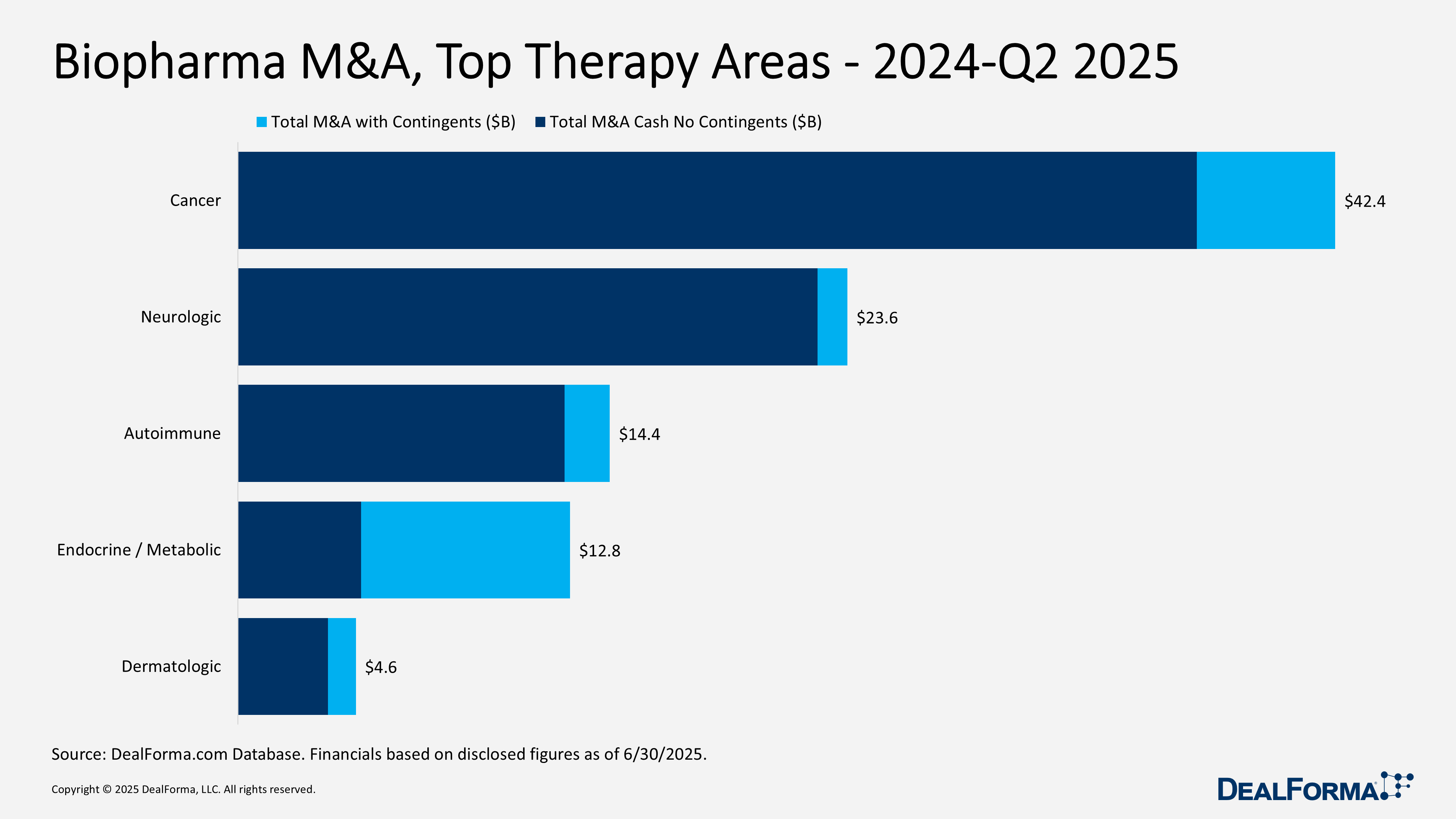
Biopharma M&A, Top Therapy Areas – 2024 – Q2 2025
From 2024 through Q2 2025, biopharma M&A reached notable scale and selectivity across therapeutic areas. Oncology led with 55 deals totaling $42.4 billion ($37.1 billion cash), averaging $1 billion upfront (median $403 million). Neurology followed with 17 transactions worth $23.6 billion ($22.4 billion cash), delivering the richest terms at $1.6 billion average and $479 million median upfront, with 95% of value paid in cash. Autoimmune recorded 13 deals for $14.4 billion ($12.6 billion cash), skewed by a few large buys, with $1.3 billion average and $250 million median upfront. Endocrine/metabolic logged 9 deals worth $12.8 billion but with just 37% in cash ($4.8 billion), pairing a $595 million average with the highest median upfront at $535 million, reflecting balanced mid-sized payouts and heavier earnout structures. Dermatology rounded out the group with 10 deals at $4.6 billion ($3.5 billion cash), averaging $497 million upfront (median $275 million).
