Biopharma and broader healthcare and life sciences R&D partnership activity continued its earlier slowdown, though not without major deals that marked the year in charts. SystImmune granted Bristol Myers Squibb exclusive rights to co-develop and co-commercialize BL-B01D1 for lung/breast cancer, involving an $800 million upfront payment, potential payments of up to $500 million, and up to $7.1 billion in milestones. Daiichi Sankyo exclusively licensed three antibody-drug conjugates to Merck for a $4 billion upfront payment, potential additional payments of $1.5 billion, and up to $16.5 billion in development and commercial milestones.
In a partnership between Nurix and Seagen, Nurix received a $60 million upfront payment and is eligible for milestones of up to $3.4 billion, and mid-single to low double-digit tiered royalties. Pfizer and Flagship Pioneering initiated research with $50 million initial investments from both, and Flagship is eligible for up to $700 million in combined milestones. Alnylam granted Roche exclusive rights for Zilebesiran, involving a $310 million upfront payment, potential milestones of up to $2.49 billion, and a 50% profit share in the U.S. Lastly, Voyager Therapeutics and Neurocrine Biosciences partnered on a $175 million upfront deal with potential milestones of up to $4.235 billion, and retaining U.S. profit split options.
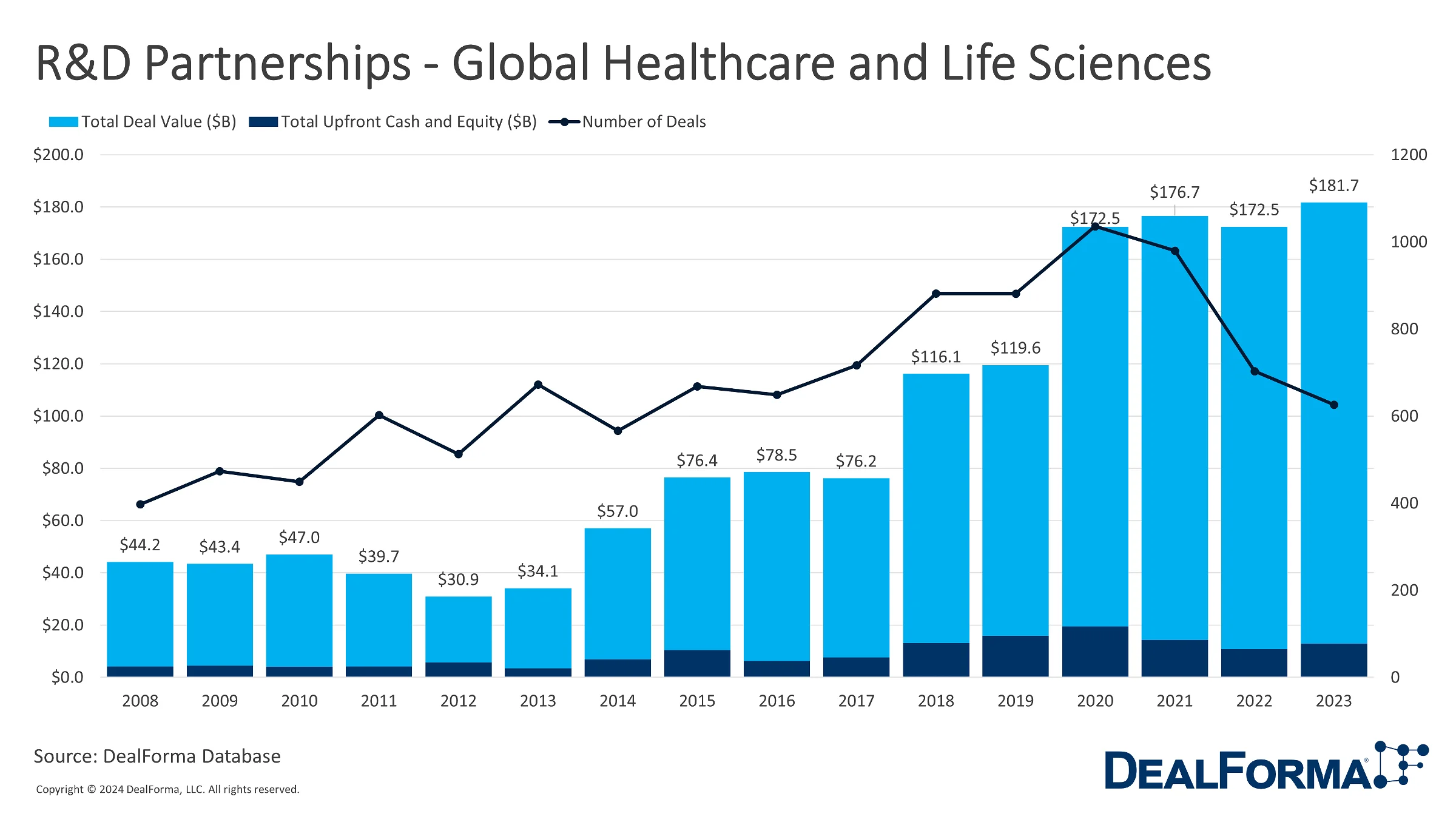
In 2023, 626 R&D partnership deals were announced, marking a slight decrease from the previous year’s 703 deals. However, the overall value of these deals experienced a significant surge thanks to the major deals signed this year, reaching $181.7 billion as opposed to $172.5 billion in 2022. At the same time, the total upfront cash and equity in these