Neurology in H1 2025 reflected a selective yet strategic landscape, with slower partnering, fewer but larger acquisitions, and steady venture funding. Key R&D partnerships included GSK’s April 2025 deal with ABL Bio to develop antibody- and RNA-based therapies using the Grabody-B BBB platform, providing $50 million upfront and up to $2.7 billion in milestones and royalties; Eli Lilly’s April 2025 collaboration with Sangamo on its STAC-BBB AAV capsid platform with $18 million upfront and up to $1.4 billion in fees and milestones; Biogen’s May 2025 agreement with City Therapeutics on RNAi-based CNS therapies with $46 million upfront ($16 million cash, $30 million convertible notes), an undisclosed option fee, and up to $1 billion in milestones plus royalties; Angelini Pharma’s May 2025 license of GRIN’s radiprodil for $50 million upfront, up to $520 million in milestones, plus royalties; and Ferrer’s co-development of Prilenia’s Phase III oral therapy Pridopidine for Huntington’s disease in April 2025 with €80 million ($91 million) upfront, up to €45 million ($51 million) near-term milestones, and up to €375 million ($426 million) in long-term regulatory, development, and commercial milestones, plus tiered royalties.
M&A reinforced neurology’s strategic reshaping: Eli Lilly’s May 2025 acquisition of SiteOne Therapeutics may yield up to $1 billion in cash including upfront and milestones; Supernus’ June 2025 acquisition of Sage Therapeutics valued at $561 million cash ($8.50/share) plus up to $234 million in CVRs; Sanofi’s May 2025 acquisition of Vigil Neuroscience, closed August 6, with $470 million cash ($8/share) plus up to $130 million in CVRs; Atai Life Sciences’ June 2025 all-stock acquisition of Beckley Psytech issued 105 million shares ($390 million) to Beckley shareholders (31% of combined company) and included a $30 million private placement, and Alto Neuroscience’s June 2025 acquisition of Chase Therapeutics included $2 million upfront, up to $72 million in development milestones, and $41 million in sales milestones.
Venture financing remained strong, supporting early- and mid-stage innovation: Draig Therapeutics raised $140 million (£107 million) in June 2025 series A; GRIN Therapeutics closed $140 million in a May 2025 series D ($65 million Angelini, $75 million Blackstone) with a $50 million upfront collaboration and up to $520 million in milestones; Neurona Therapeutics raised $102 million in April 2025; Neuron23 secured $97 million in June 2025 series D for its Phase 2 NEULARK trial; and Vima Therapeutics launched with $60 million series A in May 2025 to advance its oral therapy VIM0423 for dystonia and related movement disorders.
