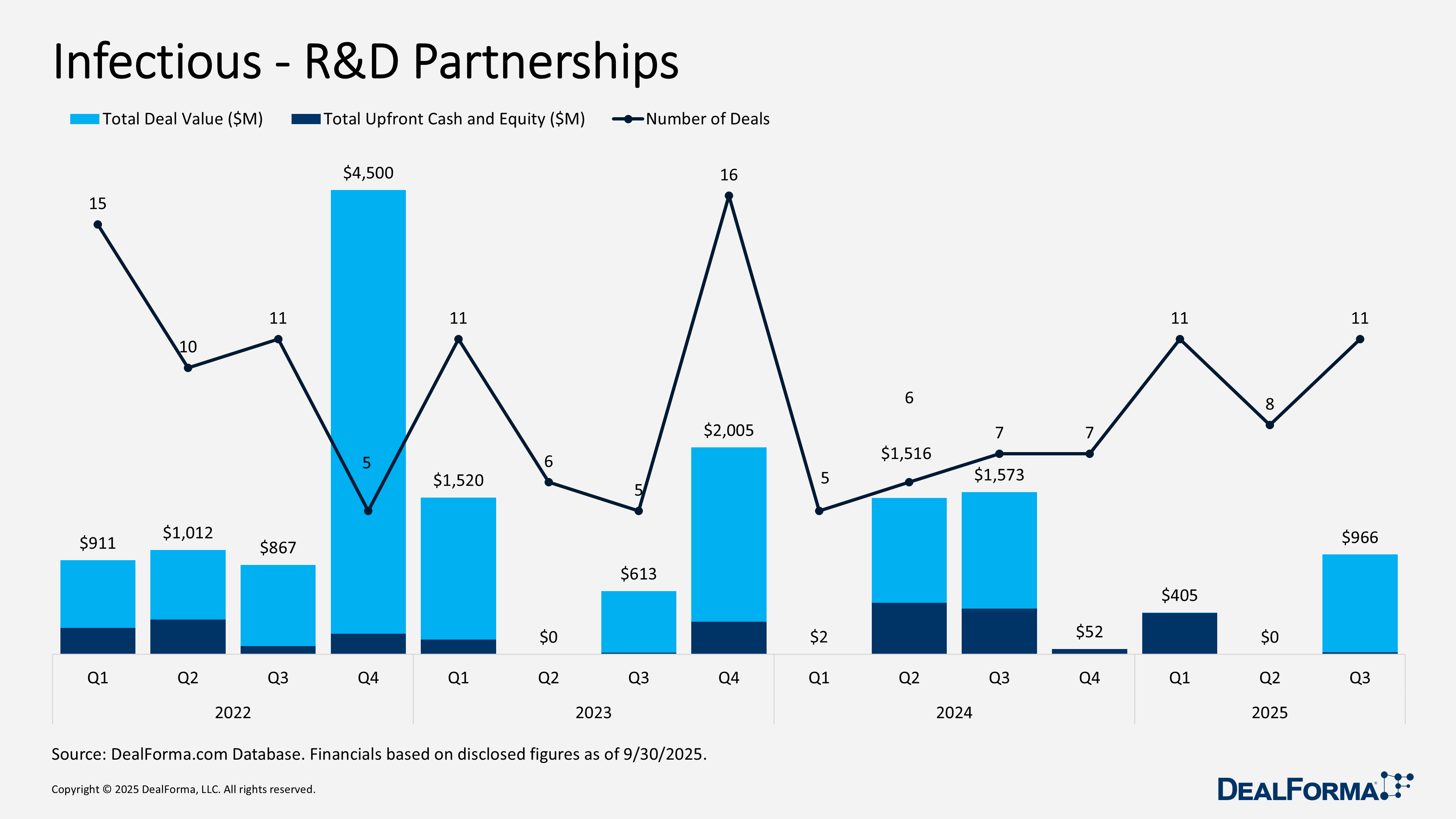
Q3 2025 saw a strong rebound in infectious disease transactions, with vigorous activity across R&D partnerships, M&A, and venture funding, reflecting improved confidence in clinical-stage assets and platform technologies. R&D partnerships totaled 11 deals worth $966 million with $19 million upfront, up from a subdued Q2. Key deals include Shionogi–BioVersys (July 2025) for preclinical BV‑500 targeting non-tuberculous mycobacteria (CHF 5 million/$6 million upfront, CHF 479 million/$601 million milestones, tiered royalties); VenatoRx–Basilea (August 2025) for Phase I/III-ready oral BL/BLI combo for complicated UTIs (up to $325 million commercial milestones); and MaaT Pharma–Clinigen (July 2025) for Phase III Xervyteg in GvHD (EUR 11 million/$12 million upfront, EUR 18 million/$21 million milestones, mid-30% royalties).