partnerships increased from $10.9 billion in 2022 to $13.0 billion in 2023, again taking into account a few record-breaking deals. While examining the last 3 years, the cumulative figures portray a substantial number of partnerships (2,309 deals) with a total deal value of $530.8 billion and upfront cash and equity totaling $38.2 billion.
Biopharma Therapeutics And Platforms Redefining R&D Partnerships For 2023
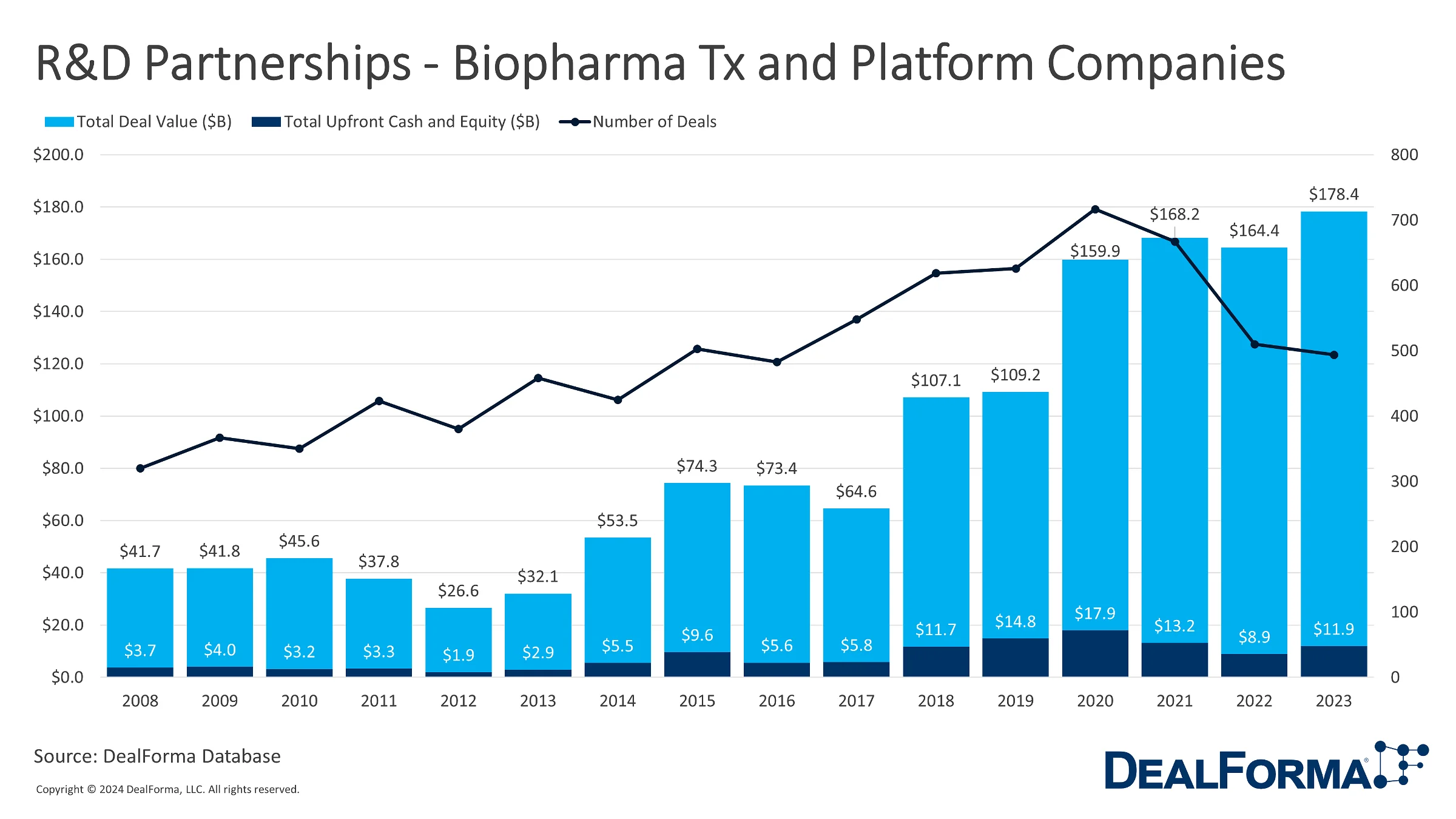
DealForma data on biopharma licensing deals for therapeutics and discovery platforms tracks the 494 announced deals, showing a slight dip from the 510 deals in 2022. However, the total value of these deals saw a significant increase, reaching $178.4 billion compared to the previous year’s $164.4 billion. Additionally, these partnerships’ overall upfront cash and equity increased from $8.9 billion in 2022 to $11.9 billion in 2023. Over