Our review of the Q3 2025 neurology activity shows a mixed but broadly active market. R&D partnerships gained momentum, with 23 deals totaling $9.2 billion, driven by larger or more incentive-rich agreements. Upfront payments remained stable, while total deal value growth suggests sponsors are structuring partnerships with long-term milestones. Notable R&D deals include Arrowhead–Novartis ($200 million upfront, $2 billion potential milestones), Merck–Skyhawk (>$2 billion potential), and VectorY–Shape ($359 million upfront/option payments, up to $1.2 billion total).
M&A activity softened, with 18 deals worth $1.9 billion and lower cash outlays, indicating increased reliance on contingent payments and earnouts. In contrast, Q2 saw stronger upfront commitments. Major transactions included AbbVie’s $1.2 billion acquisition of Gilgamesh Pharmaceuticals, Remedy Meds’ $500+ million all-stock purchase of Thirty Madison, and the NeuroX–Relief Therapeutics merger ($1.2 billion NeuroX valuation). Venture Funding, moderated, with 35 rounds raising $1.3 billion versus $1.9 billion in Q2. Investment remained concentrated in select high-potential programs, including MapLight Therapeutics’ $373 million series D, Imagine Pediatrics’ $67 million series B, and NRG Therapeutics’ $67 million series B. Overall, the sector demonstrated strong R&D activity with significant long-term value creation, experienced cautious M&A characterized by lower upfront cash and saw selective venture funding.
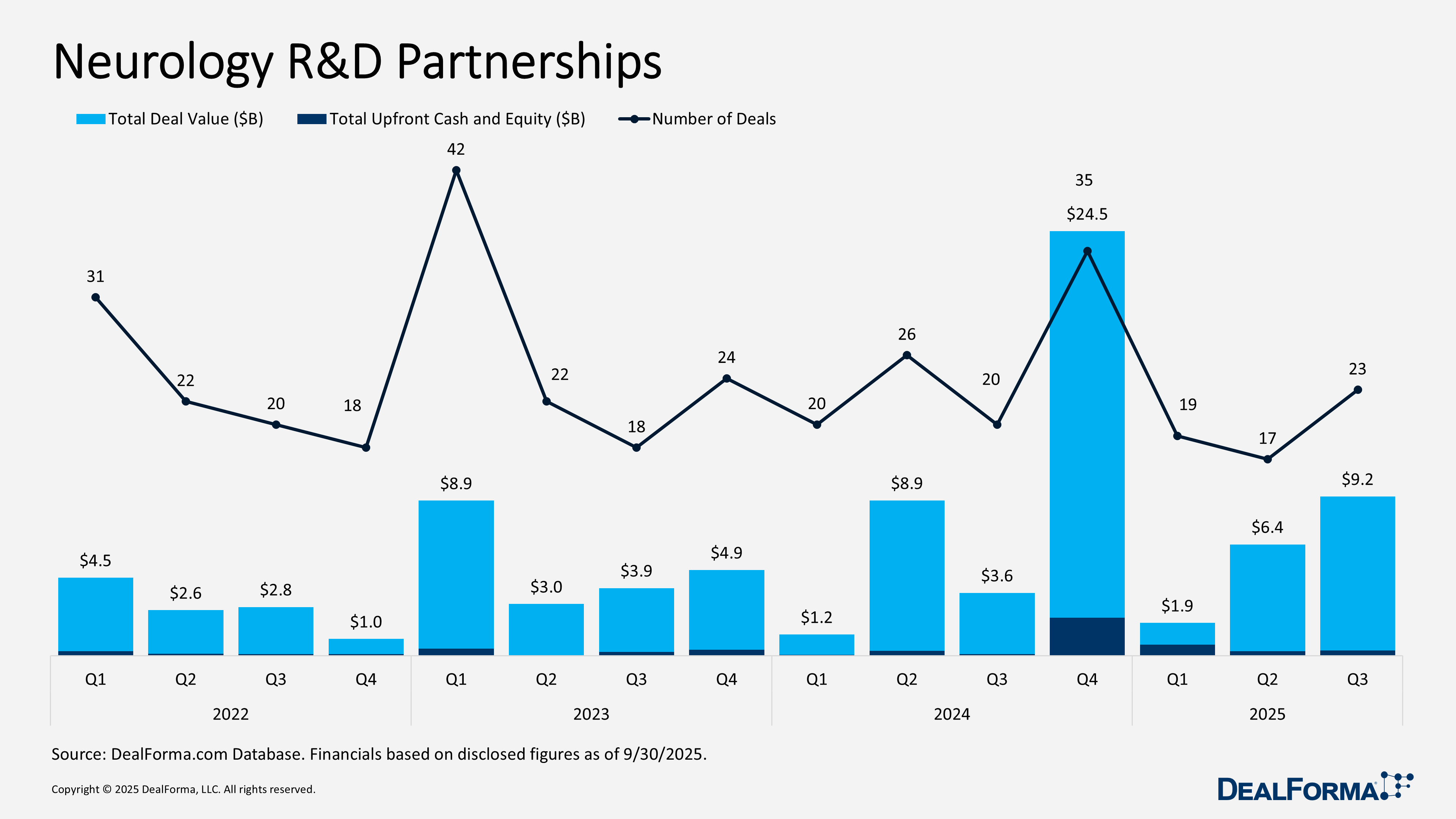
Neurology R&D Partnerships
Neurology R&D partnering accelerated in Q3 2025, with 23 deals totaling $9.2 billion and $294 million in upfront payments. A modest increase in volume and a significant rise in total deal value compared with Q2, pointing to larger or more incentive-rich structures even as upfront spending stayed stable. Q2 recorded 17 partnerships worth $6.4 billion with $254 million upfront, showing that while fewer deals closed, sponsors maintained consistent near-term capital deployment and concentrated variability in longer-term milestones. Across Q2 and Q3 combined, neurology partnerships reached 40 deals and $15.6 billion in value, with $548 million upfront.