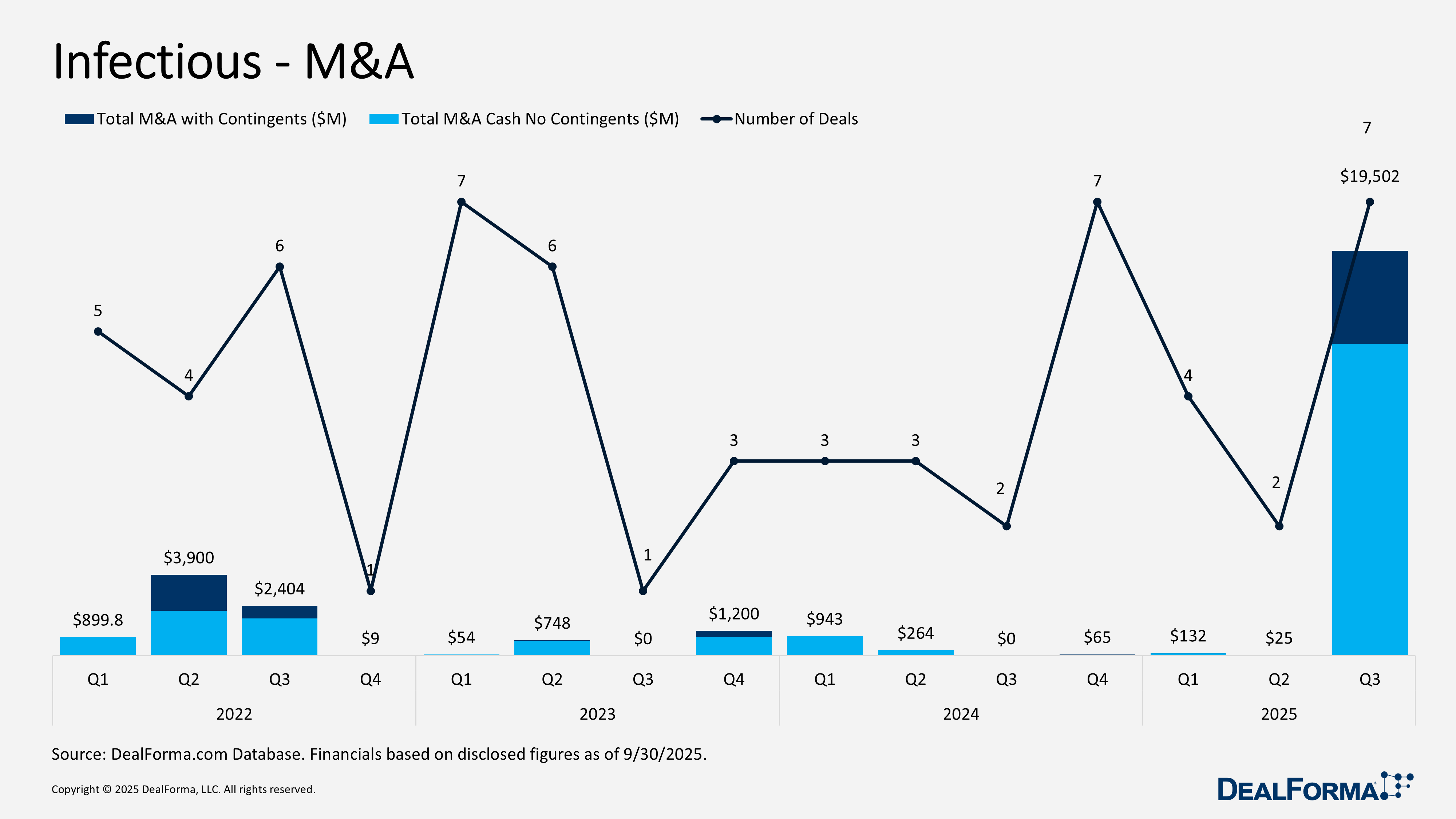
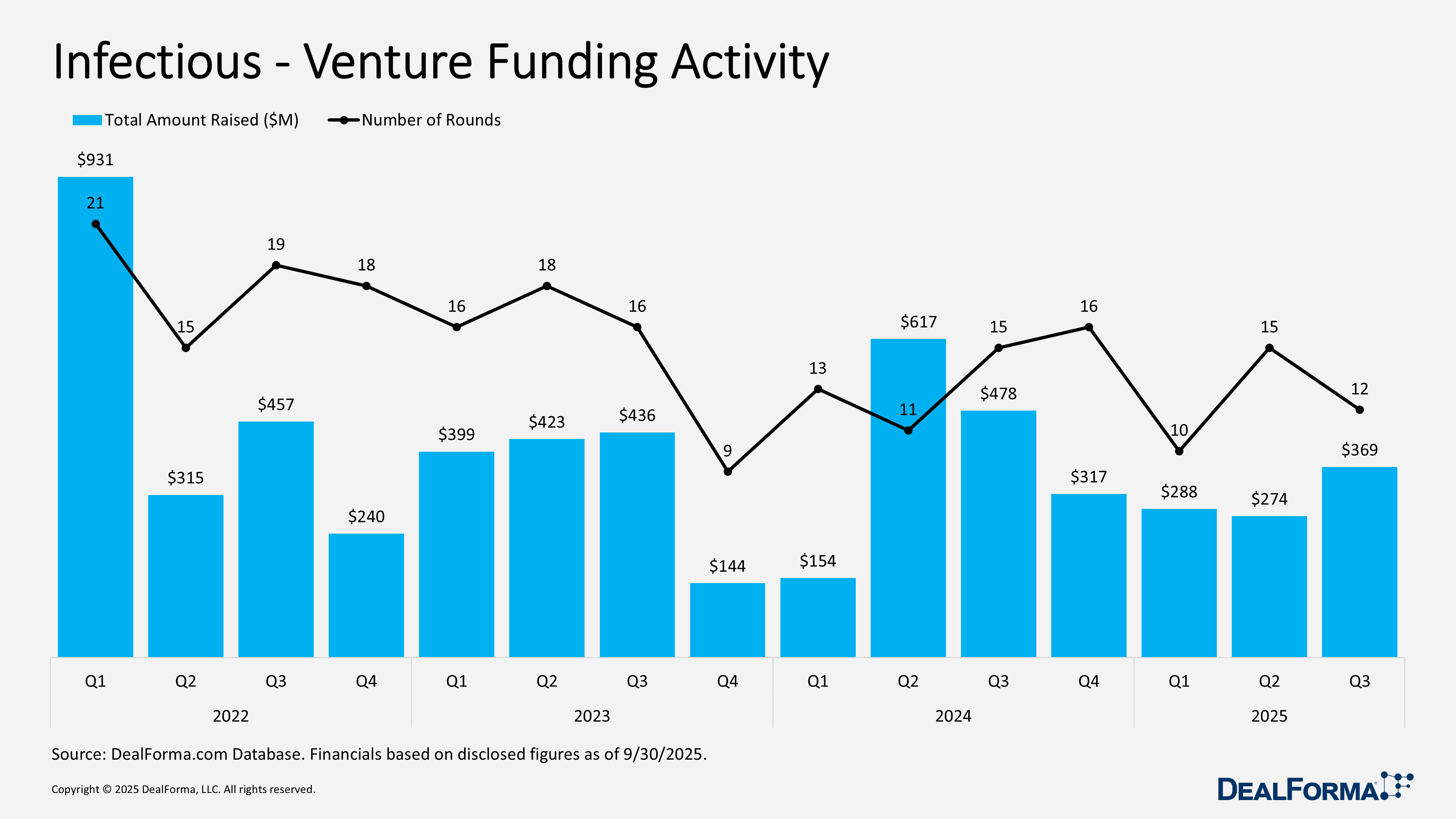
M&A surged with 7 deals totaling $19.5 billion, including contingents ($15 billion cash), versus 2 small deals in Q2. Notable transactions: BD–Waters (July 2025) Reverse Morris Trust ($17.5 billion, $4 billion pre-closing cash, $4 billion debt assumed, $9.5 billion equity, pro forma 2025 revenue $6.5 billion, adj. EBITDA $2 billion, 2030 targets $9 billion revenue/$3.3 billion EBITDA/32% margin); Sanofi–Vicebio (July 2025) for Phase I RSV/hMPV vaccines ($1.2 billion upfront, $450 million milestones, closed Dec 4, 2025); CorMedix–Melinta (Aug 2025) for approved antifungal and antibiotics ($300 million, $260 million cash/$40 million equity, up to $25 million regulatory milestones, closed Sept 2, 2025). Venture funding totaled 12 rounds for $369 million versus $274 million in Q2, reflecting larger financings. Key rounds: AusperBio series B2 ($63 million, Sept 2025) for AHB‑137 and Med‑Oligo™ platform; Mid-Atlantic Biotherapeutics ($50 million, Aug 2025) for IMT504 immunotherapy; Centivax series A ($45 million, July 2025) for universal influenza vaccine entering Phase I, HAI assay across 20+ strains. Overall, Q3 2025 marked a clear resurgence in infectious disease investments.
