Our review of biopharma therapeutics and platform deals by modality highlights how biologics and small molecules dominated dealmaking, while cell, gene, immunotherapy, and CRISPR therapies attracted large milestone-driven structures. Notable R&D deals included Arrowhead’s up to $10 billion RNA alliance with Sarepta (Nov 2024) in neurology and musculoskeletal disorders, BioNTech’s $11.1 billion bispecific antibody oncology partnership with BMS (Jun 2025), and GSK’s $7.2 billion discovery collaboration with Flagship Pioneering for respiratory and immunology (Jul 2024).
On the M&A front, Johnson & Johnson acquired Intra-Cellular Therapies for $14.6 billion (Jan 2025) to strengthen its CNS portfolio, Sanofi purchased Blueprint Medicines for $9.5 billion (Jun 2025) in systemic mastocytosis and oncology, and Mallinckrodt merged with Endo in a $6.7 billion transaction (Mar 2025), creating a diversified specialty pharma company. Venture financing remained robust, highlighted by Verdiva Bio’s $410 million series A (Jan 2025) and Kailera’s $400 million series A (Oct 2024) in obesity and cardiometabolic disorders, alongside ArsenalBio’s $325 million series C (Sep 2024) to advance CAR T-cell therapies for solid tumors.
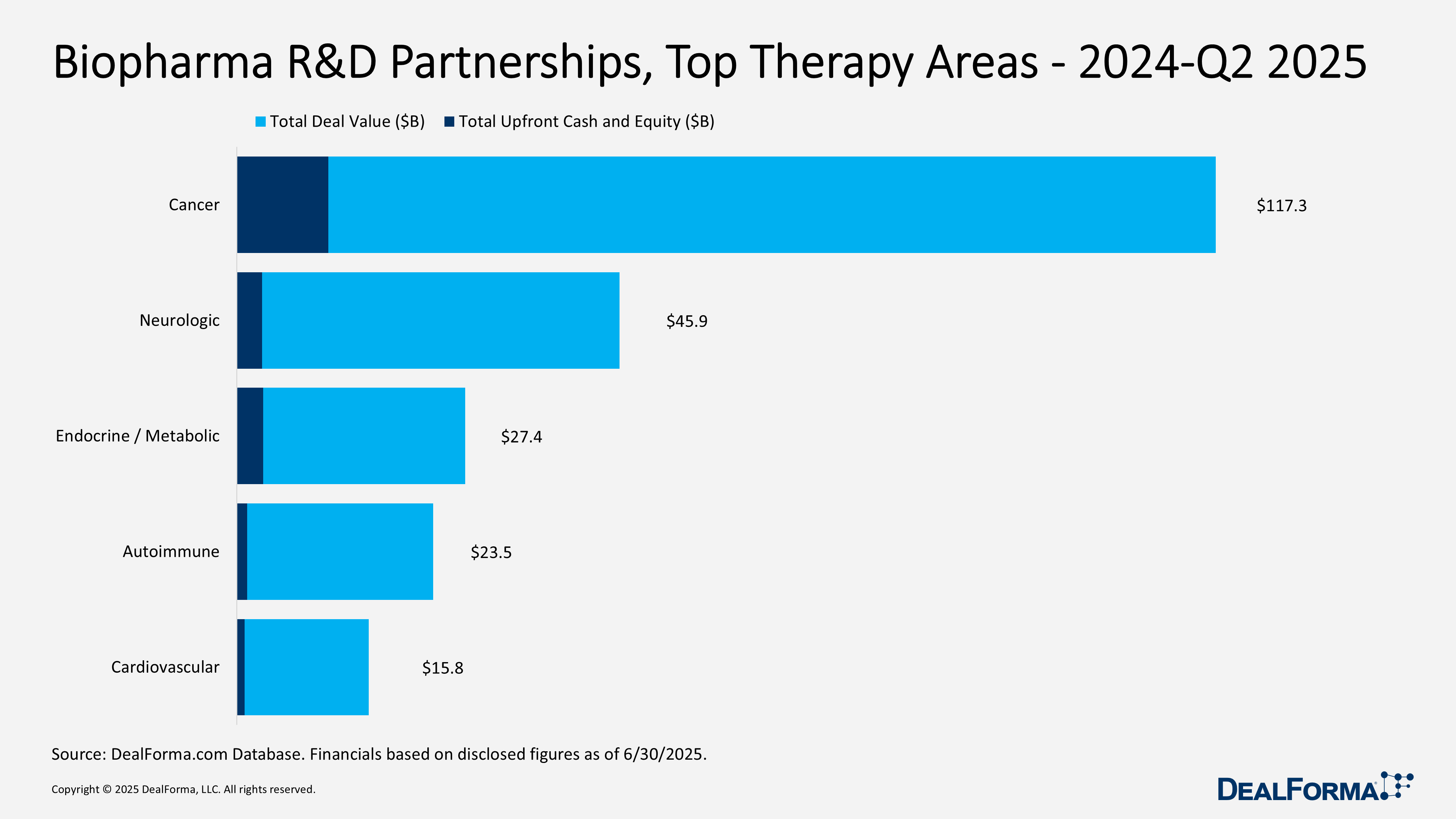
Biopharma R&D partnerships, Top Modalities – 2024 – Q2 2025
From 2024 through Q2 2025, R&D R&D partnerships were dominated by biologics, which accounted for 274 of 592 deals (46%), generating $160.6 billion of $277.4 billion total value and $13.4 billion of $20.6 billion upfront spend (average $126 million). Small molecules ranked second with 151 deals worth $55.6 billion and $4.8 billion upfront (average $80 million). Together, these two modalities represented 72% of transactions, 78% of deal value, and 88% of upfront commitments. Platform and data technologies (genomics, sequencing, screening) ranked third with 74 deals worth $29.4 billion but saw milestone-heavy structures, delivering just $900 million upfront (average $50 million). Gene therapy, though limited to 20 deals, delivered high value density at $14.4 billion total ($720 million per deal on average) but with minimal upfronts ($39 million), highlighting option-driven, back-loaded economics. Cell therapy recorded 39 deals ($10 billion total, $600 million upfront, average $65 million). Immunotherapy (23 deals; $3.8 billion; $66 million average upfront) and CRISPR (11 deals; $3.7 billion; $58 million average upfront) remained niche but showed solid per-deal terms.