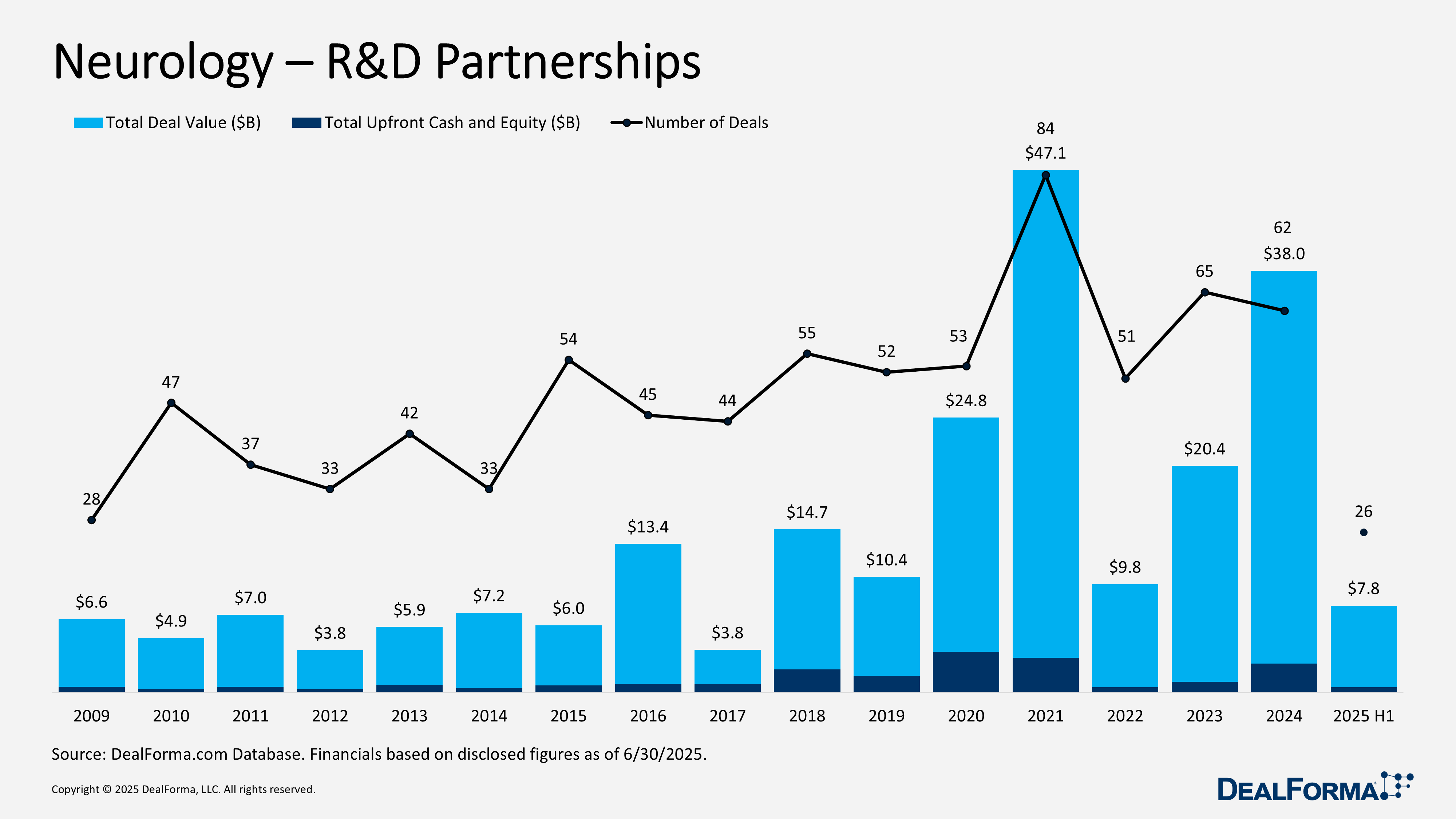
Neurology R&D Partnerships
During H1 2025, neurology R&D partnerships slowed, with 26 agreements totaling $7.8 billion and $500 million in upfront cash and equity. The average upfront was $50 million, with an average deal size of $300 million. By comparison, 2024 recorded 62 deals worth $38 billion, including $2.6 billion in upfront payments, averaging $97 million upfront and $613 million per deal. Across the combined period of 2024 through H1 2025, a total of 88 deals were signed, amounting to $45.9 billion with $3.1 billion in upfront payments. H1 2025 contributed 30% of total deal volume but only 17% of overall value and upfronts.
Top Neurology R&D Partnerships in H1 2025
ABL Bio development and commercialization deal with GSK – April 2025
GSK secured exclusive global rights from ABL Bio to develop and commercialize antibody- and RNA-based therapies, including siRNA and antisense oligonucleotides, for neurologic diseases. The collaboration leverages ABL’s Grabody-B blood-brain barrier shuttle platform. ABL Bio received £39 million ($50 million) upfront and is eligible for up to £2.1 billion ($2.7 billion) in potential payments across milestones, alongside tiered royalties.
Sangamo development and commercialization deal with Eli Lilly – April 2025
Eli Lilly obtained exclusive global rights to develop and commercialize gene therapies using Sangamo’s STAC-BBB AAV capsid platform, initially for one neurologic target with options to expand to four additional CNS indications. Sangamo will handle technology transfer, while Lilly assumes development, regulatory, and commercialization responsibilities. Sangamo received $18 million upfront and could earn up to $1.4 billion in target fees and milestones across five potential programs, plus tiered royalties.
City Therapeutics development and commercialization deal with Biogen – May 2025
Biogen secured exclusive global rights to develop and commercialize RNAi-based therapies for central nervous system diseases using City Therapeutics’ RNAi platform combined with Biogen’s delivery technology. Biogen will oversee development, regulatory, and commercialization, with an option to add one additional target. City Therapeutics received $46 million upfront ($16 million in cash and $30 million in convertible notes), an undisclosed option fee, and is eligible for up to $1 billion in development and commercial milestones, plus tiered royalties.