Infectious R&D Partnerships
In Q3 2025, infectious disease R&D partnerships rebounded strongly, with 11 deals generating $966 million in total value and $19 million in upfront cash or equity. In contrast, Q2 2025 activity was subdued, with 8 deals but no disclosed deal values or upfront payments, suggesting largely undisclosed financial terms. Across Q2 and Q3 combined, the sector recorded 19 partnerships totaling $966 million in disclosed value and $19 million upfront, with all reported economics concentrated in Q3.
Top Infectious R&D Partnerships in Q3 2025
Shionogi research partnership with BioVersys – July 2025
BioVersys entered a preclinical research partnership with Shionogi to co-develop BV-500, a small molecule therapy for non-tuberculous mycobacterial infections, combining BioVersys’ ansamycin chemistry platform with Shionogi’s infectious disease expertise. The agreement includes up to CHF 5 million, approximately $6 million, in upfront and near-term research payments, with potential milestone payments of up to CHF 479 million, approximately $601 million, tied to regulatory and commercial progress, plus tiered royalties contingent on Shionogi exercising its license option.
VenatoRx Pharmaceuticals development and commercialization deal with Basilea – August 2025
VenatoRx licensed exclusive worldwide rights to Basilea to develop and commercialize ceftibuten, ledaborbactam, and etzadroxil, an oral small molecule BL BLI combination targeting complicated urinary tract infections, including pyelonephritis. The Phase I stage asset is described as Phase III ready. Financial terms include an undisclosed upfront payment, additional undisclosed development and regulatory milestones, and up to $325 million in commercial milestones, plus tiered mid-single digit royalties, implying total deal value could exceed the disclosed amount.
MaaT Pharma development and commercialization deal with Clinigen – July 2025
MaaT Pharma licensed exclusive European rights to Clinigen to develop and commercialize Xervyteg (MaaT013), an immunotherapy for graft versus host disease, at the Phase III stage. The EMA accepted the marketing authorization application in June 2025, reducing regulatory risk. Under the agreement, MaaT Pharma will supply the finished product to Clinigen at a fixed unit price and receive EUR 11 million, approximately $12 million, upfront, with potential regulatory and commercial milestones of up to EUR 18 million, approximately $21 million, plus mid 30% royalties, supporting a meaningful long term revenue opportunity.
Infectious M&A
In Q3 2025, infectious disease M&A activity accelerated sharply, with 7 transactions generating $19.5 billion in total announced value, including contingents, and $15 billion in cash. The roughly $4.5 billion gap reflects earn outs or milestone-based payments, pointing to large, multibillion-dollar deal structures. By comparison, Q2 2025 was muted, with just 2 deals totaling $25 million and no meaningful contingent component, indicating small transaction sizes. Across Q2 and Q3 combined, the sector recorded 9 M&A deals valued at $19.5 billion with contingents and $15.1 billion in cash.
Top Infectious M&A in Q3 2025
BD’s Biosciences & Diagnostic Solutions Business merged with Waters in a Reverse Morris Trust transaction – July 2025
BD will combine its Biosciences and Diagnostic Solutions business with Waters Corporation in a Reverse Morris Trust transaction, forming a life sciences and diagnostics company focused on flow cytometry, molecular diagnostics, and infectious disease testing. The combined company will operate as Waters, listed on the NYSE as WAT, with ownership split 60.8% to Waters shareholders and 39.2% to BD shareholders. The transaction values the deal at $17.5 billion, including a $4 billion pre-closing cash distribution to BD shareholders, approximately $4 billion of debt assumed by Waters, and $9.5 billion of equity issued to BD shareholders. Pro forma 2025 revenue is expected to be $6.5 billion with $2 billion in adjusted EBITDA. Management targets $345 million in annualized EBITDA synergies by 2030, revenue of $9 billion, adjusted EBITDA of $3.3 billion, and a 32 percent operating margin by 2030. The transaction is expected to close by the end of Q1 2026.
