During 2025, global healthcare and life sciences saw a year of strategic scale, innovation, and record capital deployment across R&D, M&A, venture funding, and IPOs. R&D partnerships totaled 685 deals worth $273.6 billion, driven by large, milestone-heavy agreements including Hengrui Pharma’s $500 million upfront, up to $12 billion PDE3/4 inhibitor deal with GSK for COPD (July); Innovent Biologics’ $1.2 billion oncology partnership with Takeda for NSCLC and gastric cancer (October); BioNTech’s $1.5 billion bispecific antibody collaboration with BMS for multiple solid tumors (June); 3SBio’s $1.3 billion PD-1/VEGF bispecific antibody deal with Pfizer for NSCLC, metastatic colorectal, and gynecological cancers (Phase III 2025); and XtalPi’s AI-driven molecular glue collaboration with DoveTree across oncology, immunology, inflammation, neurology, and metabolic diseases, valued up to $5.9 billion (August).
M&A activity surged with 585 deals worth $269.3 billion, highlighted by Abbott’s $23 billion acquisition of Exact Sciences for cancer diagnostics (November); Blackstone and TPG’s $18.3 billion Hologic buyout (October); BD’s $17.5 billion Biosciences & Diagnostic Solutions merger with Waters (July); Johnson & Johnson’s $14.6 billion Intra-Cellular Therapies acquisition for CNS and psychiatric disorders (January); and Novartis’ $12 billion purchase of Avidity Biosciences for xRNA neuromuscular and rare disease programs (October).
Venture funding reached $50 billion across 1,197 rounds, featuring BVI Medical’s $1 billion ophthalmic device raise (March); OURA’s $900 million health and wearable tech round (October); Neuralink’s $650 million series E for brain-computer interface expansion (June); Kailera Therapeutics’ $600 million series B for next-generation obesity therapies (October); and Isomorphic Labs’ $600 million AI drug discovery round under Alphabet (March).
Public markets rebounded with 48 IPOs raising $14.1 billion, led by Medline’s $7.2 billion medical-surgical debut (October); Caris Life Sciences’ $494 million AI-driven molecular profiling listing (May); Lumexa Imaging’s $463 million diagnostic imaging IPO (November); Hinge Health’s $437 million digital musculoskeletal care offering (March); and Heartflow’s $364 million Nasdaq listing for AI coronary diagnostics (July), reflecting a robust year of scale, innovation, and capital inflow.
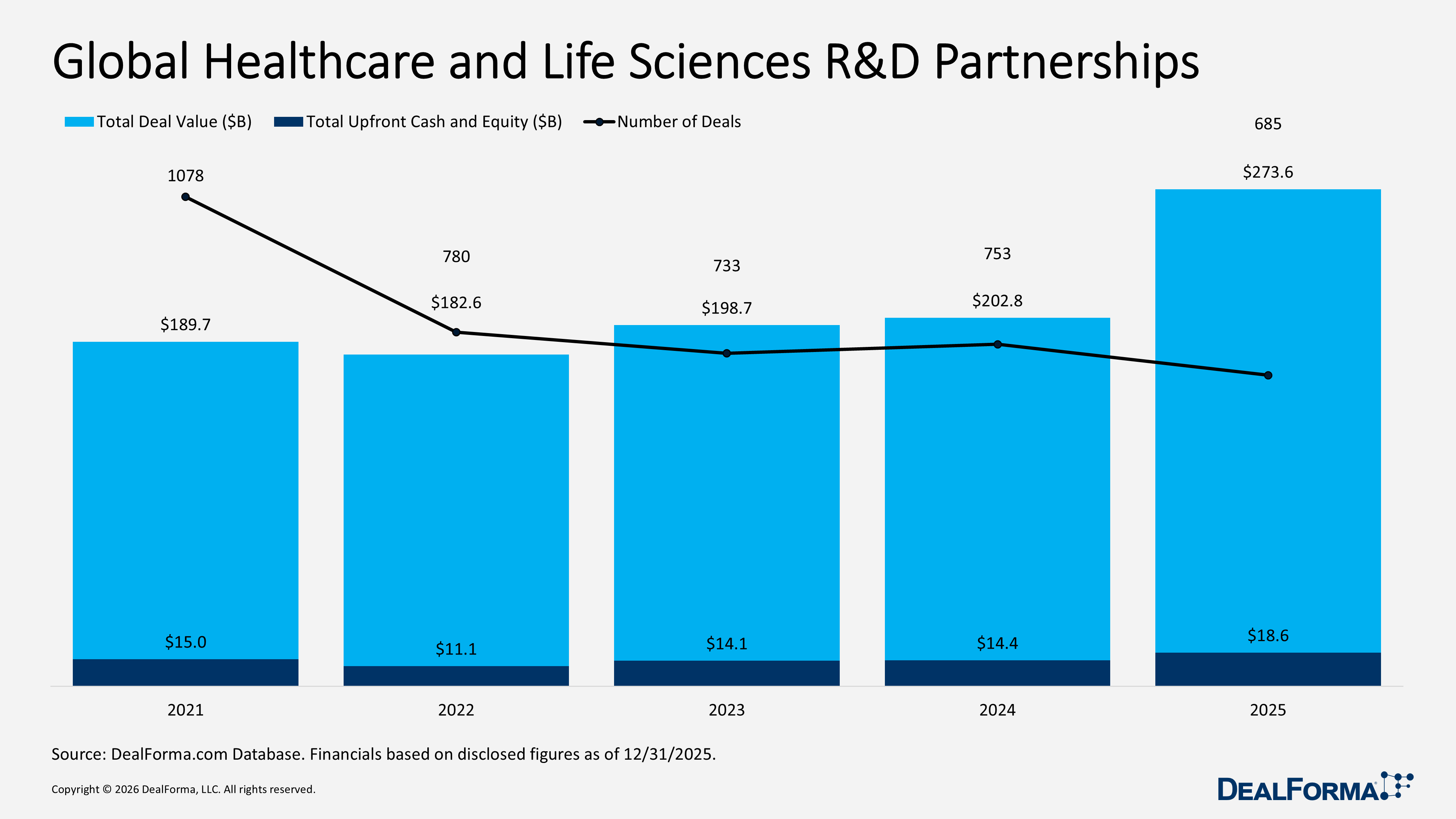
Global Healthcare and Life Sciences R&D Partnerships
In 2025, global healthcare and life sciences R&D partnerships declined to 685 deals but generated a higher $273.6 billion in total value, a shift toward fewer, larger transactions. Average deal size increased, while upfront cash and equity remained unchanged at $97 million, indicating that most value continued to be driven by milestone and contingent payments rather than significant upfront commitments. By comparison, 2024 recorded a higher 753 deals but a lower $202.8 billion in total value, with $76 million in upfront funding, pointing to a predominantly mid-sized deal environment. Across 2024 and 2025 combined, the sector completed 1,438 partnerships totaling $476.3 billion, supported by $33 billion in upfront cash and equity.
Top 5 R&D Partnerships Global Healthcare and Life Sciences – 2025
Hengrui Pharma development and commercialization deal with GSK – July 2025
