H1 2025 autoimmune dealmaking remained strong, led by biologics and boosted by AI-driven discovery, multi-target platforms, and targeted delivery, with large pharma focusing on late-stage assets via selective M&A and milestone-heavy partnerships. On the R&D front, five high-value collaborations stood out, RemeGen–Vor Bio (June 2025, $125 million upfront/stock warrants plus over $4 billion in potential milestones), Syneron Bio–AstraZeneca (March 2025, up to $75 million near-term and $3.4 billion in milestones), Earendil Labs–Sanofi (April 2025, $175 million near-term and $1.7 billion in milestones), WuXi Biologics Candid (January 2025, up to $925 million), and BridGene–Takeda (February 2025, up to $816 million).
M&A activity was equally strategic, AbbVie–Capstan (June 2025, up to $2.1 billion cash), Alumis Acelyrin (February–May 2025, $737 million all-stock), Paratek–Optinose (March–May 2025, up to $330 million), Nerva–Mahana (May 2025, undisclosed), and Scinai–Pincell option deal (March 2025, undisclosed). Collectively, these transactions highlight a dual-track trend large pharmas bolstering cell, gene, and antibody platforms, while digital health and targeted delivery technologies gain momentum as differentiated growth drivers in the autoimmune.
Autoimmune R&D Partnerships by Tech Grouping – H1 2025
Autoimmune R&D activity remained strong in H1 2025, with 21 partnerships totaling $14.7 billion. Biologic platforms, including antibodies, nucleic acid therapies, and proteins, led the field, accounting for nearly 80% of the total deal value ($11.3 billion) and $345 million in upfront payments. Genomics and sequencing-based collaborations followed with six deals worth $1.8 billion and $125 million upfront. Small molecule programs secured three deals totaling $816 million, while two immunotherapy partnerships added $765 million. Overall, upfront payments reached $551 million.
Top Autoimmune R&D Partnerships – H1 2025
RemeGen development and commercialization deal with Vor Bio – June 2025
Vor Bio secured exclusive global rights (ex-China, Hong Kong, Macau, Taiwan) to develop and commercialize telitacicept, a dual-target fusion protein (BlyS/BAFF and APRIL) for generalized myasthenia gravis (gMG). Already approved in China for gMG, SLE, and RA, the drug is in a global Phase III trial for gMG. RemeGen will receive $45 million upfront and $80 million in stock warrants, with potential for over $4 billion in milestones plus tiered royalties. The structure underscores high-value interest in late-stage autoimmune assets with established regional approvals.
Syneron Bio development and commercialization deal with AstraZeneca – March 2025
AstraZeneca gained rights to develop and commercialize macrocyclic peptides from Syneron Bio’s Synova platform for chronic conditions, including rare, autoimmune, and metabolic diseases. Syneron Bio will receive up to $75 million in upfront and near-term milestone payments, plus an undisclosed equity investment. The agreement includes up to $3.4 billion in potential development and commercial milestones, along with tiered royalties highlighting the strategic value of protein-based discovery platforms in addressing high-burden disease areas.
Earendil Labs development and commercialization deal with Sanofi – April 2025
Sanofi secured exclusive global rights to HXN-1002 and HXN-1003, bispecific antibodies discovered via Earendil Labs’ AI-enabled, high-throughput platform for autoimmune and inflammatory bowel diseases, including Crohn’s disease and colitis. Earendil Labs will receive $125 million upfront and up to $50 million in near-term milestones, with total potential development and commercial milestones reaching $1.7 billion, plus tiered royalties from the high single to low double digits. The deal reflects a growing appetite for AI-driven antibody discovery in high-need autoimmune indications.
WuXi Biologics development and commercialization deal with Candid – January 2025
Candid obtained exclusive global rights to preclinical tri-specific T-cell engager antibodies targeting BCMA, CD20, and CD19, developed via WuXi Biologics’ WuXiBody platform for autoimmune and inflammatory diseases. IND-enabling studies are planned for H1 2026. WuXi Biologics could receive up to $925 million in upfront, development, and commercial milestone payments, plus royalties, underscoring the strategic interest in multi-target antibody platforms for next-generation autoimmune therapies.