Sanofi acquiring Vicebio – July 2025
Sanofi acquired Vicebio, including its Phase I RSV and hMPV vaccines (VXB‑241, VXB‑211), preclinical trivalent vaccine VXB‑251, two preclinical antivirals for avian influenza and SARS‑CoV, and the molecular clamp technology licensed from UniQuest. Vicebio shareholders received $1.2 billion upfront and are eligible for up to $450 million in development and regulatory milestones. The transaction closed on December 4, 2025, and supports Sanofi’s multivalent vaccine development strategy.
CorMedix acquiring Melinta Therapeutics – August 2025
CorMedix acquired Melinta Therapeutics, including its approved antifungal Rezzayo for candidemia and invasive candidiasis, which is in Phase III for prophylactic use in allogeneic bone marrow transplant patients (trial completion 1H 2026), along with key antibiotics Kimyrsa, Orbactiv, Vabomere, Minocin, Baxdela, and Toprol‑XL. Melinta shareholders received $300 million, $260 million in cash, and $40 million in equity via pre‑funded warrants and are eligible for up to $25 million in regulatory milestones tied to Rezzayo prophylaxis approval, plus tiered royalties on Rezzayo and low single‑digit royalties on Minocin US net sales. The transaction closed on September 2, 2025.
Infectious Venture Funding Activity
In Q3 2025, infectious disease venture funding improved, with 12 rounds raising $369 million, reflecting higher capital deployment despite fewer financings than the prior quarter. By contrast, Q2 2025 recorded 15 rounds but raised only $274 million, signaling smaller average deal sizes. Across Q2 and Q3 combined, the sector attracted $643 million across 27 rounds, with Q3 accounting for 57% of total capital while Q2 contributed more transactions.
Top Infectious Venture Funding Activity – Q3 2025
AusperBio – Series B2 – $63M – September 2025
AusperBio Therapeutics closed a $63 million series B2 financing to advance its targeted oligonucleotide therapies for a functional cure of chronic hepatitis B. The round was co-led by Qiming Venture Partners and a global strategic investor, with participation from existing backers including HanKang Capital, CDH Investments, YuanBio Venture Capital, Sherpa Capital, and Genesis Capital. Proceeds will support clinical development of lead candidate AHB‑137, including pivotal and Phase II combination trials, global Phase II expansion, advancement of the Med‑Oligo™ ASO platform, preclinical pipeline progression, scaling commercial manufacturing, and long-term growth.
Mid-Atlantic BioTherapeutics – Series Unspecified – $50M – August 2025
Mid‑Atlantic Biotherapeutics (MABT), a Doylestown, PA-based biotech developing immunotherapy for neurological disorders and cancer, received up to $50 million from Legend Innovation Life Science Fund to support clinical development, research, and regulatory activities. The company’s proprietary IMT504 platform leverages the body’s immune response and has demonstrated safety and efficacy in multiple primate models and humans. Founded in 2011 and led by CEO Dr. David Horn, MABT focuses on neurodegenerative diseases such as Alzheimer’s, Parkinson’s, and ALS, aiming to address hard-to-treat conditions through advanced biotechnology and AI-driven approaches.
Centivax – Series A – $45M – July 2025
Centivax, a biotech company developing universal vaccines and therapies, has closed an oversubscribed $45 million series A round led by Steve Jurvetson of Future Ventures, with participation from NFX, BOLD Capital Partners, Base4 Capital, Kendall Capital Partners, Amplify Partners, and existing investors. The funding will advance Centivax’s first clinical candidate, a universal influenza vaccine, through CMC development into a Phase I trial within eight months. The study will assess the safety and efficacy of the hemagglutination inhibition (HAI) assay against more than 20 influenza strains, including current, historical mismatch, and pandemic strains, providing a benchmark comparison against standard-of-care vaccines to demonstrate its broad protective potential.
Also check out Infectious Disease R&D Partnerships, M&A, Ventures, and IPO Activity – 2024 Review