Top Deals R&D partnerships 2024 – Q2 2025
Arrowhead development and commercialization deal with Sarepta Therapeutics – November 2024
Arrowhead granted Sarepta exclusive global rights to seven RNA programs across neurology, pulmonary, and musculoskeletal diseases, with options for six more under a five-year collaboration. Deal terms included $500 million upfront, $325 million in equity (35% VWAP premium), $250 million annually, and up to $300 million near-term milestones, plus development and sales milestones bringing potential value to more than $10 billion with royalties. In 2025, Sarepta’s restructuring added provisions that could return rights to Arrowhead if payments lapse. Arrowhead has since received $100 million in DM1 trial milestones (with another $100 million pending), while Sarepta raised $224 million through sales and buybacks of 12 million Arrowhead shares.
BioNTech development and commercialization deal with BMS – June 2025
BioNTech licensed BMS exclusive global rights to BNT-327, a bispecific antibody targeting PD-L1/VEGF-A, in development for >10 solid tumor indications. Originating from BioNTech’s Biotheus acquisition, the asset is in Phase III for small and non-small cell lung cancers, with a Phase III in TNBC expected in late 2025. BioNTech will supply the study drug and co-develop with BMS, retaining rights in the US, EU, UK, China, and Turkey, while BMS books sales elsewhere; costs and profits are shared equally. Terms include $1.5 billion upfront, $2 billion in non-contingent anniversary payments through 2028, and up to $7.6 billion in milestones, for a total potential $11.1 billion deal.
GSK research partnership with Flagship Pioneering – July 2024
GSK entered a discovery-stage research partnership with Flagship Pioneering to develop up to 10 medicines and vaccines, initially focused on respiratory and immunology. Research will leverage Flagship’s bioplatform companies and diverse modalities, including vaccines, small molecules, RNA, and biologics. The collaboration is jointly funded with $150 million upfront. GSK holds exclusive options to license all resulting programs. Flagship and its bioplatforms are eligible for up to $720 million per program in upfront, development, and sales milestones plus royalties, representing a potential $7.2 billion total deal value across 10 programs.
