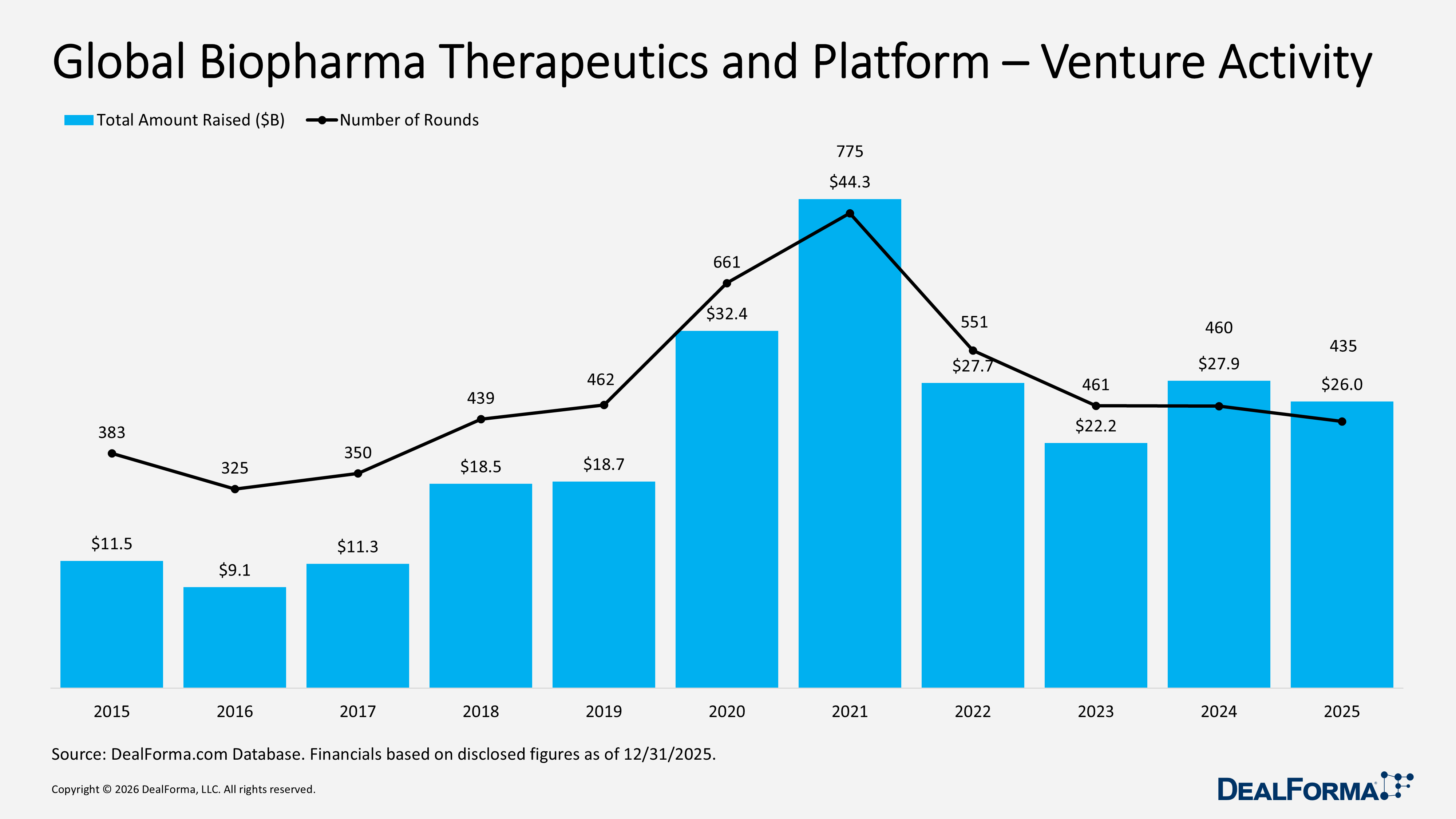
Biopharma therapeutics and platform venture funding dropped in 2025 compared to 2024, with 435 rounds raising $26B versus 460 rounds and $27.9B in 2024, while the average round size remained stable at $67M ($66M in 2024), bringing the two-year total to 895 rounds and $53.8B. The U.S. remained the dominant market ($18.5B, 256 rounds), followed by Europe ($5.4B, 123 rounds) and China and Hong Kong ($1.1B, 22 rounds), while major hubs SF Bay Area ($6.3B, 65 rounds) and Boston ($4.5B, 60 rounds) continued to anchor venture activity despite lower deal volumes. Funding remained concentrated in series A ($7.9B, 121 rounds) and series B ($6.6B, 82 rounds), with series D+ ($3B) and seed ($1.4B) gaining momentum.
By therapeutic area, cancer led funding ($7.7B, 138 rounds) followed by neurologic ($3.7B) and endocrine/metabolic and autoimmune ($2.4B each). In comparison, small molecules dominated modalities ($7.3B, 28%), ahead of AI/ML ($3.2B), antibodies ($3.2B), and gene therapy ($2.4B). Large financings continued to anchor the market, led by Kailera Therapeutics’ $600M series B (October 2025) to advance KAI-9531, a dual GLP-1/GIP obesity therapy in Phase 3. Verdiva Bio raised $410M series A (January 2025) to develop oral GLP-1 and amylin obesity therapies. Tubulis secured €344M ($401M) series C (October 2025) to advance ADC candidates TUB-040 and TUB-030 for solid tumors. MapLight Therapeutics completed a $373M series D (July 2025) supporting ML-007C-MA for schizophrenia and Alzheimer’s disease psychosis, while Eikon Therapeutics raised $351M series D (February 2025), bringing total private funding to over $1.1B to expand its advanced drug discovery platform.
In 2025, global biopharma therapeutics and platform venture funding remained firm, with 435 rounds raising $26B in total and an average of $67M per round, indicating similar support for large financings despite a slight decline in deal count. In 2024, the sector recorded 460 rounds raising $27.9B, with a similar $66M average round size. Combined, 2024 and 2025 generated 895 rounds totaling $53.8B.