the three years, 1,671 deals were announced, resulting in a cumulative deal value of $511.0 billion, with upfront cash and equity totaling $33.9 billion.
R&D Partnerships At Every Stage – Highlights From 2023
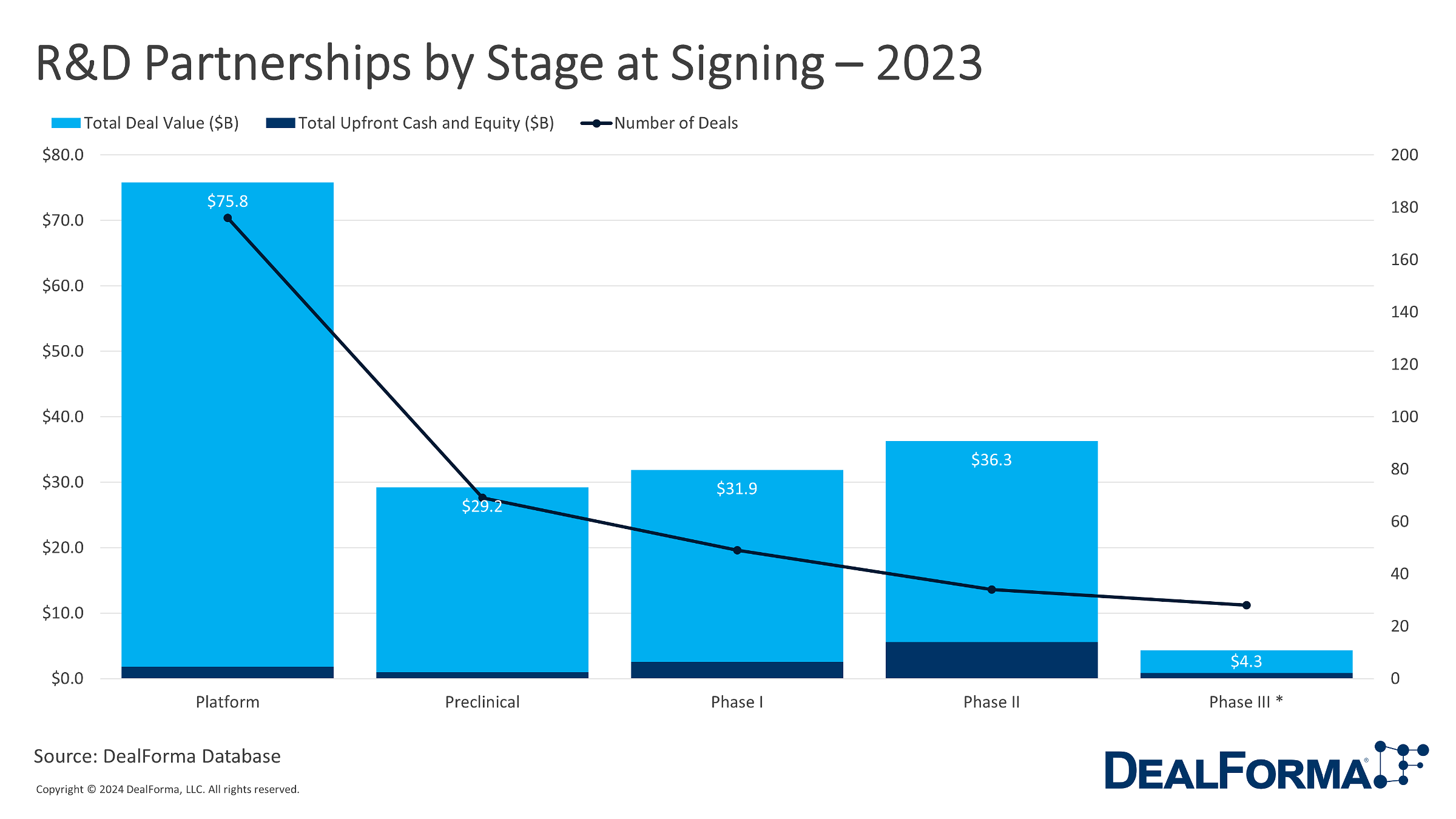
A look at biopharma R&D licensing and partnership deal values by stage at signing shows the amount of deal activity for discovery platforms. In 2023, 176 deals contributed to a total deal value of $75.8 billion, with upfront cash and equity amounting to $1.8 billion for deals signed at discovery and platform stages. Following this, preclinical partnerships comprised 69 deals with a total deal value of $29.2 billion and upfront cash and equity of $900 million. Partnerships at clinical stages, particularly Phase I, II, and III, demonstrated substantial values, reaching $31.9 billion, $36.3 billion, and $4.3 billion, respectively.