Top Neurology R&D Partnership in Q3 2025
Arrowhead development and commercialization deal with Novartis – September 2025
Arrowhead granted Novartis exclusive global rights to ARO SNCA, a preclinical siRNA therapy targeting alpha synuclein for Parkinson’s disease and other synucleinopathies. The collaboration leverages Arrowhead’s TRiM RNAi platform, with Novartis also holding an option to add additional TRiM-based targets. Arrowhead will complete all CTA enabling preclinical work, after which Novartis will take over clinical development, manufacturing, medical affairs, and commercialization. Financial terms include a $200-million upfront payment and up to $2 billion in potential development, regulatory, and sales milestones, along with tiered royalties.
Merck KGaA research partnership with Skyhawk Therapeutics – August 2025
Merck entered a research partnership with Skyhawk Therapeutics to identify RNA-targeting small molecules for neurological disorders using Skyhawk’s SkySTAR platform. Skyhawk will lead discovery and preclinical activities, while Merck holds an exclusive global option to advance and commercialize any resulting therapies. If Merck exercises this option, it will take on all further development and commercialization. Skyhawk is eligible for more than $2 billion in upfront and milestone payments, along with tiered royalties.
VectorY Therapeutics research partnership with Shape Therapeutics – September 2025
VectorY Therapeutics partnered with Shape Therapeutics to develop vectorized antibody therapies for three neurodegenerative programs using Shape’s engineered AAV5-derived SHP-DB1 capsid, designed for deep-brain penetration and reduced toxicity. VectorY holds an option to develop and commercialize resulting therapies, supporting IV delivery for ALS, Huntington’s, and Alzheimer’s. Financial terms include an undisclosed upfront and option payments totaling $359 million, with Shape eligible for up to $1.2 billion in total deal value, including $338 million in milestones for rare disease programs, $504 million for non-rare programs, plus tiered royalties.