Top Biopharma M&A – 2024 – Q2 2025
Johnson & Johnson acquiring Intra-Cellular Therapies – January 2025
In January 2025, Johnson & Johnson agreed to acquire Intra-Cellular Therapies for $132 per share in cash (40% premium), valuing the deal at $14.6 billion. Intra-Cellular’s portfolio is anchored by Caplyta, approved for schizophrenia, bipolar disorder, depression, sleep disorders, and autism, with additional late-stage development in Parkinson’s disease, heart failure, anxiety, and psychosis. The pipeline also includes ITI-1020 (Phase I, oncology), ITI-333 (pain), and multiple preclinical CNS programs. The acquisition, completed April 2, 2025, positions J&J to expand its neuroscience and CNS franchise, with Intra-Cellular continuing as a standalone business unit.
Sanofi acquiring Blueprint Medicines – June 2025
In June 2025, Sanofi announced the $9.5 billion acquisition of Blueprint Medicines, paying $129 per share in cash (27% premium), including $9.1 billion upfront and up to $400 million in milestones tied to BLU-808. Blueprint’s portfolio is anchored by Ayvakit, approved for GIST and systemic mastocytosis, alongside late-stage elenestinib (Phase III, systemic mastocytosis) and BLU-808 (Phase II, chronic urticaria and allergic rhinoconjunctivitis, with expansion into asthma and mast cell activation syndrome). The pipeline also includes discovery programs in solid tumors, breast cancer, and protein degraders targeting CDK2 and CDK4. Sanofi completed the acquisition on July 18, 2025, with Blueprint shares delisted from NASDAQ.
Mallinckrodt acquiring Endo in a stock and cash transaction – March 2025
In March 2025, Mallinckrodt announced a $6.7 billion stock-and-cash acquisition of Endo, adding 27 approved products, 101 generics, and 40 sterile injectables in development. Endo’s marketed portfolio includes Aveed and Testim (hypogonadism), Frova (migraine), Nascobal (anemia), Edex (erectile dysfunction), Percocet (pain), Supprelin-LA (precocious puberty, prostate cancer), Theo-24 (respiratory), and Valstar (oncology), among others, spanning CNS, cardiovascular, infectious disease, and metabolic indications. The merger will give Mallinckrodt shareholders 50.1% and Endo shareholders 49.9% of the combined company, with plans to divest generics and sterile injectables into a standalone entity. Endo shareholders will also receive $80 million in cash. Shareholders approved the deal on June 13, 2025, and Mallinckrodt completed the acquisition on August 4, 2025, creating a diversified specialty pharma platform.
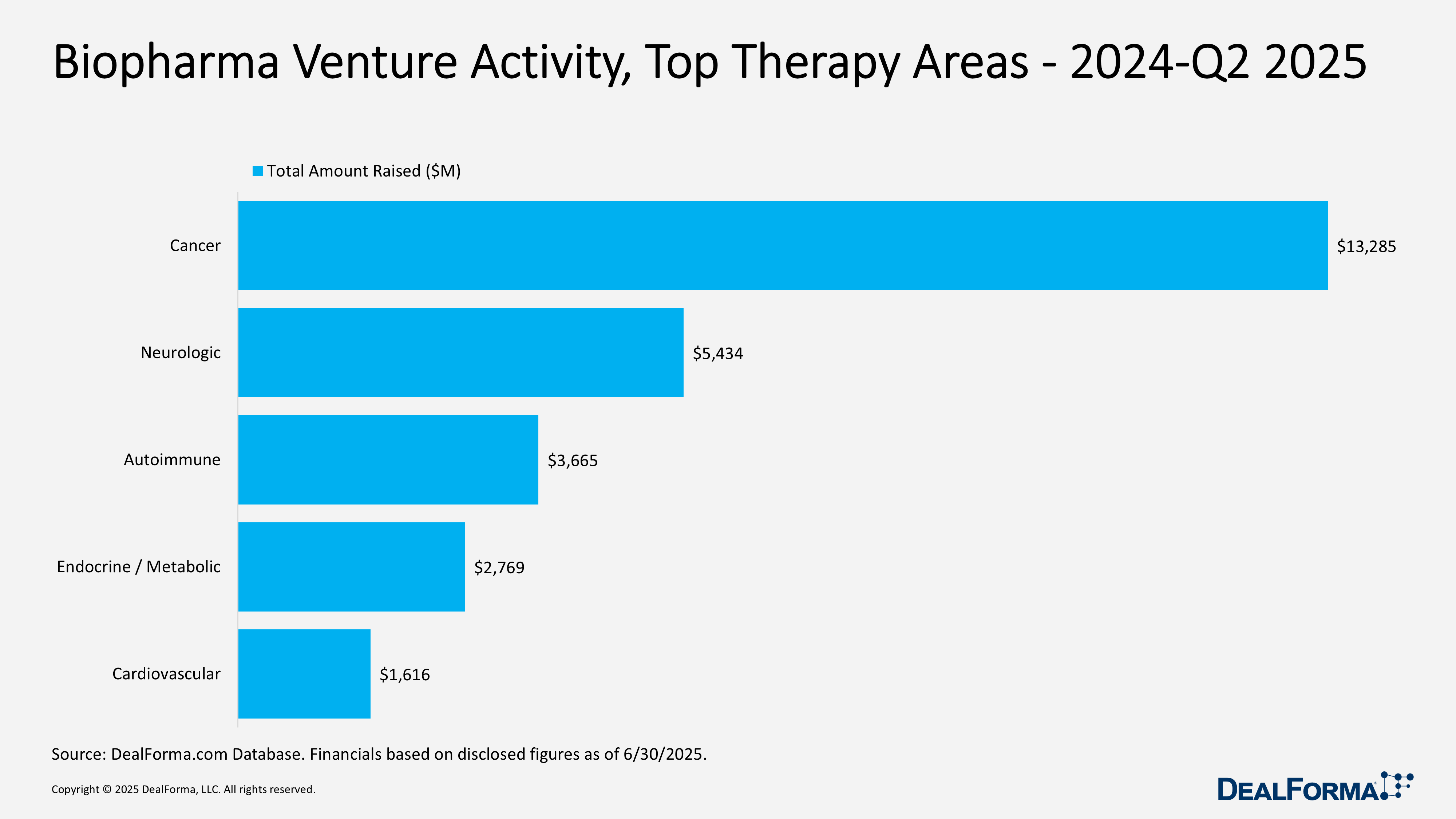
Biopharma Venture Activity, Top Therapy Areas – 2024 – Q2 2025