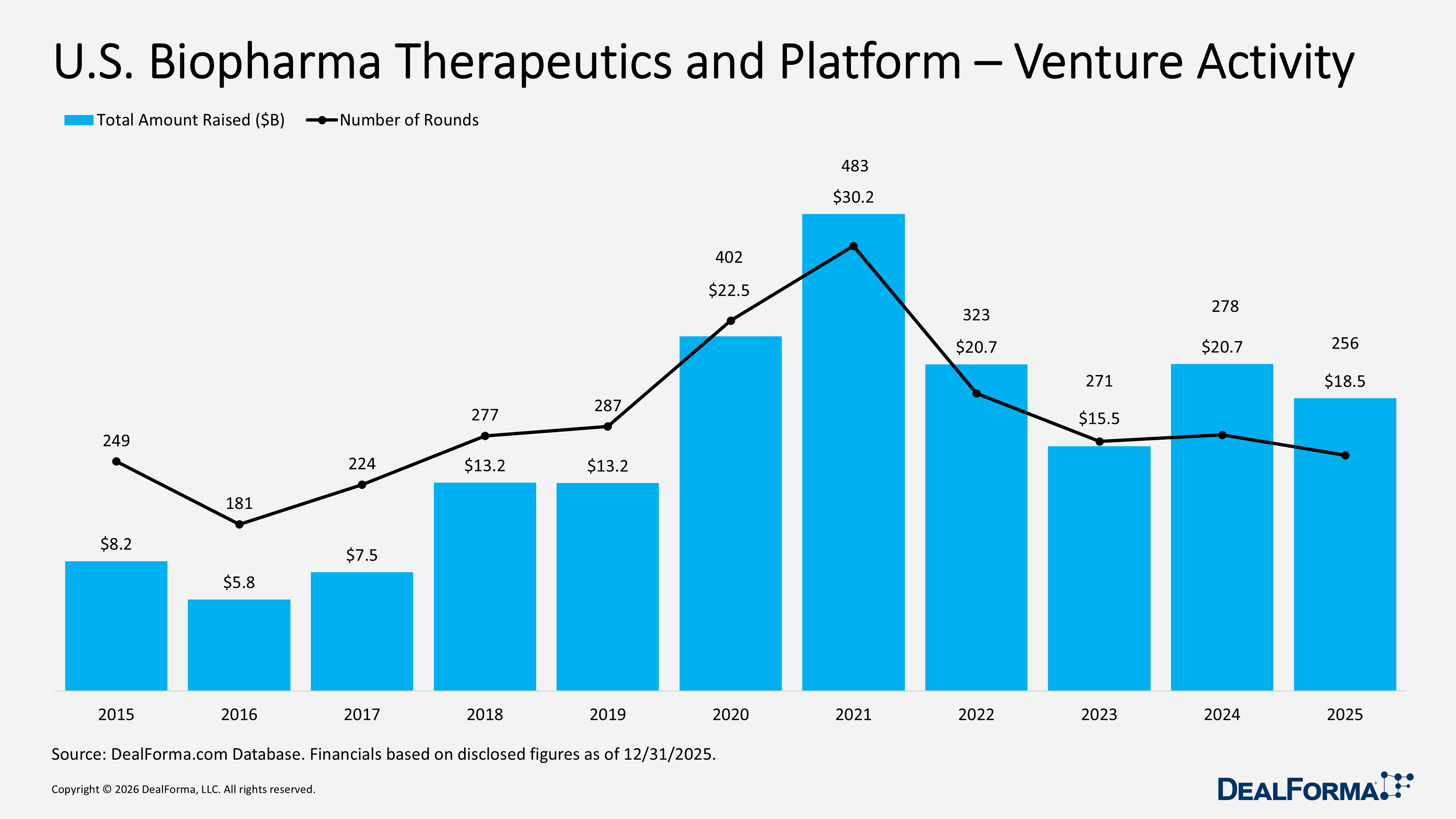
U.S. Biopharma Therapeutics and Platform Venture Activity
In 2025, U.S. biopharma therapeutics and platform venture funding declined from 2024 levels but remained robust, with 256 rounds raising $18.5B and an average of $80M per round. While deal volume and total capital eased, support was evident for relatively large financings. In 2024, activity was slightly stronger with 278 rounds raising $20.7B, and a comparable $81M average round size. Combined, 2024 and 2025 recorded 534 rounds totaling $39.2B in the U.S.
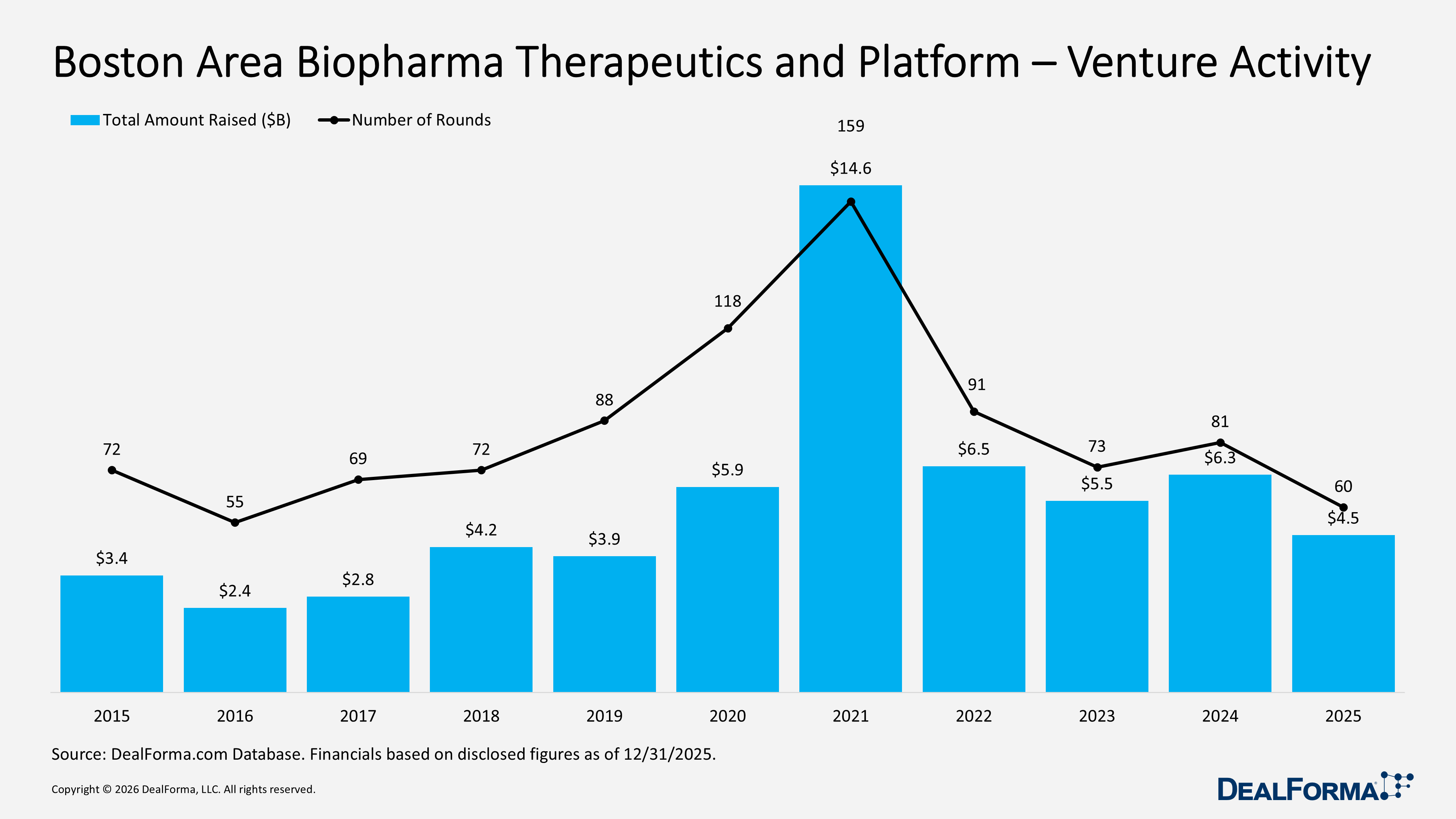
Boston Area Biopharma Therapeutics and Platform Venture Activity
In 2025, the Boston Area biopharma therapeutics and platform venture market declined from 2024 levels, with 60 rounds raising $4.5B, compared with 81 rounds and $6.3B in 2024, while the average round size remained stable at $84M versus $85M, signifying the slowdown was driven by fewer deals rather than weaker financing sizes. Across 2024 and 2025 combined, the region recorded 141 rounds totaling $11B, reinforcing Boston’s position as a leading biopharma funding hub.
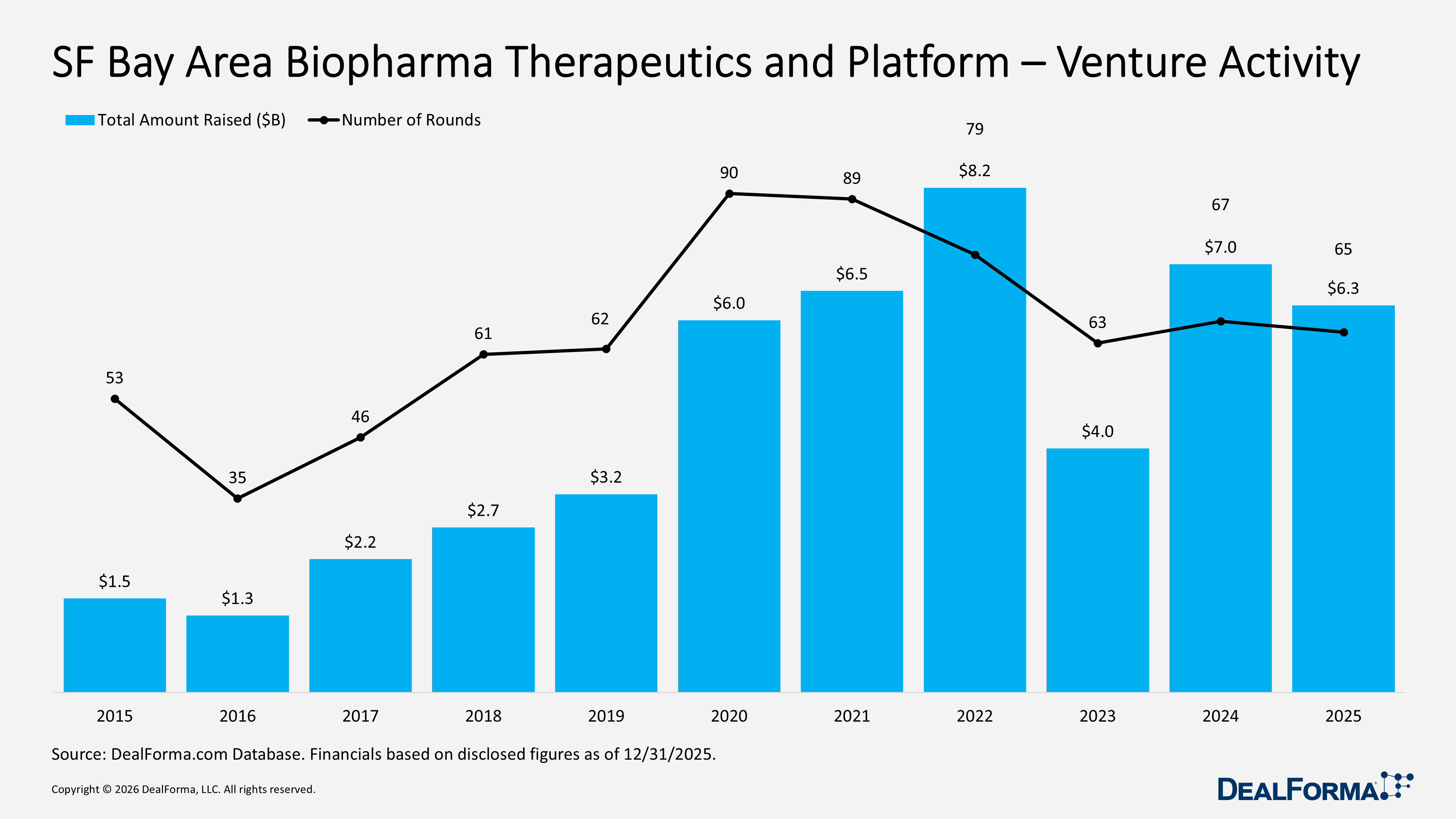
SF Bay Area Biopharma Therapeutics and Platform Venture Activity
In 2025, the San Francisco Bay Area, biopharma therapeutics and platform venture market declined slightly from 2024, with 65 rounds raising $6.3B, compared with 67 rounds and $7B in 2024, while the average round size fell to $101M from $116M, specifying the lower capital raised in 2025 was driven by smaller average checks rather than meaningful change in deal volume. Across 2024 and 2025 combined, the Bay Area generated 132 rounds totaling $13B, stressing sustained funding scale, with 2024 standing out as the year with higher total capital and larger average rounds.