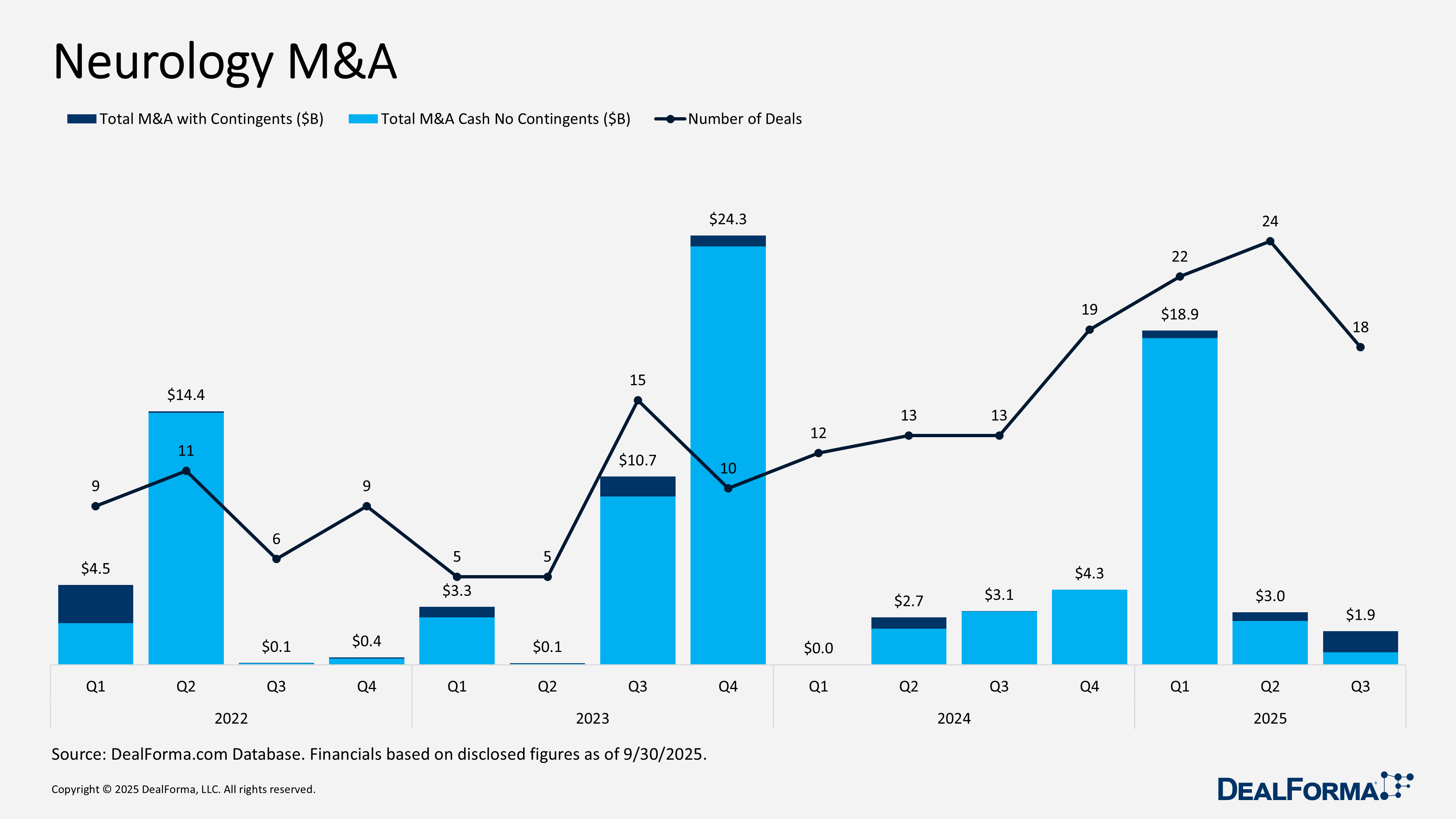
Neurology M&A
Neurology M&A activity softened in Q3 2025, with 18 deals valued at $1.9 billion, including contingents, and only $710 million paid in cash. This reflects both smaller transactions and a heavier use of earnouts, which accounted for about $1.2 billion of the total. By comparison, Q2 was materially stronger with 24 deals totaling $3 billion and $2.5 billion in cash consideration, indicating buyers were more willing to commit upfront and with greater certainty. Taken together, Q2 and Q3 delivered 42 deals worth $4.9 billion and $3.2 billion in cash.
Top Neurology M&A in Q3 2025
AbbVie acquiring Gilgamesh Pharmaceuticals – August 2025
AbbVie acquired Gilgamesh Pharmaceuticals to secure Bretisilocin, a Phase II 5-HT2A receptor agonist and 5-HT releaser for major depressive disorder. Gilgamesh spun off its other programs, including blixeprodil and the cardio-safe ibogaine analog M1/M4 agonist program, into a new entity, Gilgamesh Pharma, Inc., which will continue development of existing collaborations with AbbVie. Financial terms include up to $1.2 billion in total consideration, covering upfront and development milestones. The acquisition was completed on October 17, 2025.