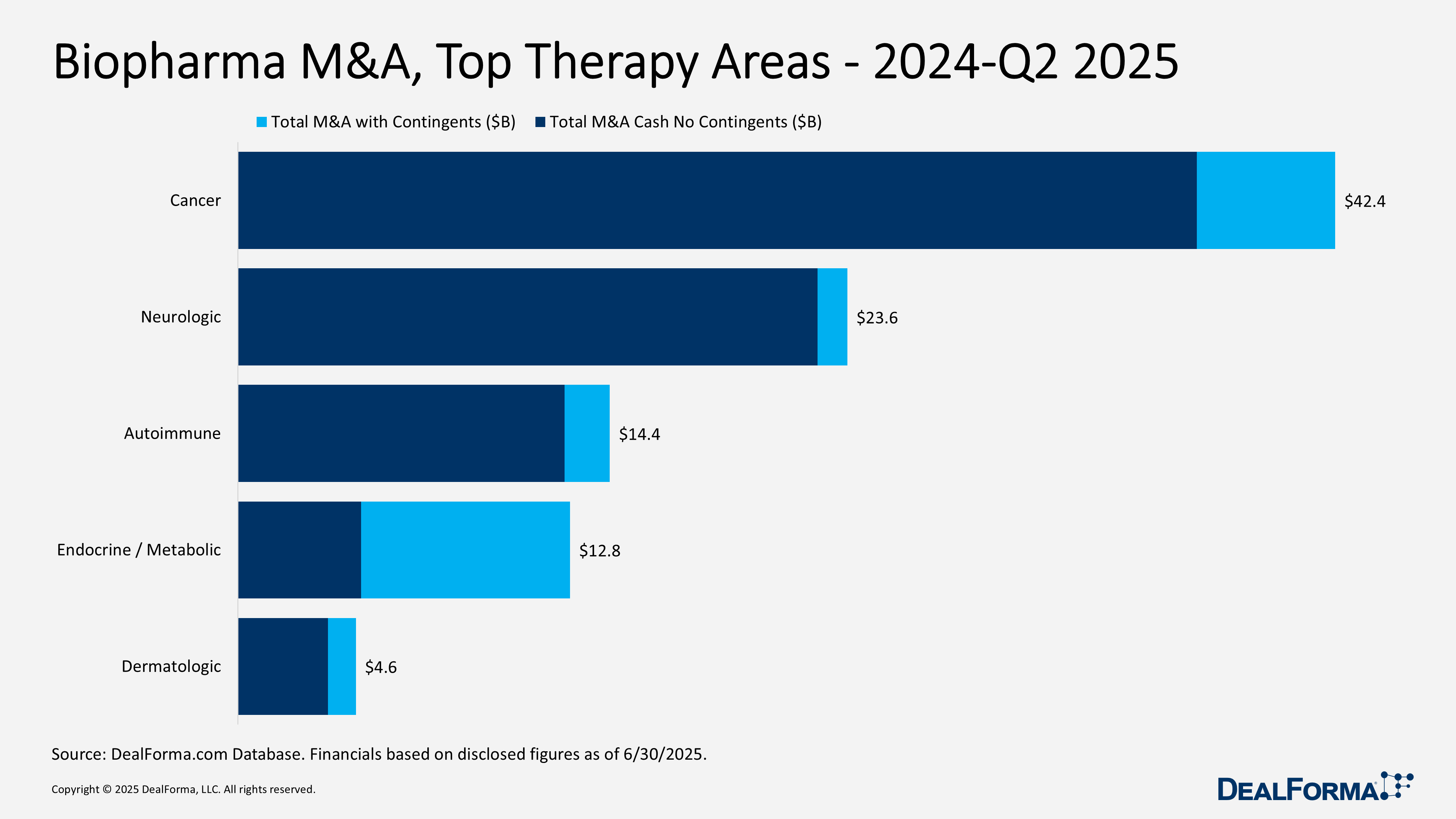
Biopharma M&A, Top Modalities – 2024 – Q2 2025
From 2024 through mid-2025, biopharma M&A remained concentrated in established modalities. Small molecules dominated with 62 of 136 deals (46%), totaling $65.9 billion ($55.8 billion cash) and averaging $1.4 billion upfront. Biologics followed with 54 deals worth $31.5 billion ($21.9 billion cash) and $645 million average upfront. Specialized areas were fewer but carried premium terms. Immunotherapy saw only 4 acquisitions, yet the highest averages at $1.7 billion upfront ($7.4 billion total; $6.7 billion cash). Gene therapy and vectors (5 deals) reached $3.3 billion ($2.2 billion cash; $718 million average upfront), CRISPR (3 deals) $1.5 billion ($1.1 billion cash; $533 million average), and cell therapy (8 deals) $3.1 billion ($2 billion cash; $329 million average). In total, 136 transactions generated $112.7 billion ($89.6 billion cash).