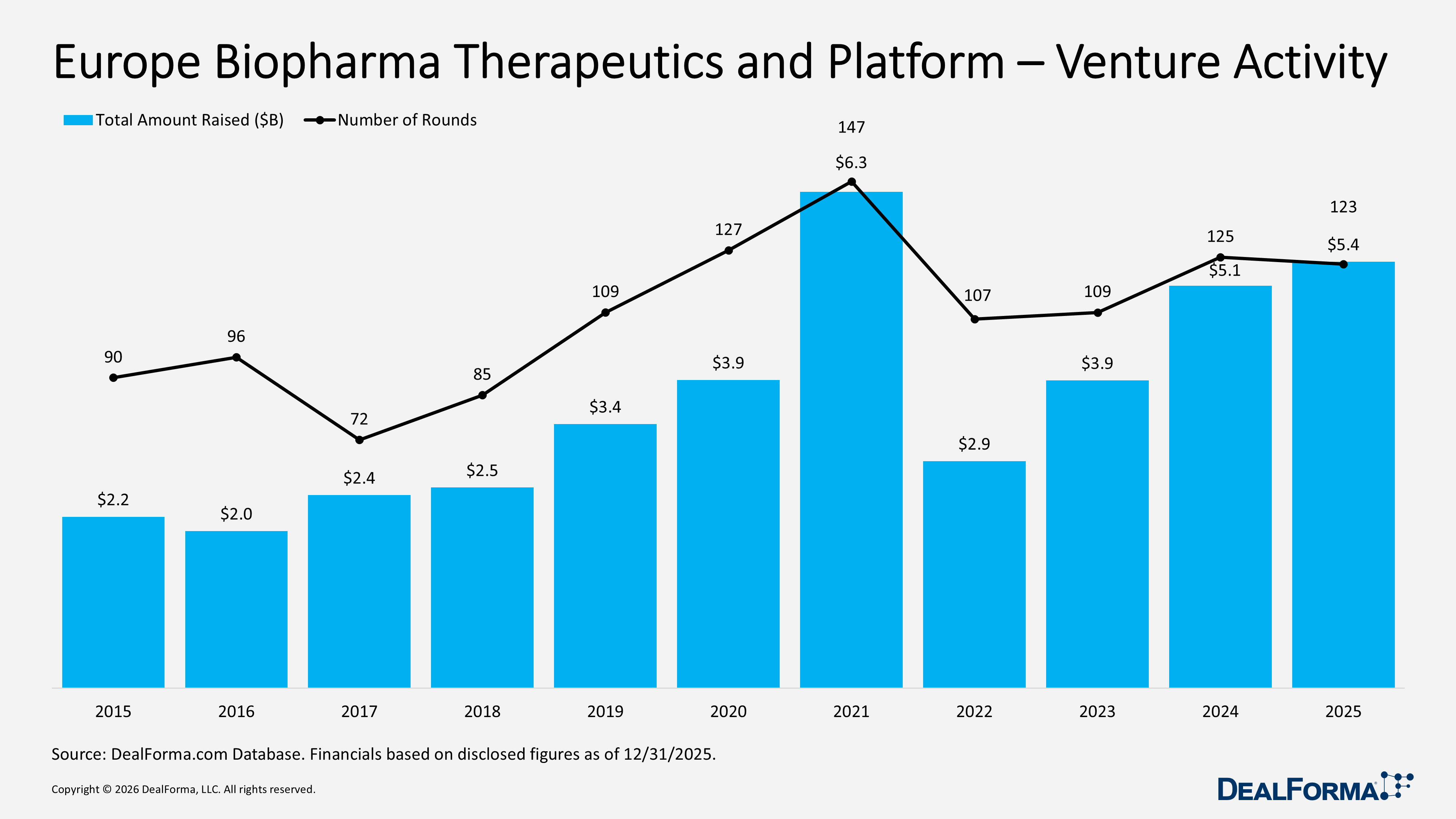
Europe Biopharma Therapeutics and Platform Venture Activity
In 2025, Europe’s biopharma therapeutics and platform venture market remained stable in deal activity with 123 rounds, while total funding increased to $5.4B, and the average round size rose to $50M, indicating slightly larger financings despite a marginal decline in deal count. In 2024, the region recorded 125 rounds raising $5.1B, with a smaller $43M average round size. Across 2024 and 2025, Europe generated 248 rounds totaling $10.5B.