GRIN development and commercialization deal with Angelini – May 2025
Angelini Pharma obtained exclusive rights outside North America to commercialize radiprodil, a selective GluN2B NMDA receptor modulator in Phase II development for GRIN-related neurodevelopmental disorder (GRIN-NDD), including tuberous sclerosis complex (TSC) and focal cortical dysplasia (FCD) type II. GRIN will retain rights in the United States, Canada, and Mexico, and will lead global development, with both parties sharing certain clinical costs. GRIN received $50 million upfront and is eligible for up to $520 million in milestones, plus tiered royalties.
Prilenia development and commercialization deal with Ferrer – April 2025
In April 2025, Prilenia granted Ferrer rights to co-develop, manufacture, and commercialize Pridopidine, an oral sigma-1 receptor agonist in Phase III for Huntington’s disease, across Europe, MENA, Southern Africa, Latin America, and CIS. Prilenia retains rights in North America, Japan, and the Asia-Pacific. The drug is under EMA review with CHMP opinion expected in H2 2025. Ferrer and Prilenia will also co-develop the therapy for additional indications within the licensed regions. Financial terms include €80 million ($91 million) upfront, up to €45million ($51 million) in near-term milestones, and up to €375 million ($426 million) in regulatory, development, and commercial milestones.
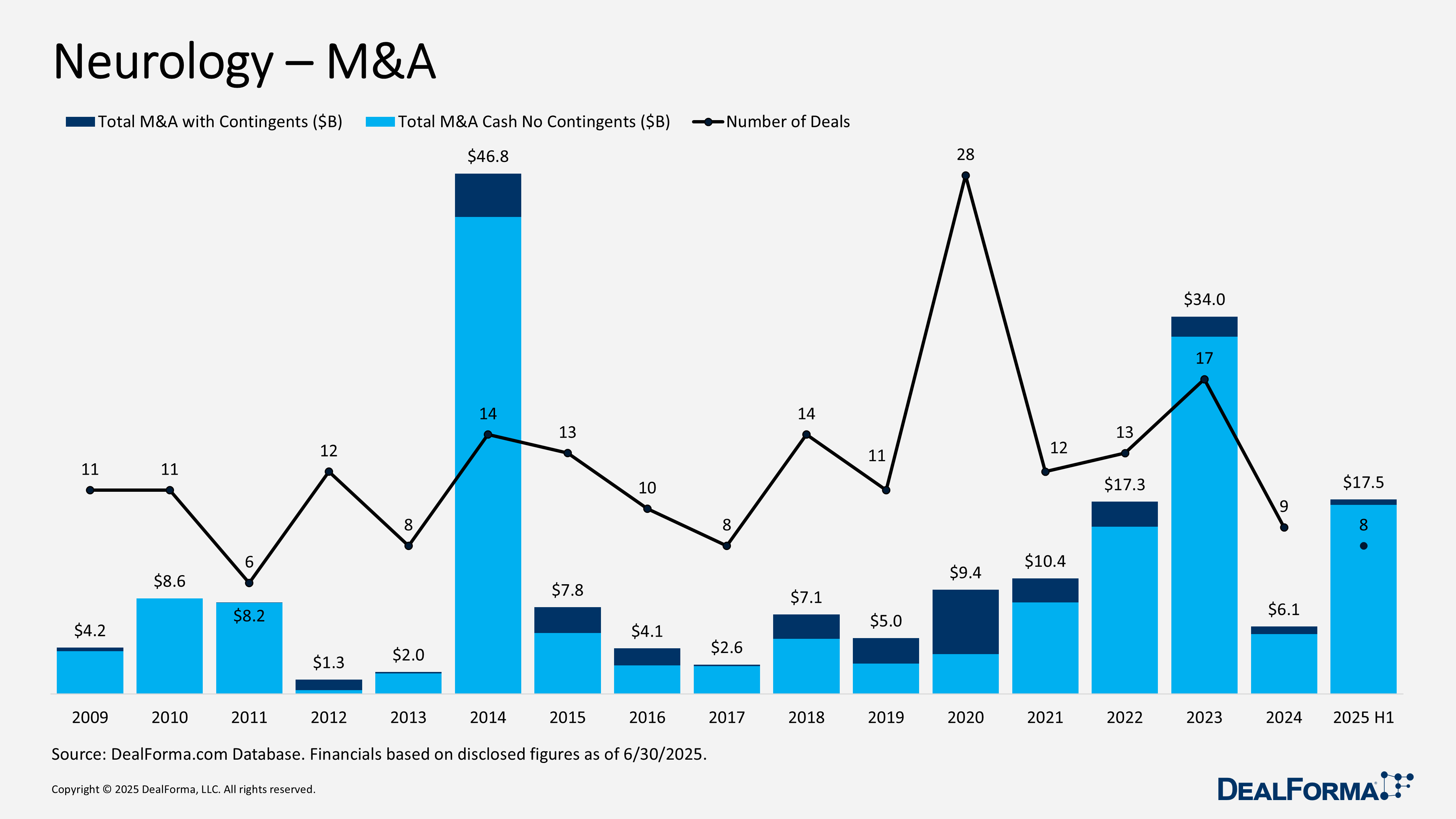
Neurology M&A
During H1 2025, neurology M&A were fewer in number but significantly larger in scale. Eight acquisitions generated $17.5 billion in headline value ($2.2 billion per deal) and $17 billion in cash excluding contingents ($2.1 billion per deal). The average upfront reached $2.9 billion, reflecting a tilt toward mega-deals, while earnouts were minimal at $500 million, only 3% of total value. By comparison, 2024 recorded 9 deals worth $6.1 billion ($680 million per deal) and $5.4 billion in cash, with a much lower average upfront of $674 million. Across 2024 through H1 2025, there were 17 transactions totaling $23.6 billion, including $22.4 billion in cash.
Top Neurology M&A in H1 2025
Eli Lilly acquiring SiteOne Therapeutics – May 2025
Eli Lilly agreed to acquire SiteOne Therapeutics, whose lead program, STC-004, a Nav1.8 inhibitor for pain, is in Phase I with readiness for Phase II. The acquisition strengthens Lilly’s pain pipeline. SiteOne shareholders may receive up to $1 billion in cash through an upfront payment and milestone-based considerations (details undisclosed).