Top Deals M&A 2024 – Q2 2025
Johnson & Johnson acquiring Intra-Cellular Therapies – January 2025
J&J acquired Intra-Cellular Therapies for $132 per share in cash, a 40% premium, valuing the deal at $14.6 billion. Intra-Cellular’s lead asset, Caplyta, is approved for schizophrenia, bipolar disorder, depression, autism, and sleep disorders, with additional indications in development. The pipeline includes Phase II lenrispodun (Parkinson’s disease, heart failure), lumateperone deuterated (anxiety, psychosis), Phase I ITI-1020 (cancer), ITI-333 (pain), and multiple preclinical PDE1 inhibitors for CNS disorders. Following completion on April 2, 2025, Intra-Cellular now operates as a J&J business unit.
Sanofi acquiring Blueprint Medicines – June 2025
Sanofi acquired Blueprint Medicines for $129 per share in cash, a 27% premium, valuing the deal at $9.5 billion ($9.1 billion upfront plus up to $400 million in milestones tied to BLU-808). Blueprint shares ceased trading on NASDAQ at closing. Blueprint’s portfolio includes approved Ayvakit (GIST and systemic mastocytosis), late-stage elenestinib (Phase III, systemic mastocytosis), and mid-stage BLU-808 (Phase II, chronic urticaria and allergic rhinoconjunctivitis, with POC studies planned in asthma and mast cell activation). The pipeline also features early programs in solid tumors, breast cancer, and targeted protein degraders (CDK2, CDK4).
Mallinckrodt acquiring Endo – March 2025
Mallinckrodt acquired Endo in a cash-and-stock transaction valued at an enterprise value of $6.7 billion. Endo shareholders received $80 million in cash and will own 49.9% of the combined company, with Mallinckrodt shareholders holding 50.1%. The merged entity is listed on the NYSE, with plans to spin off Endo’s generics and sterile injectables businesses as a separate company. Endo brought a portfolio of 27 approved branded drugs, including Aveed (hypogonadism), Frova (migraine), Nascobal (anemia), Edex (erectile dysfunction), Percocet (pain), and Supprelin-LA (precocious puberty, prostate cancer), alongside more than 100 generics and 40 sterile injectables in development.
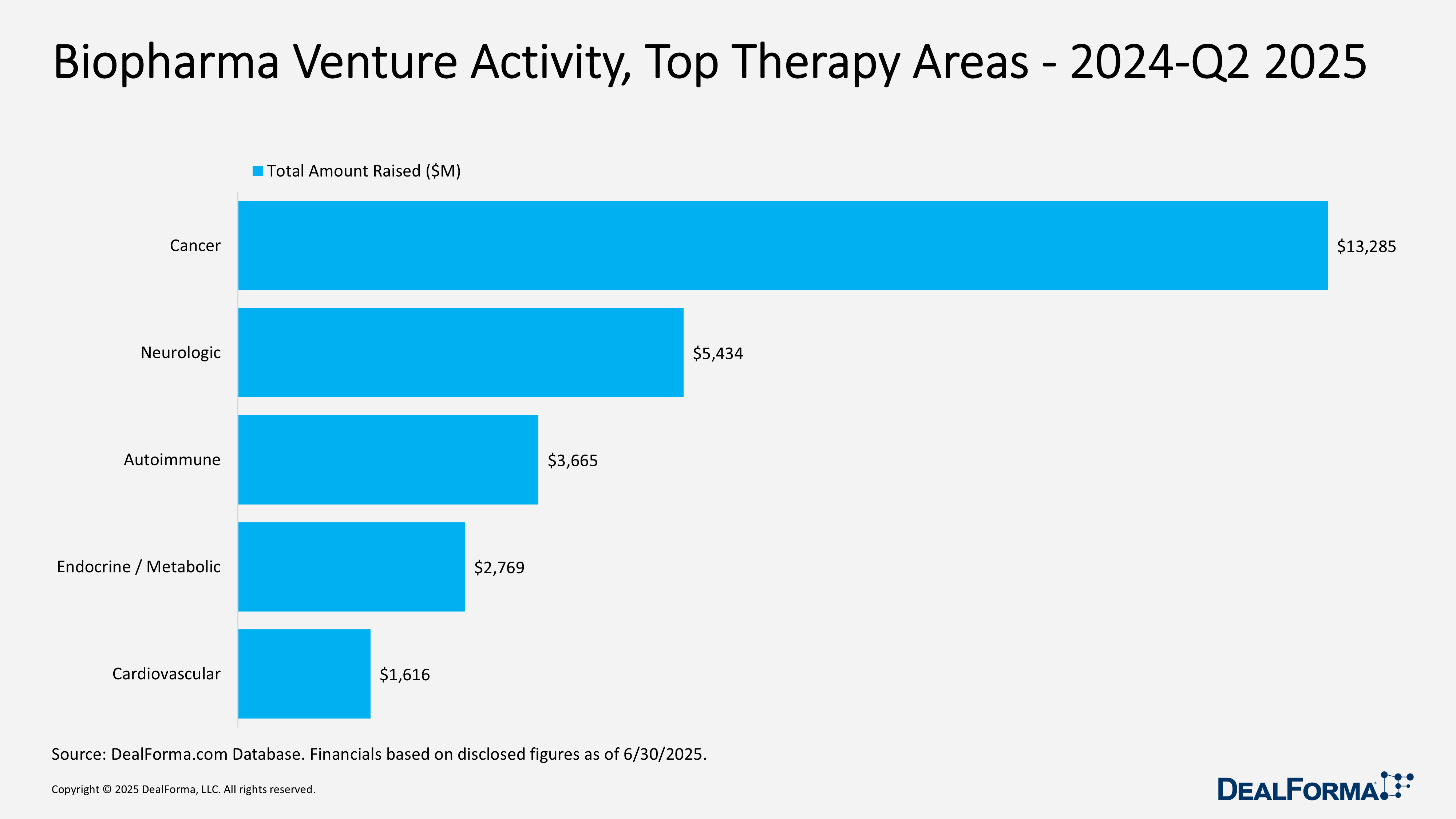
Biopharma Venture Activity, Top Modalities – 2024 – Q2 2025