Leading Modalities In Biopharma R&D Partnerships – 2023
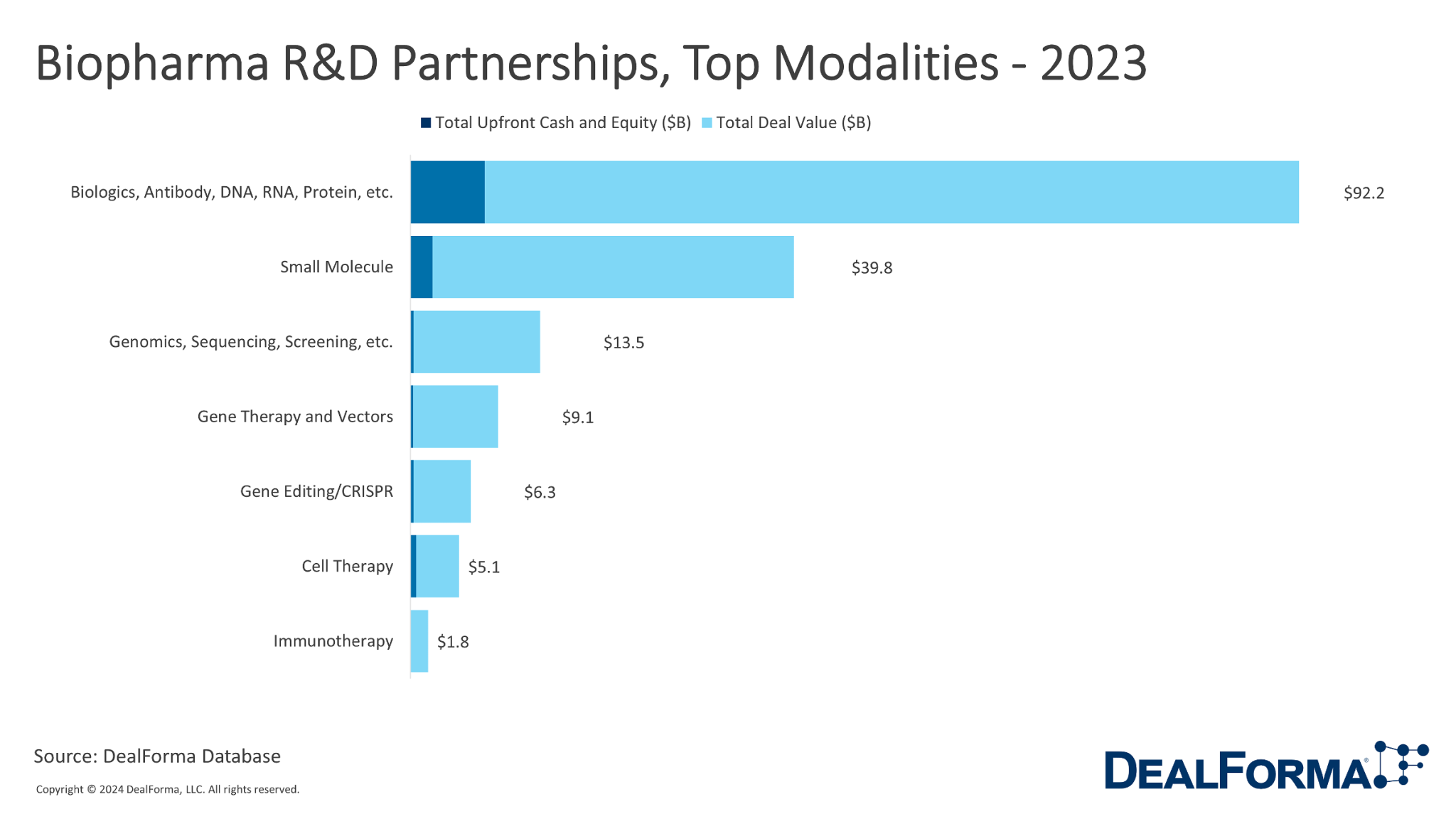
DealForma data on biopharma R&D partnerships in 2023 classifies partnerships based on primary technology and sheds light on the prevailing modalities within the industry. The predominant modality was still biologics, covering antibodies, DNA, RNA, proteins, and more grouped here for simplicity and available in detail in the DealForma database. This accounted for 186 deals, contributing to a substantial total deal value of $92.2 billion and upfront cash and equity of $7.7 billion. Following closely were small molecule partnerships, comprising 118 deals with a total deal value of $39.8 billion and upfront cash and equity of $2.3 billion.
