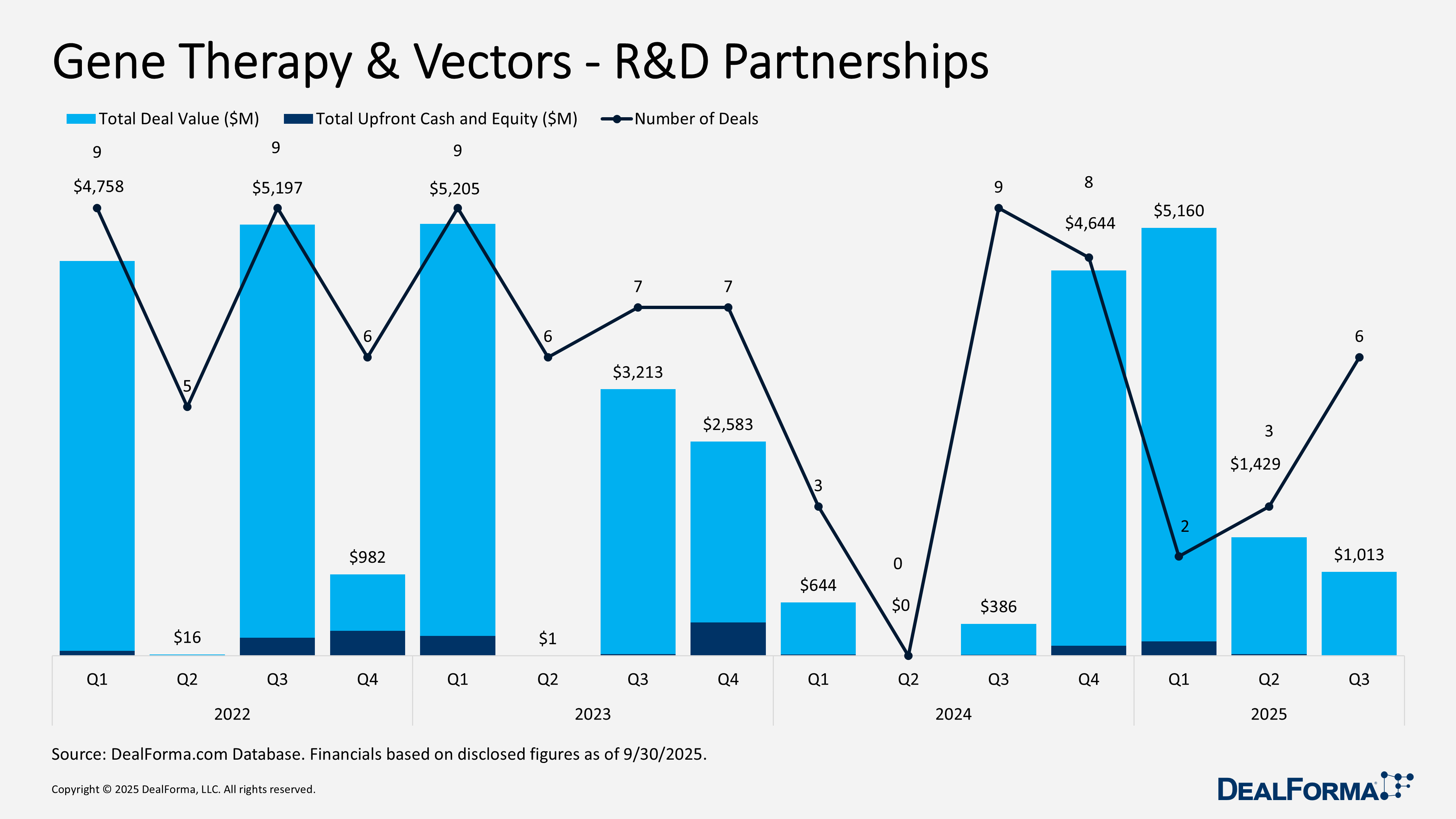
Q3 2025 reflected sustained strategic interest in gene therapy and vectors, with disciplined R&D partnerships and a minor rebound in venture funding, particularly for later-stage and platform-driven assets. The sector recorded 6 R&D partnerships totaling approximately $1 billion in announced value, but only $8 million in upfront payments, reinforcing the continued shift toward milestone-heavy deal structures. Notable transactions included the JCR Pharmaceuticals & Alexion platform collaboration, carrying up to $825 million in development and commercial milestones, and the Ocugen & Kwangdong Phase III regional license with up to $188 million in milestones and a 25% royalty, highlighting selective interest in de-risked assets.