Remedy Meds acquiring Thirty Madison in an all-stock transaction – September 2025
Remedy Meds is acquiring Thirty Madison in an all-stock transaction. Thirty Madison delivers telehealth-based, personalized care for chronic conditions, combining virtual consultations, customized treatments, and home delivery. The acquisition will expand Remedy Meds’ telehealth platform, integrating Thirty Madison’s brands to broaden access, accelerate treatment, and enhance operational scale across men’s, women’s, and metabolic health. The deal, expected to close in Q4 2025, values Thirty Madison at over $500 million upfront.
NeuroX reverse merging with Relief Therapeutics – July 2025
NeuroX is reverse merging with Relief Therapeutics to secure a public listing on the SIX Swiss Exchange. The combined company will focus on digital therapies for neurological disorders, integrating NeuroX’s AI platform and MindMaze product portfolio with Relief’s market services. Upon completion, NeuroX shareholders will hold 91% of the merged entity, and Relief shareholders 9%, based on valuations of CHF 1 billion ($1.2 billion) for NeuroX and CHF 100 million ($125 million) for Relief. The merger, expected to close in Q4 2025, was approved by Relief shareholders on November 17, 2025, creating MindMaze Therapeutics Holding SA. The transaction includes issuing 140 million new shares, restructuring capital, and appointing a leadership team from NeuroX and MindMaze. On December 15, 2025, the business combination between MindMaze and NeuroX was completed. The newly combined entity now operates under the name MindMaze Therapeutics Holding SA.
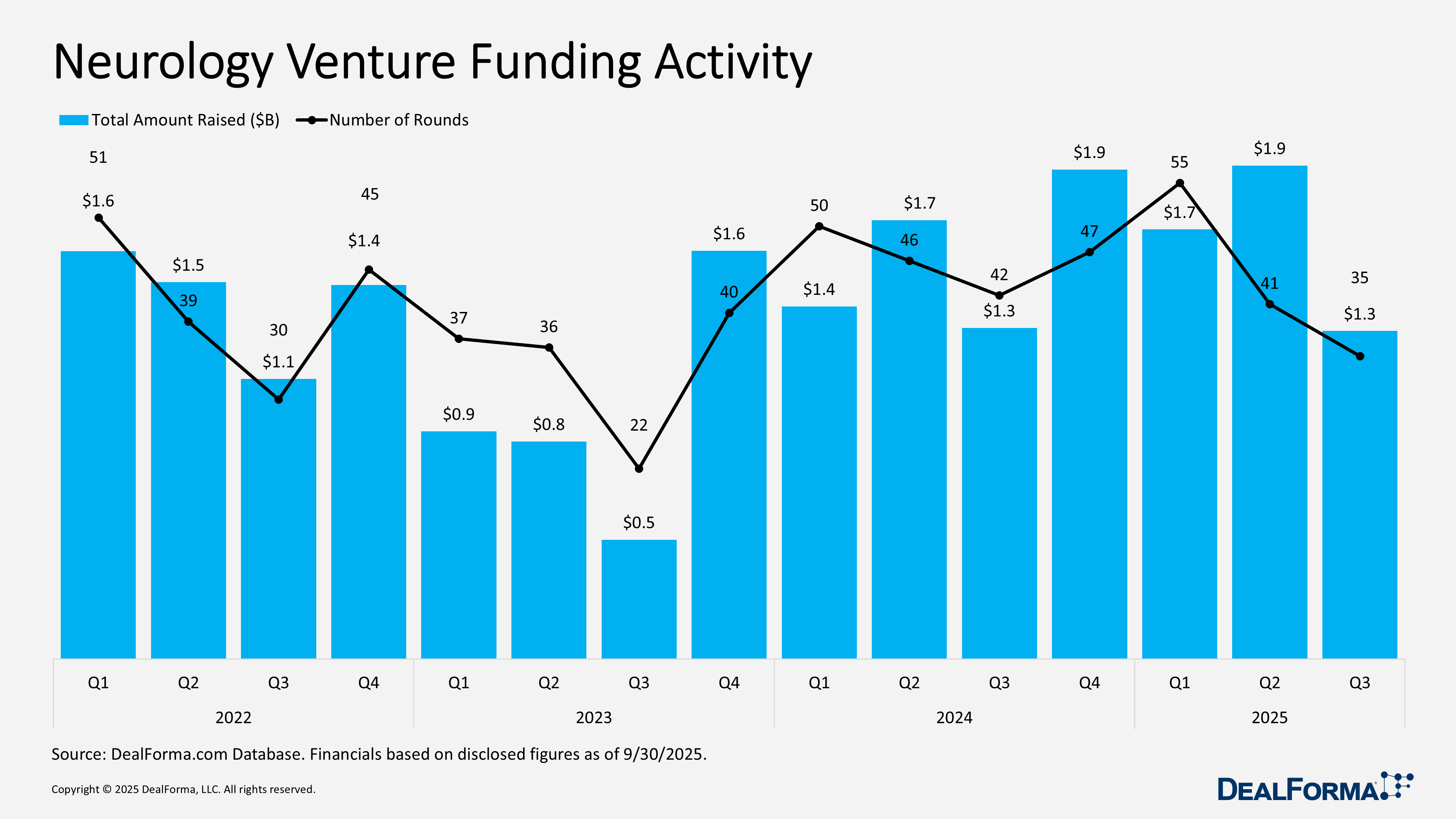
Neurology Venture Funding Activity
Neurology venture funding moderated in Q3 2025, with 35 rounds totaling $1.3 billion, reflecting both fewer financings and lower capital deployment than the prior quarter. Q2 was notably stronger, with 41 rounds raising $1.9 billion, signaling a more risk-on environment and the presence of several larger rounds that boosted aggregate funding. Across Q2 and Q3, the sector recorded 76 rounds and $3 billion.
Top Neurology Venture Funding in Q3 2025
MapLight Therapeutics – Series D – $373M – July 2025