Supernus acquiring Sage Therapeutics – June 2025
Supernus agreed to acquire Sage Therapeutics, whose portfolio includes FDA-approved zurzuvae for postnatal depression, Phase II–ready SAGE-324 for epilepsy, Phase I SAGE-319 for neurodevelopmental disorders, and two preclinical assets (SAGE-817 and SAGE-039). Sage shareholders will receive $8.50 per share in cash (27% one-day premium), valuing the deal at $561 million, plus up to $3.50 per share in contingent value rights (CVRs) worth an additional $234 million, tied to zurzuvae’s performance. CVR milestones include: $1 if U.S. net sales reach $250 million by 2027; $1 if sales reach $300 million by 2028; $1 if sales reach $375 million by 2030; and $0.50 upon first commercial sale in Japan for major depressive disorder by June 30, 2026.
Sanofi acquiring Vigil Neuroscience – May 2025
Sanofi announced the acquisition of Vigil Neuroscience, adding two key TREM2 agonist programs: Iluzanebart (VGL-101), a Phase II monoclonal antibody for ALSP, a rare neurodegenerative disease, and VG-3927, a Phase I oral small molecule for Alzheimer’s disease. Vigil will terminate its Amgen collaboration on VGL-101 before closing. Sanofi had previously partnered with Vigil on TREM2 in 2024 and invested during the company’s series A. Vigil shareholders will receive $8 per share in cash (246% one-day premium), valuing the deal at approximately $470 million, plus up to $2 per share in CVRs worth an additional $130 million, tied to the first commercial sale of VG-3927.
atai Life Sciences acquiring Beckley Psytech in an all-stock transaction – June 2025
Atai Life Sciences announced an all-stock acquisition of Beckley Psytech, forming a combined company to be named Atai Beckley. Beckley’s pipeline includes Phase IIb intranasal BPL-003 for treatment-resistant depression and alcohol use disorder, and ELE-101 for major depressive disorder, alongside several preclinical novel chemical entities. Before closing, Beckley will spin out Eleusis Holdings and its subsidiaries. Under the terms, Beckley shareholders (excluding Atai, which already holds shares) will own 31% of the combined company on a fully diluted basis. They will receive 105 million newly issued Atai shares valued at approximately $390 million (€0.10 per share), subject to a lock-up releasing 1/12 of the shares monthly. Concurrently, Atai raised $30 million in a private placement with Ferring Ventures and Adage Capital Partners to support development.