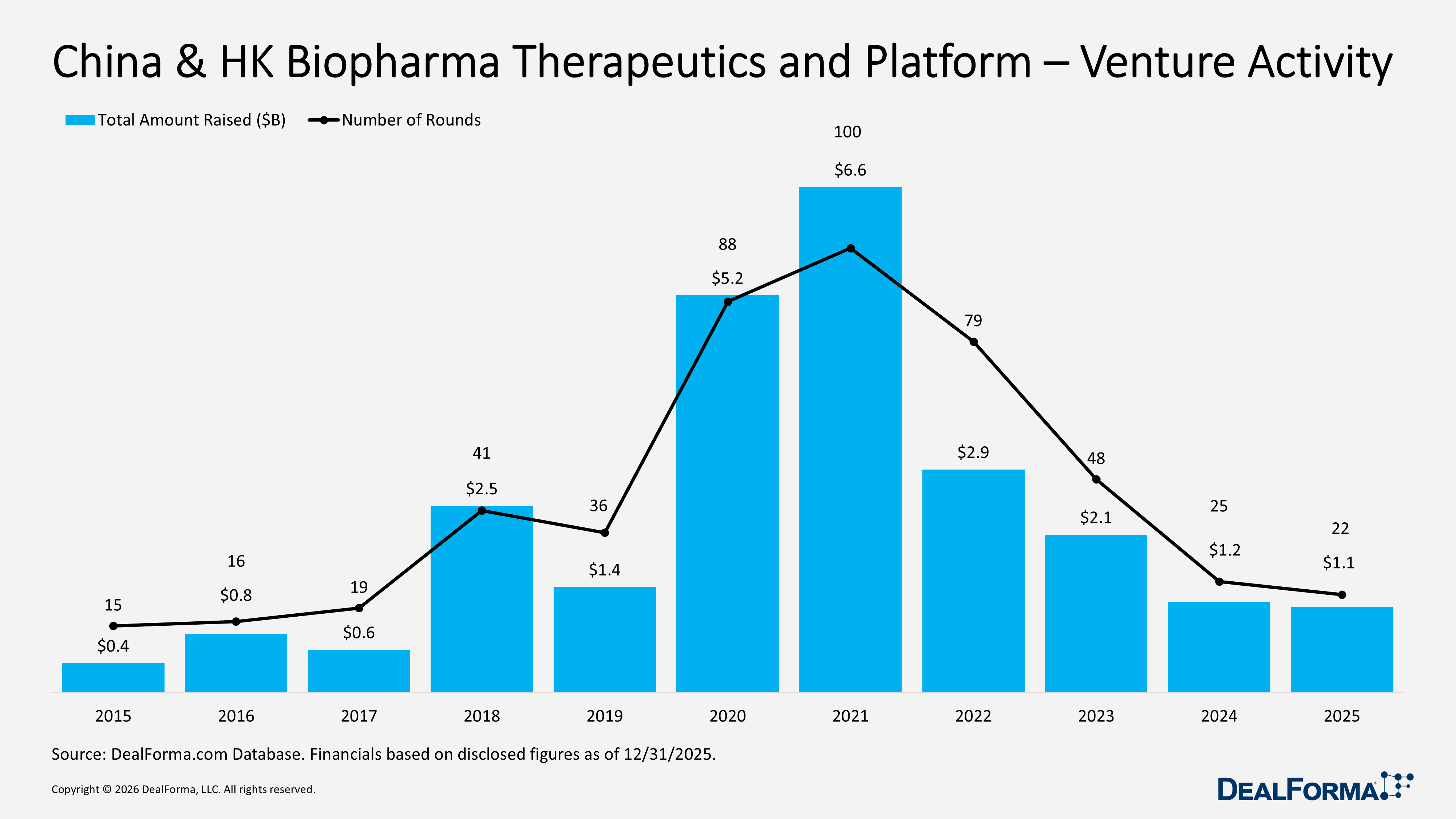
China & HK Biopharma Therapeutics and Platform Venture Activity
In 2025, China and Hong Kong biopharma therapeutics and platform venture funding declined from 2024 levels, with 22 rounds raising $1.1B, compared with 25 rounds and $1.2B in 2024, while the average round size fell to $59M from $63M, reflecting slightly lower deal activity and smaller financing. Across 2024 and 2025 combined, China & Hong Kong posted 47 rounds totaling $2.3B, emphasizing a smaller venture market compared to the U.S. and Europe.
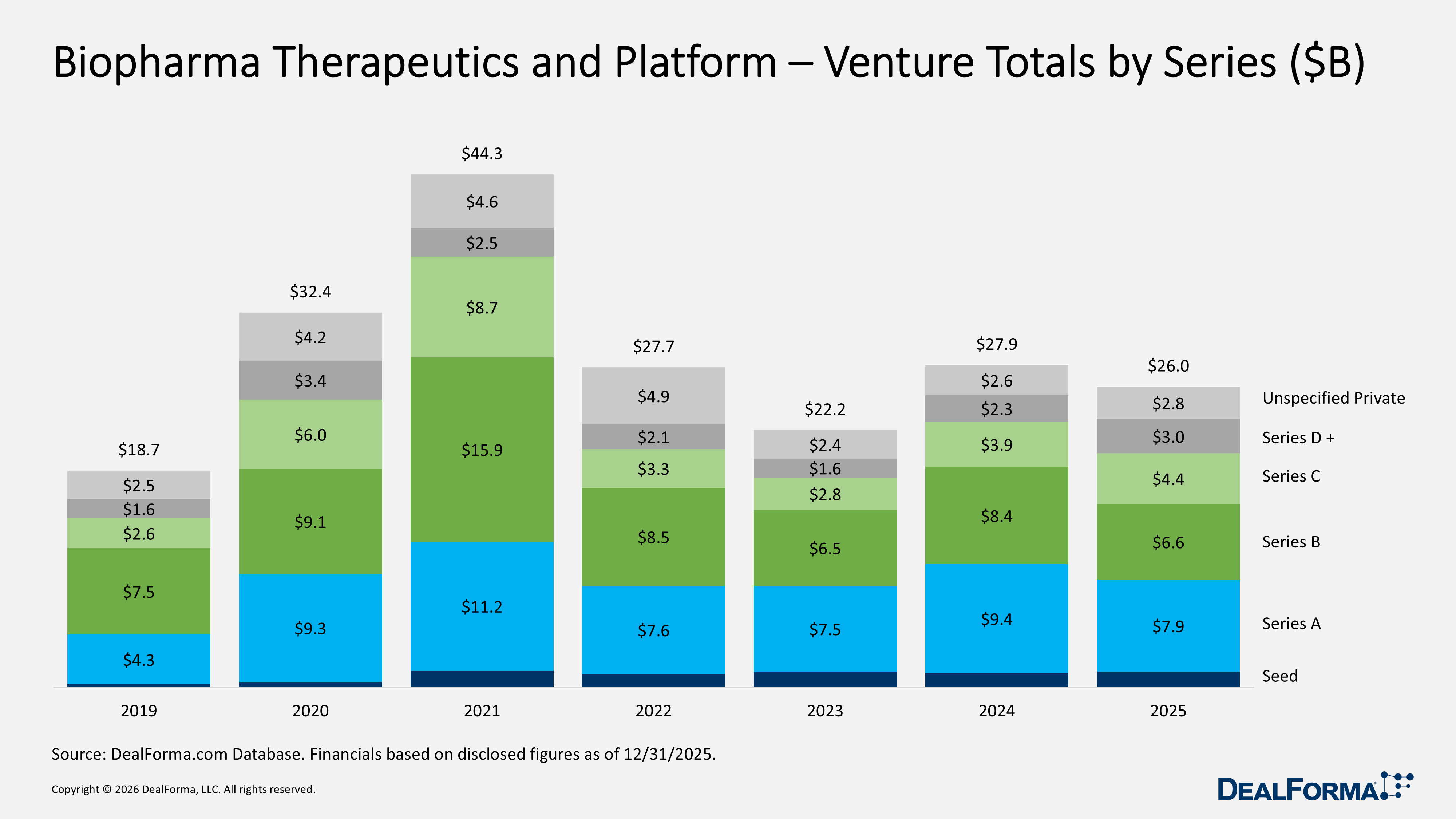
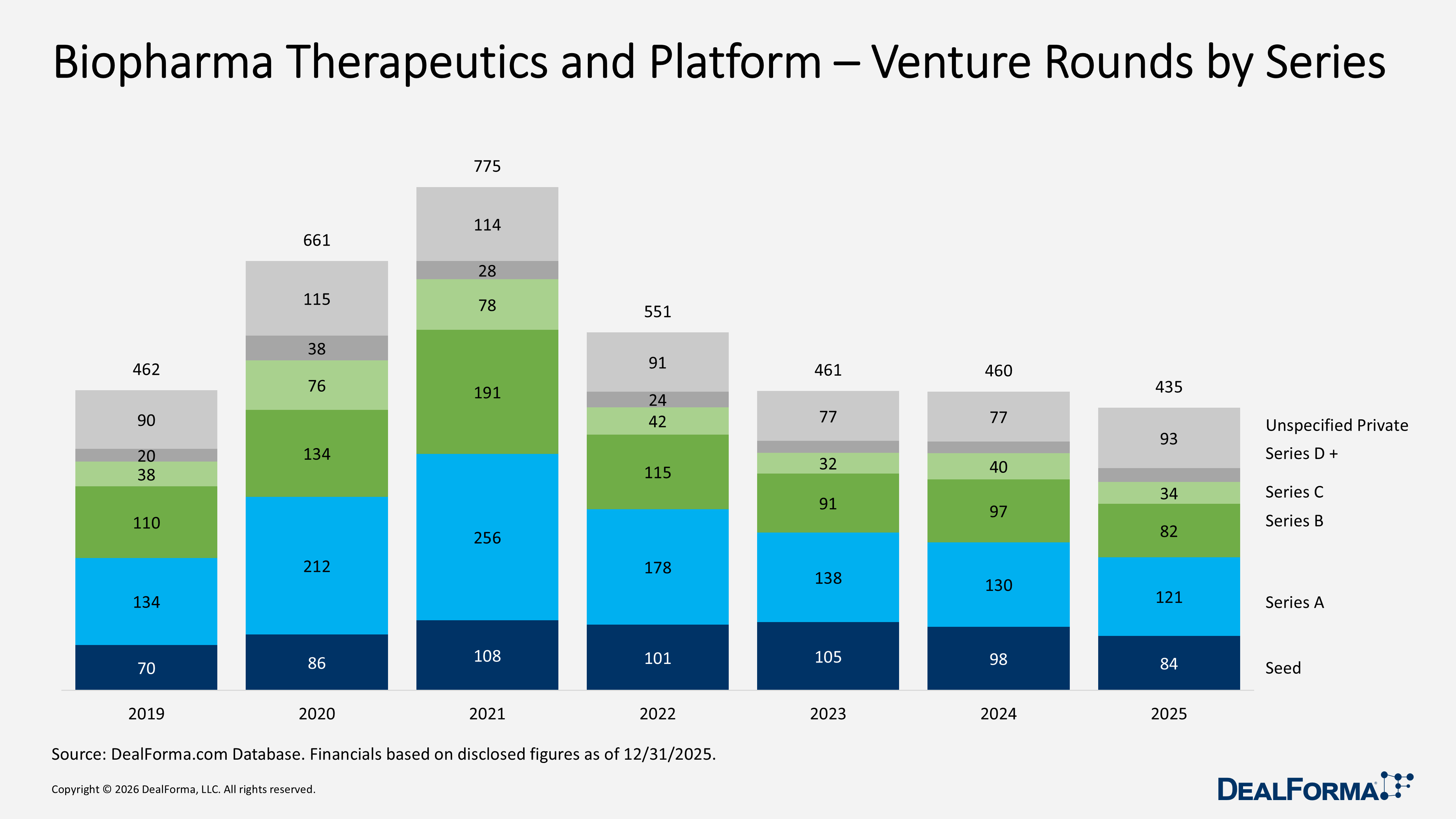
Biopharma Therapeutics and Platform – Venture Totals by Series
In 2025, biopharma therapeutics and platform venture funding totaled $26B across 435 rounds, with series A leading at $7.9B across 121 rounds and series B raising $6.6B from 82 rounds. Later-stage series D and beyond grew to $3B across 21 rounds, while seed funding increased to $1.4B despite fewer deals (84 rounds), signifying a trend toward concentrating more capital in selected early-stages. In 2024, total funding was slightly higher at $27.9B across 460 rounds, driven by larger series A ($9.4B, 130 rounds) and series B ($8.4B, 97 rounds) totals, while series C was lighter at $3.9B across 40 rounds and series D+ was smaller at $2.3B across 18 rounds.
In 2025, unspecified private rounds increased to 93, totaling $2.8B, up from 77 rounds and $2.6B in 2024, reflecting a rise in private financings not clearly categorized by stage. Across 2024 and 2025 combined, the sector recorded 895 rounds totaling $53.8B, with the largest capital allocations in series A ($17.3B) and series B ($15B), indicating that mid-stage rounds remain the main driver of funding, even as 2025 saw growing momentum in seed, later-stage, and unspecified private investments.