MapLight Therapeutics announced an oversubscribed $373 million series D financing co-led by Forbion and Life Sciences at Goldman Sachs Alternatives, with participation from new investors including Sanofi, T. Rowe Price-advised accounts, and Avego BioScience Capital, as well as existing backers such as Novo Holdings, 5AM Ventures, and Blue Owl Healthcare Opportunities. The funding will advance MapLight’s lead program, ML-007C-MA, an oral fixed-dose combination of the M1/M4 muscarinic agonist ML-007 with a peripherally acting anticholinergic, designed to enhance CNS efficacy while reducing peripheral side effects. Proceeds will support ongoing Phase 2 trials in schizophrenia and Alzheimer’s disease psychosis, exploration of additional indications, and the continued development of other clinical and preclinical pipeline programs. MapLight is a clinical-stage biopharmaceutical company focused on CNS disorders.
Imagine Pediatrics – Series B – $67M – September 2025
Imagine Pediatrics, a tech-enabled pediatric medical group providing 24/7 virtual and in-home care for children with special health care needs, has raised $67 million in series B funding. Existing investors Oak HC/FT, Optum Ventures, and Rubicon Founders participated, alongside new strategic investment from the Autism Impact Fund and others. Founded in 2022, Imagine Pediatrics serves roughly 40,000 children nationwide through an integrated medical, behavioral, and social care model. The funding will support national expansion, enhance the company’s proprietary technology platform, and drive clinical innovation to close care gaps and improve access to specialized pediatric services under value-based care arrangements.
NRG Therapeutics – Series B – $67M – September 2025
NRG Therapeutics closed an oversubscribed £50 million ($67 million) series B to advance Parkinson’s studies and achieve proof of concept in ALS/MND. Led by SV Health Investors’ Dementia Discovery Fund, the round included British Business Bank, M Ventures, Novartis Venture Fund, Criteria Bio Ventures, and existing investors Omega Funds and Brandon Capital. The financing will support Phase 1b Parkinson’s trials and ALS/MND development of NRG5051, a first-in-class oral mPTP inhibitor targeting mitochondrial dysfunction and neuronal death, with potential to expand across other neurodegenerative indications.
Also check out Neurology R&D Partnerships, M&A, Ventures and IPOs – H1 2025 Review