BridGene development and commercialization deal with Takeda – February 2025
Takeda obtained exclusive rights to develop and commercialize small molecule therapies discovered via BridGene’s IMTAC chemoproteomics platform for autoimmune and neurological diseases. BridGene will receive up to $46 million in upfront and preclinical milestone payments, with potential for $770 million in clinical and commercial milestones, plus tiered royalties. The collaboration highlights growing interest in proteomics-driven small molecule discovery for immunology and neurology.
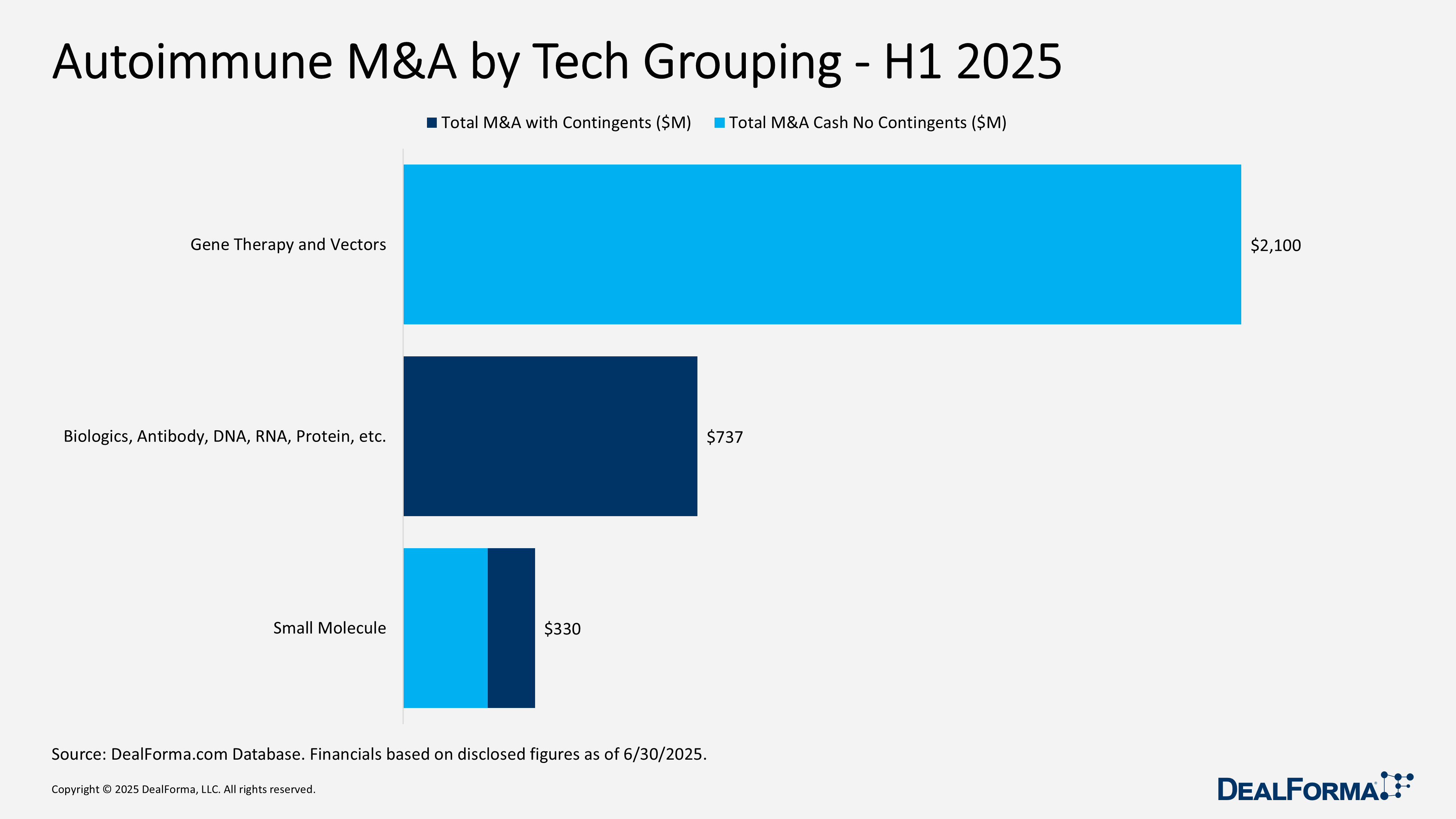
Autoimmune M&A by Tech Grouping – H1 2025
Autoimmune M&A activity was limited but high in value in H1 2025, with just five deals totaling $3.2 billion. Notably, a single gene therapy acquisition accounted for $2.1 billion, all paid upfront highlighting strong interest in potentially curative treatments. Biologic platform deals followed, totaling $737 million across two acquisitions, though both were structured entirely around future milestones, reflecting buyer caution with early-stage assets. Two small-molecule acquisitions added $330 million, including $212 million upfront, indicating ongoing but modest investment in traditional approaches. Upfront payments made up 73% of total deal value, primarily driven by the gene therapy transaction.