Alto Neuroscience acquiring Chase Therapeutics – June 2025
Alto Neuroscience announced the acquisition of Chase Therapeutics, adding two Phase II assets to its neurology pipeline. Lead candidate CTC-501 (to be renamed ALTO-207) is a fixed-dose combination of pramipexole and ondansetron for treatment-resistant depression, designed to mitigate pramipexole’s dose-limiting side effects. CTC-413 (to be renamed ALTO-208) is a neurokinin-1 (NK-1) receptor antagonist in development for Parkinson’s disease. Alto plans to initiate a pivotal Phase IIb trial of ALTO-207 in mid-2026, with topline data expected in 2027. Deal terms include $2 million upfront, up to $72 million in development milestones, and up to $41 million in sales milestones.
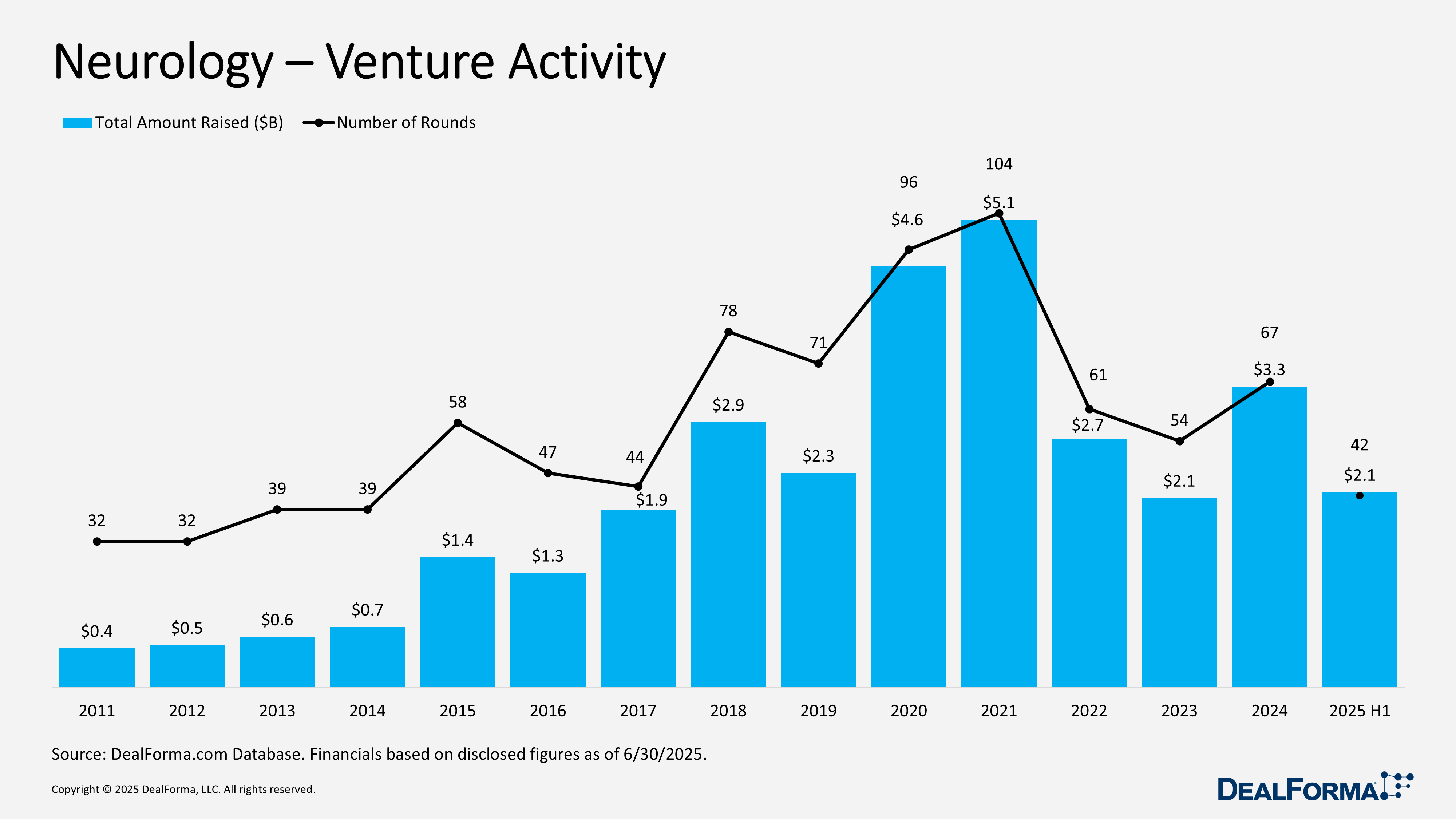
Neurology Venture Activity
During H1 2025, neurology venture funding recorded 42 rounds totaling $2.1 billion, with the average round size steady at $55 million. Activity slowed in pace but maintained consistent ticket sizes. In 2024, investors completed 67 rounds raising $3.3 billion, also averaging $55 million per round. Across 2024 through H1 2025, a total of 109 rounds raised $5.4 billion.