Genomics, sequencing, screening, gene therapy, vectors, and gene editing/CRISPR played significant roles, each with varying deal numbers and values. Despite having fewer deals, Cell Therapy and Immunotherapy demonstrated their importance in the industry, contributing to the overall partnership landscape. When considering all modalities collectively, there were 434 deals, amounting to a total deal value of $167.6 billion, with upfront cash and equity totaling $11.6 billion.
Top Therapy Areas In Biopharma R&D Partnerships For 2023
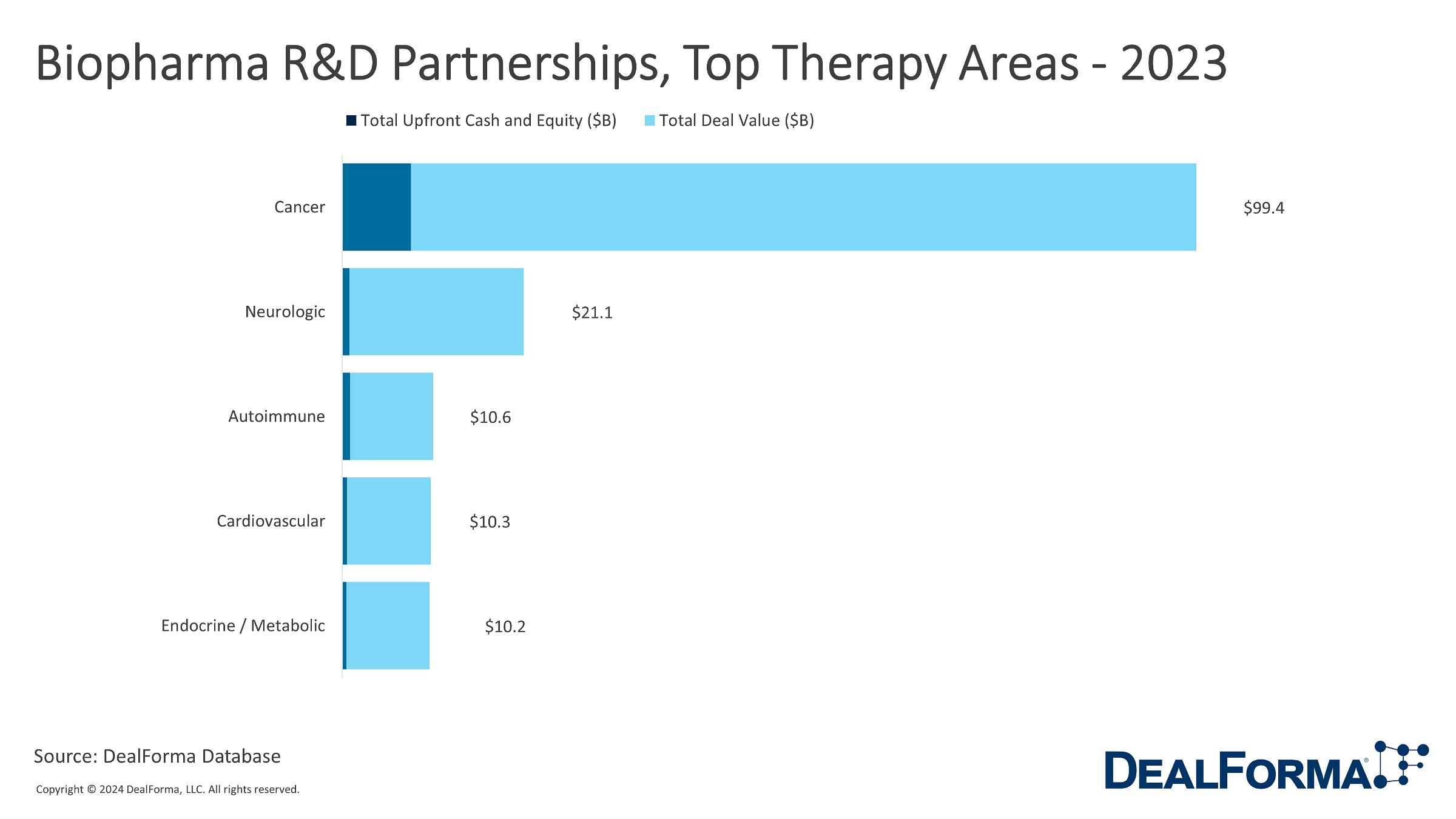
Biopharma R&D partnerships in 2023, categorized by primary therapy area, provide insights into the industry’s focus areas of collaborative efforts. Cancer remained the dominant therapy area, with 178 deals contributing to a substantial total deal value of $99.4 billion and upfront cash and equity of $8.0 billion. Other therapy such as neurologic, autoimmune, cardiovascular, and endocrine/metabolic, also witnessed considerable partnership activity, each with varying deal numbers and values. Despite having fewer deals, ophthalmic, infectious, and inflammation therapy areas played significant roles in the partnership landscape. When considering therapy areas collectively, there were 842 deals, resulting in a total deal value of $164.0 billion, with upfront cash and equity totalling $11.5 billion.
Impactful Partnerships Shaping Biopharma R&D In 2023
SystImmune and Bristol Myers Squibb Partnership – (December 2023)
SystImmune granted Bristol Myers Squibb (BMS) exclusive rights to co-develop and co-commercialize BL-B01D1 for lung and breast cancer treatment. Key figures include an $800 million upfront payment to SystImmune, potential near-term payments of up to $500 million, and up to $7.1 billion in development, regulatory, and commercial milestones. Tiered royalties apply outside the U.S., and BMS is eligible for royalties in Mainland China where SystImmune retains.
Daiichi Sankyo and Merck Partnership – (October 2023)