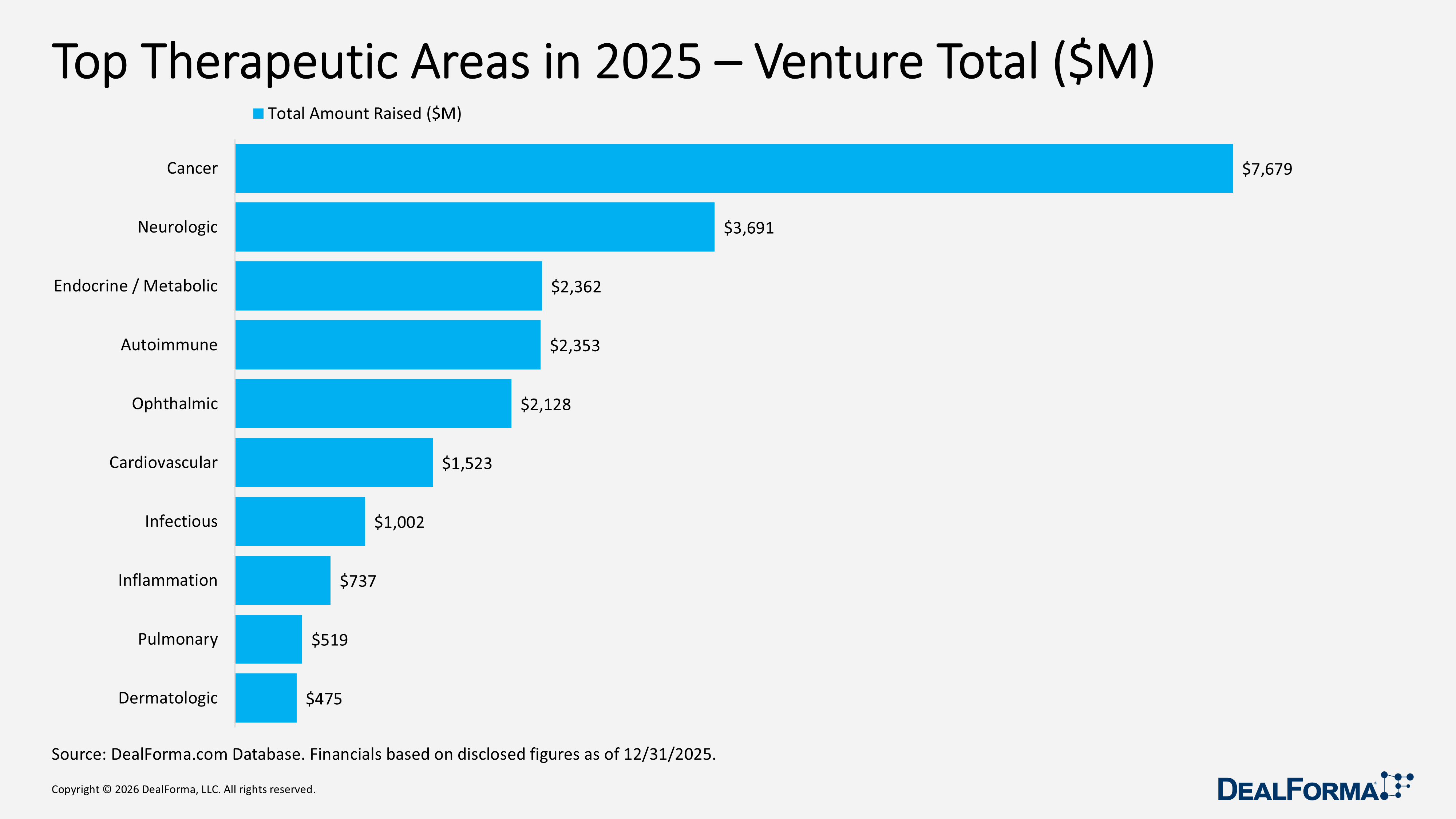
Top Therapeutic Areas in 2025 – Venture Totals
In 2025, venture funding by therapeutic area was dominated by cancer, which led both in deal count and capital, with 138 rounds raising $7.7B (about 30% of total funding) at an average of $61M per round. Neurologic followed with 70 rounds totaling $3.7B (14% share; $57M average). Endocrine/metabolic and autoimmune each raised roughly $2.4B (9% share), but with contrasting profiles: endocrine/metabolic had the largest average rounds at $118M across 24 deals, while autoimmune distributed capital over 34 rounds at $81M average. Other notable areas included ophthalmic with $2.1B across 22 rounds (8% share; $106M average), reflecting later-stage financings, and cardiovascular with $1.5B over 24 rounds ($76M average), showing steady mid-sized deals. Smaller active segments included infectious disease at $1B over 29 rounds ($36M average), and inflammation, pulmonary, and dermatologic funding at $737M and $475M (2-3% share; $47-$53M averages).
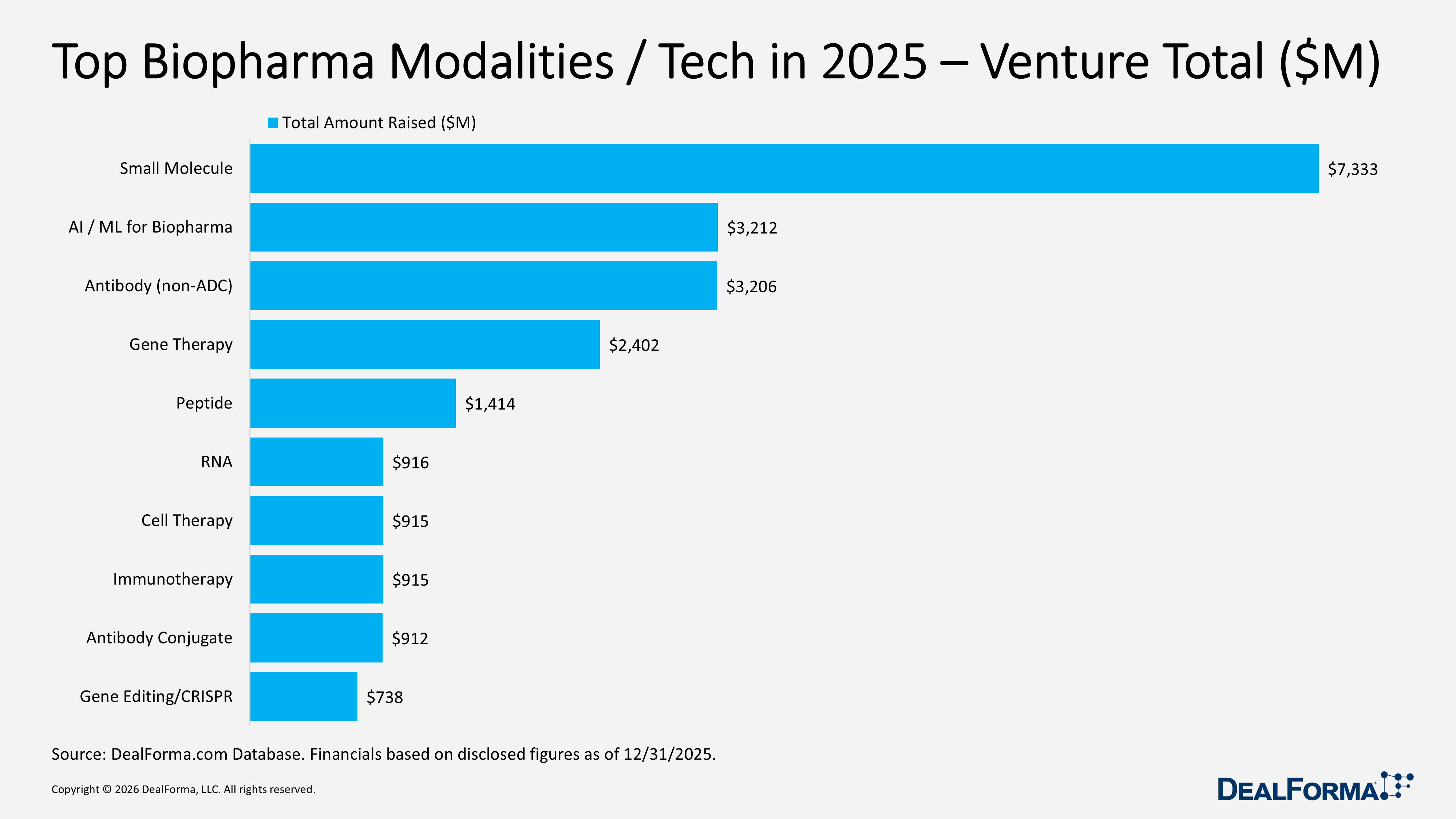
Top Biopharma Modalities / Tech in 2025 – Venture Totals
In 2025, venture funding by biopharma modality was led by small molecules, which combined the highest deal flow (130 rounds) with the largest capital pool ($7.3B, 28% of total) at an average of $62M per round. The next tier included AI/ML for biopharma (52 rounds, $3.2B, 12%, $68M average) and non-ADC antibodies (51 rounds, $3.2B, 12%, $67M average). Gene therapy followed with $2.4B across 27 rounds (9%) and a higher $92M average, indicating fewer but larger financings. Certain modalities stood out for high average round sizes despite smaller deal counts, including antibody-drug conjugates (ADCs) at $912M across 8 rounds, $114M average, and peptides at $1.4B over 16 rounds, $109M average. The mid-to-long tail included RNA ($916M, 16 rounds, $61M average), cell therapy ($915M, 25 rounds, $48M average), immunotherapy ($915M, 11 rounds, $83M average), and gene editing/CRISPR ($738M, 10 rounds, $82M average).
Prominent Biopharma Therapeutics and Platforms Venture Funding in 2025
Kailera Therapeutics – Series B – $600M – October 2025
Kailera Therapeutics, Inc., a clinical-stage biopharma focused on next-generation obesity therapies, raised $600M in a series B round led by Bain Capital Private Equity, fully funded at closing. The proceeds will advance its obesity portfolio, including the global Phase 3 program for lead candidate KAI-9531, a dual GLP-1/GIP receptor agonist with potential best-in-class weight loss. The round also included new investors such as Adage Capital, CPP Investments, Invus, Janus Henderson, Perseverance Capital, QIA, Royalty Pharma, Surveyor Capital, and T. Rowe Price accounts, alongside existing investors Atlas Venture, Bain Capital Life Sciences, RTW Investments, and Sirona Capital, reflecting broad institutional support.
Verdiva Bio – Series A – $410M – January 2025