Top Neurology Venture Activity in H1 2025
Draig Therapeutics – Series A – $140M – June 2025
Draig Therapeutics, a clinical-stage biotech developing therapies for neuropsychiatric disorders, emerged from stealth with $140 million (£107 million) raised over the past nine months. The oversubscribed series A was led by Access Biotechnology, with participation from Canaan Partners, SR One, Sanofi Ventures, Schroders Capital, and seed investors SV Health Investors (company co-founder) and ICG. Founded in 2024 through a partnership between Cardiff University’s Medicines Discovery Institute and SV Health Investors, Draig is advancing a pipeline targeting core neurotransmission pathways. Proceeds will fund four clinical trials, including two Phase II studies of lead candidate DT-101 in major depressive disorder.
GRIN Therapeutics – Series D – $140M – May 2025
GRIN Therapeutics closed a $140 million series D financing, including a $65 million strategic equity investment from Angelini Pharma and $75 million from existing investor Blackstone Life Sciences. In parallel, GRIN entered a collaboration granting Angelini commercial rights to its lead asset, radiprodil, outside North America. The collaboration provides GRIN with a $50 million upfront payment and up to $520 million in potential milestones, plus tiered royalties and shared development costs. GRIN will retain exclusive rights in the U.S., Canada, and Mexico while continuing to lead global development. Funding from both transactions will support operations and advance radiprodil into a planned pivotal Phase 3 trial for GRIN-related neurodevelopmental disorder in 3Q 2025.
Neurona – Series Unspecified – $102M – April 2025
Neurona Therapeutics closed an upsized, oversubscribed $102 million financing with participation from Fidelity, The Column Group, Soleus Capital, Viking, Cormorant, Schroders Capital, LYFE, Euclidean, UCB Ventures, Willett, UC Investments, YK Bioventures, Berkeley Frontier Fund, Ysios, Alexandria, and Spur Capital. Proceeds will advance Neurona’s allogeneic regenerative cell therapy pipeline for chronic neurological disorders, led by NRTX-1001 for drug-resistant mesial temporal lobe epilepsy (MTLE).
Neuron23 – Series D – $96.5M – June 2025
Neuron23 closed a $97 million series D financing led by a healthcare investor with participation from Westlake Village BioPartners, SoftBank Vision Fund 2, Redmile, Blue Owl, Kleiner Perkins, HBM Healthcare Investments, and Acorn Bioventures. Proceeds will support the global Phase 2 NEULARK trial of NEU-411, a selective, brain-penetrant LRRK2 inhibitor for early Parkinson’s disease. The randomized, double-blind study (NCT06680830) will enroll ~150 participants and evaluate efficacy, safety, pharmacokinetics, and pharmacodynamics over one year.
Vima Therapeutics – Series A – $60M – May 2025
Vima Therapeutics Launches with $60 million series A to Advance Oral Therapy for Dystonia. Vima Therapeutics launched with a $60 million series A financing led by Atlas Venture, with participation from Access Industries and Canaan. Proceeds will advance VIM0423, a potential first-in-class oral therapy for dystonia and related movement disorders. The company, based in Cambridge, Massachusetts, is addressing a significant unmet need in dystonia, a chronic neurological condition marked by involuntary muscle contractions. Isolated dystonia affects more than 100,000 people in the United States, while dystonia and related movement disorders together impact over 1 million Americans.
Also check out Neurology R&D Partnerships, M&A and Venture Funding by Tech – Q1 2025 Review