From 2024 through Q2 2025, biopharma venture funding reached $26.8 billion across 448 rounds (avg $60 million). Oncology dominated with 227 financings raising $13.3 billion (avg $63 million), accounting for roughly half of total capital. Neurology followed with 109 rounds and $5.4 billion (avg $55 million). Autoimmune drew $3.7 billion over 47 rounds, with larger average checks of $85 million, while endocrine/metabolic posted the highest per-round average at $92 million from 39 rounds ($2.8 billion total). Cardiovascular closed with 26 financings worth $1.6 billion (avg $65 million).
Top Biopharma Venture Activity – 2024 – Q2 2025
Xaira – Series A – $1B – April 2024
In May 2025, ARCH Venture Partners and Foresite Labs (Foresite Capital) launched Xaira Therapeutics with $1 billion in funding, joined by F-Prime, NEA, Sequoia, Lux, Lightspeed, Menlo, Two Sigma, and SV Angel. Headquartered in the San Francisco Bay Area, Xaira integrates machine learning, data generation, and therapeutic development to accelerate drug discovery. Co-founded by David Baker, Ph.D. (University of Washington), the company leverages advanced protein- and antibody-design models (RFdiffusion, RFantibody) and incorporates technologies spun out of Illumina’s functional genomics unit and Interline’s proteomics group. The platform aims to design novel connections between biological targets, engineered molecules, and human diseases.
Neuralink – Series E – $650M – June 2025
In June 2025, Neuralink raised $650 million in series E funding to expand patient access and develop new brain-computer interface devices. The round drew backing from ARK Invest, DFJ Growth, Founders Fund, G42, Human Capital, Lightspeed, QIA, Sequoia, Thrive, Valor Equity, and Vy Capital, among others. Neuralink’s N1 implant, with 1,024 electrodes across 64 threads, is designed to enable paralyzed patients to control digital devices with their minds. The company previously raised $280 million in series D (Aug 2023) and has now secured over $1.2 billion to date.
Verdiva Bio – Series A – $410M – January 2025
Verdiva Bio launched in June 2025 as a clinical-stage biotech developing oral and injectable therapies for obesity and cardiometabolic disorders. The company closed an oversubscribed $411 million series A, co-led by Forbion and General Atlantic, with backing from RA Capital, OrbiMed, Logos, Lilly Asia Ventures, and LYFE Capital. Its lead candidate is a potential first-in-class, once-weekly oral obesity treatment designed to improve patient access and affordability.
Also check out Biopharma Therapeutics and Platforms IPO Activity – Q2 2025 Review