Verdiva Bio UK-based clinical-stage biopharmaceutical company focused on obesity and cardiometabolic disorders, has raised $410M in an oversubscribed series A round, co-led by Forbion and General Atlantic, with participation from RA Capital Management, OrbiMed, Logos Capital, Lilly Asia Ventures, and LYFE Capital. The funding will advance Verdiva’s pipeline of next-generation oral and injectable therapies, including a Phase II-ready oral GLP-1 receptor agonist (ecnoglutide), a potential first-in-class oral amylin agonist, and a long-acting subcutaneous amylin agonist, all licensed from China-based Sciwind Biosciences for global rights outside Greater China and South Korea. The programs aim for first-in-class or best-in-class status, targeting weight loss and maintenance using proprietary oral delivery technology to improve dosing convenience, scalability, and patient access.
Tubulis – Series C – $401M – October 2025
Tubulis completed the second and final closing of its series C round totaling €344M (US $401M), led by Venrock Healthcare Capital Partners with new investors Fidelity, Janus Henderson, Blackstone Multi-Asset Investing, Wellington Management, and Ascenta Capital, alongside existing backers including Nextech Invest, EQT Life Sciences, Frazier Life Sciences, Andera Partners, Deep Track Capital, Bayern Kapital, Fund+, HTGF, OCCIDENT, and Seventure Partners. The funding will support pivotal trials for the lead ADC candidate, TUB-040; explore earlier treatment lines in ovarian cancer; expand combination regimens and new solid tumor indications; and advance the broader pipeline, including clinical-stage TUB-030 and preclinical programs, while further developing proprietary ADC platform technologies.
MapLight Therapeutics – Series D – $372.5M – July 2025
MapLight Therapeutics raised $373M in an oversubscribed series D financing, co-led by Forbion and Life Sciences at Goldman Sachs Alternatives, with participation from Sanofi, T. Rowe Price Investment Management accounts, and Avego BioScience Capital, alongside existing investors including Novo Holdings, 5AM Ventures, and Blue Owl Healthcare Opportunities. The proceeds will support the development of ML-007C-MA, an oral fixed-dose combination targeting M1 and M4 muscarinic receptors, advancing ongoing Phase 2 trials for schizophrenia and Alzheimer’s disease psychosis, while funding additional indications and broader clinical and preclinical programs.
Eikon Therapeutics – Series D – $350.7M – February 2025
Eikon Therapeutics, a pivotal-stage biotechnology company focused on integrating engineering, molecular biology, and data science to accelerate drug discovery, announced the initial closing of a $351M series D financing. Since its founding in 2019, the company has raised more than $1.1B in private funding. The round was led by existing investors with participation from new backers, including Lux Capital, Alexandria Venture Investments, AME Cloud Ventures, The Column Group, E15 VC, Foresite Capital, General Catalyst, Soros Capital, StepStone Group, T. Rowe Price Associates accounts, and UC Investments, reflecting strong institutional support for Eikon’s next-generation drug discovery platform.
Also check out Biopharma Therapeutics and Platforms M&A – 2025 Review