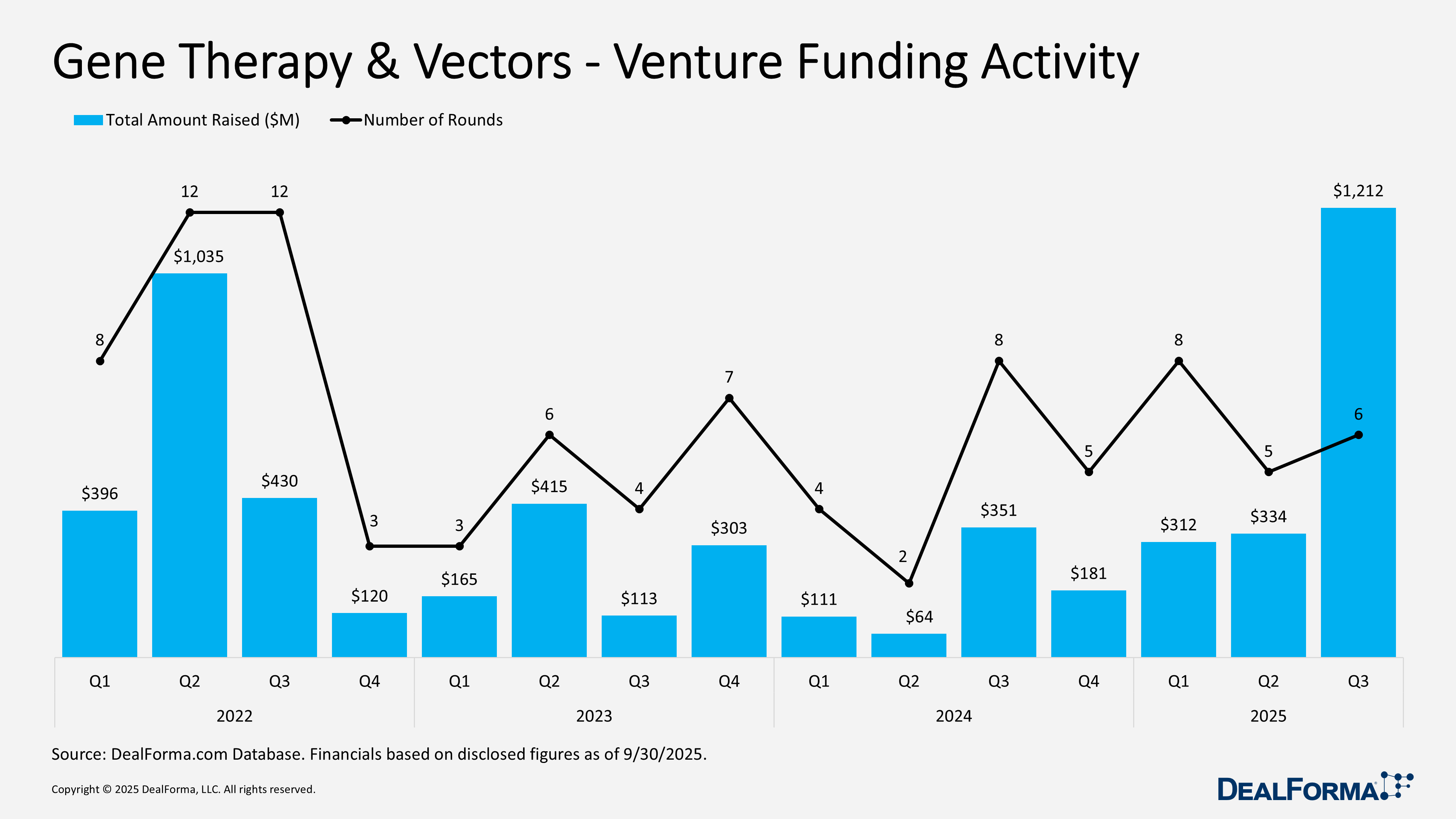
Venture funding accelerated sharply, with 6 rounds raising $1.2 billion, up from $334 million in Q2, driven by larger financings led by Kriya Therapeutics $320 million series D, alongside Epigenic Therapeutics $60 million series B and YolTech Therapeutics $45 million series B, signalling scalable gene therapy platforms and clinically advanced programs.