Top Autoimmune M&A – H1 2025
AbbVie acquiring Capstan – June 2025
AbbVie agreed to acquire Capstan for up to $2.1 billion in upfront cash (subject to adjustments), gaining its targeted lipid nanoparticle (tLNP) platform for in vivo delivery of RNA payloads, including mRNA, to engineer specific cell types. Capstan’s pipeline includes Phase I CPTX-2309, an in vivo tLNP anti-CD19 CAR-T therapy for B cell-mediated autoimmune diseases, CPTX-2506 for BCMA-expressing plasma cells in cancer and autoimmune indications, and additional early-stage programs in immunology, oncology, and fibrosis. The acquisition strengthens AbbVie’s position in next-generation cell and gene therapies for autoimmune and cancer markets.
Nerva (Mindset Health) acquiring Mahana Therapeutics – May 2025
Nerva acquired Mahana Therapeutics, a private developer of digital therapeutics for gastrointestinal diseases, including IBS. The acquisition integrates Mahana’s CBT-based treatment content into Nerva’s hypnotherapy app, enhancing its brain–gut health portfolio for IBD and gastrointestinal cancer-related care. Financials were undisclosed.
Scinai option to acquire Pincell – March 2025
Scinai Bioservices (a unit of Scinai Immunotherapeutics) secured an option to acquire Pincell, whose lead program, PC-111, is a preclinical mAb targeting the Fas/FasL pathway for rare dermatologic diseases, including pemphigus vulgaris, Stevens–Johnson syndrome, and toxic epidermal necrolysis. PC-111 holds EMA orphan drug designation for pemphigus, with FDA orphan and breakthrough designation filings planned. Development has been transferred to a Scinai subsidiary, supported by a joint application for non-dilutive European funding. Financials were undisclosed.
Paratek acquiring Optinose to take the company private – March 2025
Paratek completed the acquisition of Optinose, taking the company private for up to $330 million. The deal includes $9 per share in cash (50% one-day premium) and up to $5 per share in contingent value rights (CVRs) tied to sales milestones for Xhance (fluticasone propionate), $1 per share if annual sales reach $150 million before 2028, and an additional $4 per share if sales hit $225 million by 2029. Optinose’s portfolio includes approved Xhance, a drug–device therapy for chronic rhinosinusitis, and Onzetra Xsail, a nasal powder formulation of sumatriptan for migraines. Optinose shares were delisted from NASDAQ following the deal close in May 2025.
Alumis acquiring Acelyrin in an all-stock transaction – February 2025
Alumis acquired Acelyrin in an all-stock transaction valued at approximately $737 million, forming a combined clinical-stage biopharma focused on immune-mediated diseases. The merger closed in May 2025, with Alumis shareholders owning 52% and Acelyrin shareholders 48% of the new entity. Acelyrin’s lead asset, lonigutamab, is a Phase II IGF-1 receptor antagonist for thyroid eye disease. The deal expands Alumis’ late-stage pipeline to include ESK-001, lonigutamab, and A-005. Acelyrin now operates as a wholly owned subsidiary, and its shares have been delisted from NASDAQ.
Also check out Autoimmune R&D Partnerships, M&A and Venture Funding – H1 2025 Review