From 2024 through Q2 2025, venture capital flowed primarily to biologics and small molecules, which together accounted for 408 of 537 rounds (76%) and $23.6 billion of $30.1 billion raised (79%). Biologics led slightly with $12.9 billion across 208 rounds (average $69 million), while small molecules attracted $10.7 billion across 200 rounds (average $58 million). Secondary modalities remained active but smaller in scale. Cell therapy raised $2.3 billion across 52 rounds (average $47 million), and immunotherapy secured $2.1 billion from 34 rounds with higher check sizes (average $64 million). Gene therapy and vectors drew $1.5 billion over 33 rounds (average $48 million), while CRISPR, though limited to 10 rounds, delivered the highest average ticket at $86 million. The blended average round size across all modalities was $56 million.
Top Deals Venture Activity 2024 – Q2 2025
Verdiva Bio – Series A – $410M – January 2025
Verdiva Bio, a UK-based clinical-stage biopharma, launched with a $410 million oversubscribed series A co-led by Forbion and General Atlantic, joined by RA Capital, OrbiMed, Logos Capital, Lilly Asia Ventures, and LYFE Capital. The company secured exclusive global rights (ex-China, South Korea) to Sciwind Biosciences’ portfolio, including ecnoglutide, a Phase 2-ready oral GLP-1 agonist; a once-weekly oral amylin agonist; and a long-acting subcutaneous amylin agonist, each for monotherapy or in combination with GLP-1. Backed by one of the largest obesity-focused series A financings, Verdiva is positioned as a next-generation competitor in obesity and cardiometabolic therapies.
Kailera Therapeutics – Series A – $400M – October 2024
Kailera Therapeutics launched with a $400 million series A co-led by Atlas Venture, Bain Capital Life Sciences, and RTW Investments, with Lyra Capital participating. The company licensed four metabolic disease assets from Jiangsu Hengrui (ex-China), led by KAI-9531, an injectable GLP-1/GIP dual agonist with positive Phase 2 obesity and type 2 diabetes data. Additional programs include KAI-7535, an oral GLP-1 agonist; a once-daily oral tablet version of KAI-9531; and KAI-4729, an injectable GLP-1/GIP/glucagon tri-agonist, advancing its strategy in obesity and metabolic disorders.
ArsenalBio – Series C – $325M – September 2024
Arsenal Biosciences (ArsenalBio), a clinical-stage company developing programmable CAR T-cell therapies for solid tumors, closed an oversubscribed $325 million series C. New investors included ARCH Venture Partners, Milky Way Investments Group, Regeneron Ventures, NVentures, Luma Group, T. Rowe Price, and Rock Springs Capital, joining existing backers such as PICI, SoftBank Vision Fund 2, Bristol-Myers Squibb, Westlake Village BioPartners, Kleiner Perkins, Byers Capital, and Hitachi Ventures. Proceeds will advance lead CAR T programs, expand the solid tumor pipeline, and support new discovery tools and manufacturing innovations.
Also check out Biopharma Therapeutics and Platforms 2024-Q2 2025