Gene Therapy & Vectors R&D Partnerships
In Q3 2025, gene therapy and vector R&D partnerships remained active, with 6 deals totaling $1 billion in announced value. However, upfront cash and equity payments were limited to $8 million, indicating a preference for milestone-driven structures. By contrast, Q2 2025 saw fewer transactions, 3 deals, but a higher aggregate value of $1.4 billion and $18 million in upfronts, reflecting larger individual partnerships with slightly stronger near-term cash components. Across Q2 and Q3 2025, the sector recorded 9 deals worth $2.4 billion in total value, with $26 million paid upfront.
Top R&D Partnership Gene Therapy & Vectors in Q3 2025
JCR Pharmaceuticals development and commercialization deal with Alexion – July 2025
JCR Pharmaceuticals granted Alexion exclusive rights to develop and commercialize up to 5 gene therapy programs using JCR’s JUST AAV capsid platform, targeting undisclosed indications. The agreement includes an undisclosed upfront payment, up to $225 million in research and development milestones, and up to $600 million in sales-based milestones, plus tiered royalties, contingent upon the pursuit of all 5 programs. In September 2025, JCR and Alexion achieved undisclosed research milestones, triggering additional milestone payments.
Ocugen development and commercialization deal with Kwangdong Pharmaceutical – September 2025
Ocugen granted Kwangdong exclusive rights to develop and commercialize OCU-400, a Phase III modifier gene therapy for retinitis pigmentosa, in Korea. Under the agreement, Ocugen will retain responsibility for manufacturing and commercial supply, while Kwangdong will lead local regulatory approval and commercialization, supported by Ocugen’s ongoing Phase III data and planned US BLA filing in 2026. The deal includes up to $8 million in upfront and near-term development milestones, with additional commercial milestones exceeding $180 million over 10 years, tied to sales performance, plus a 25% royalty, providing Ocugen with a long-term revenue stream with limited regional execution risk.
Gene Therapy & Vectors Venture Funding Activity
In Q3 2025, venture funding in gene therapy and vectors rebounded with 6 rounds raising $1.2 billion, driven by significantly larger financings. Sharp increase from Q2 2025, when 5 rounds raised $334 million, reflecting similar deal volume but smaller check sizes. Quarter over quarter, total funding rose by $878 million while the number of rounds increased marginally, pushing the average round size from approximately $67 million in Q2 to about $202 million in Q3. Across Q2 and Q3 2025 combined, the sector raised $1.5 billion over 11 rounds, with most capital concentrated in Q3.
Top Venture Funding Activity Gene Therapy & Vectors in Q3 2025
Kriya Therapeutics – Series D – $320M – September 2025
Kriya Therapeutics closed a $320 million series D financing to advance its pipeline of gene therapies targeting chronic diseases with high unmet need. The oversubscribed round was co-led by Patient Square Capital and Premji Invest, with participation from Peter Thiel, Narya Capital, The T1D Fund, and other long-term investors, and was completed at a significant valuation increase versus the prior round. Proceeds will fund clinical trials across Kriya’s 5 lead programs and support continued investment in its research and manufacturing platform. The pipeline targets large commercial indications, including geographic atrophy, thyroid eye disease, type 1 diabetes, metabolic-associated steatohepatitis, and trigeminal neuralgia. The financing strengthens Kriya’s balance sheet and positions the company to progress multiple clinical-stage assets while scaling its integrated manufacturing capabilities.
Epigenic Therapeutics – Series B – $60M – September 2025
Epigenic Therapeutics raised $60 million in a series B financing led by Lapam Capital, with participation from existing and new investors. Proceeds will support clinical development of EPI 003 for chronic hepatitis B and EPI 001 for hypercholesterolemia, as well as advancement of preclinical programs. The company’s EPIREG platform enables targeted gene silencing without DNA cutting, offering a differentiated safety profile, supported by its EpiTax in vivo delivery system. The financing strengthens Epigenic’s ability to progress its pipeline and expand its gene modulation platform.
YolTech Therapeutics – Series B – $45M – September 2025
YolTech Therapeutics closed an approximately $45 million series B financing led by the AstraZeneca CICC healthcare investment fund. Proceeds will support the advancement of the company’s clinical-stage in vivo gene editing programs and global development strategy. Founded in 2021, YolTech is developing one-time in vivo gene editing therapies supported by an integrated platform spanning proprietary CRISPR nucleases, base editors, and lipid nanoparticle delivery. Its lead program, YOLT 201 for transthyretin amyloidosis, entered Phase I, IIa trials in 2024, with additional clinical programs targeting familial hypercholesterolemia, primary hyperoxaluria type 1, and beta thalassemia sickle cell disease.
Also check out Gene Therapy & Vectors R&D Partnerships, M&A and Venture Funding – H1 2025 Review