Daiichi Sankyo exclusively licensed 3 antibody-drug conjugates (ADCs) to Merck for worldwide co-development and co-commercialization, excluding Japan. Key numbers include an upfront payment of $4 billion for Daiichi, potential additional payments of $1.5 billion at 12 and 24 months, and up to $16.5 billion in potential development and commercial milestones. Merck will handle 75% of the initial $2 billion in R&D costs, and royalties apply to Japanese sales.
Nurix and Seagen Cancer Therapies Partnership – (September 2023)
Nurix granted Seagen exclusive rights for multiple cancer therapies using targeted protein degradation technology. Key figures include a $60 million upfront payment for Nurix, potential milestones of up to $3.4 billion, and mid-single to low double-digit tiered royalties. Nurix has the option for U.S. profit-sharing and co-promotion on two products.
Pfizer and Flagship Pioneering Research Partnership – (July 2023)
Pfizer entered a research partnership with Flagship Pioneering, obtaining an exclusive global option to license 10 single-asset therapies. Initial investments from both companies amount to $50 million each, with Flagship eligible for up to $700 million in combined development and commercial milestones for 10 programs. The agreement also includes undisclosed royalties.
Alnylam and Roche Partnership for Zilebesiran – (July 2023)
Alnylam granted Roche exclusive worldwide rights for Zilebesiran, an RNAi therapy for hypertension. Key numbers comprise a $310 million upfront payment for Alnylam, potential milestones of up to $2.49 billion, and a 50% profit share in the U.S. Outside the U.S., Alnylam receives low double-digit royalties on net sales.
Voyager Therapeutics and Neurocrine Biosciences Partnership – (January 2023)
Neurocrine Biosciences gained exclusive worldwide rights for the GBA1 program from Voyager Therapeutics. Key numbers involve an upfront payment of $175 million for Voyager, potential development and commercial milestones of up to $4.235 billion, and tiered royalties on net sales. Voyager retains the option for U.S. profit-sharing and co-promotion on two resulting products.