GSK licensed exclusive worldwide rights outside Greater China from Hengrui Pharma to develop and commercialize HRS-9821, a Phase I small-molecule PDE3/4 inhibitor for COPD, with development expected to include a dry-powder inhaler formulation. The agreement also grants GSK exclusive options to license up to 11 additional early-stage programs after Phase I development by Hengrui. Financial terms include a $500 million upfront payment and up to $12 billion in potential development, regulatory, and sales milestones, plus tiered royalties.
Innovent Biologics development and commercialization deal with Takeda – October 2025
Takeda entered a broad oncology partnership with Innovent Biologics, securing exclusive worldwide rights outside China to develop and commercialize two late-stage cancer programs: IBI-363, a Phase III–ready PD-1/IL-2a-bias bispecific antibody for NSCLC, and IBI-343, a Claudin 18.2–targeting antibody drug conjugate in Phase III for gastric cancer. The deal also includes an option to license IBI-3001, an early-stage bispecific antibody drug conjugate for solid tumors. Financial terms comprise $1.2 billion upfront, including a $100 million equity investment, and up to $10.2 billion in potential milestones plus royalties. IBI-363 will be co-developed globally with shared costs and profits in the US on a 60/40 basis, while Takeda holds primary global commercialization and manufacturing responsibilities outside China.
BioNTech development and commercialization deal with BMS – June 2025
BioNTech granted Bristol Myers Squibb exclusive worldwide rights to develop and commercialize BNT-327, a bispecific antibody targeting PD-L1 and VEGF-A for multiple solid tumors, with a broad late-stage clinical program including global Phase III trials in small cell and non-small cell lung cancer and a planned Phase III study in triple-negative breast cancer by year-end 2025. Financial terms include $1.5 billion upfront, up to $2 billion in non-contingent anniversary payments through 2028, and up to $7.6 billion in development, regulatory, and commercial milestones. The partners will share development and manufacturing costs and global profits equally, with BioNTech booking sales in the US and retaining key market authorizations, while BMS books sales in the rest of the world.
3SBio development and commercialization deal with Pfizer – July 2025
Shenyang Sunshine Pharmaceutical and 3SBio’s subsidiary, 3S Guojian, granted Pfizer exclusive worldwide rights outside China to develop, manufacture, and commercialize SSGJ-707, a PD-1/VEGF bispecific antibody for NSCLC, metastatic colorectal cancer, and gynecological cancers. The agreement includes options for Pfizer to commercialize the drug in China and to manufacture it in the US. Financial terms comprise $1.3 billion upfront, a $100 million equity investment, up to $4.8 billion in development, regulatory, and commercial milestones, and double-digit tiered royalties, with an additional $150 million option fee payable in July 2025. The program is set to enter Phase III trials in China in 2025.
XtalPi development and commercialization deal with DoveTree – August 2025
XtalPi entered a broad AI-driven drug discovery partnership with DoveTree, granting exclusive worldwide rights to develop and commercialize molecular glue therapies across oncology, immunology, inflammation, neurology, and metabolic diseases. The collaboration combines XtalPi’s AI and machine learning platform with DoveTree’s biological and R&D capabilities. Financial terms include $51 million upfront, up to $49 million in near-term payments, and up to $5.9 billion in development and commercial milestones, plus tiered royalties.
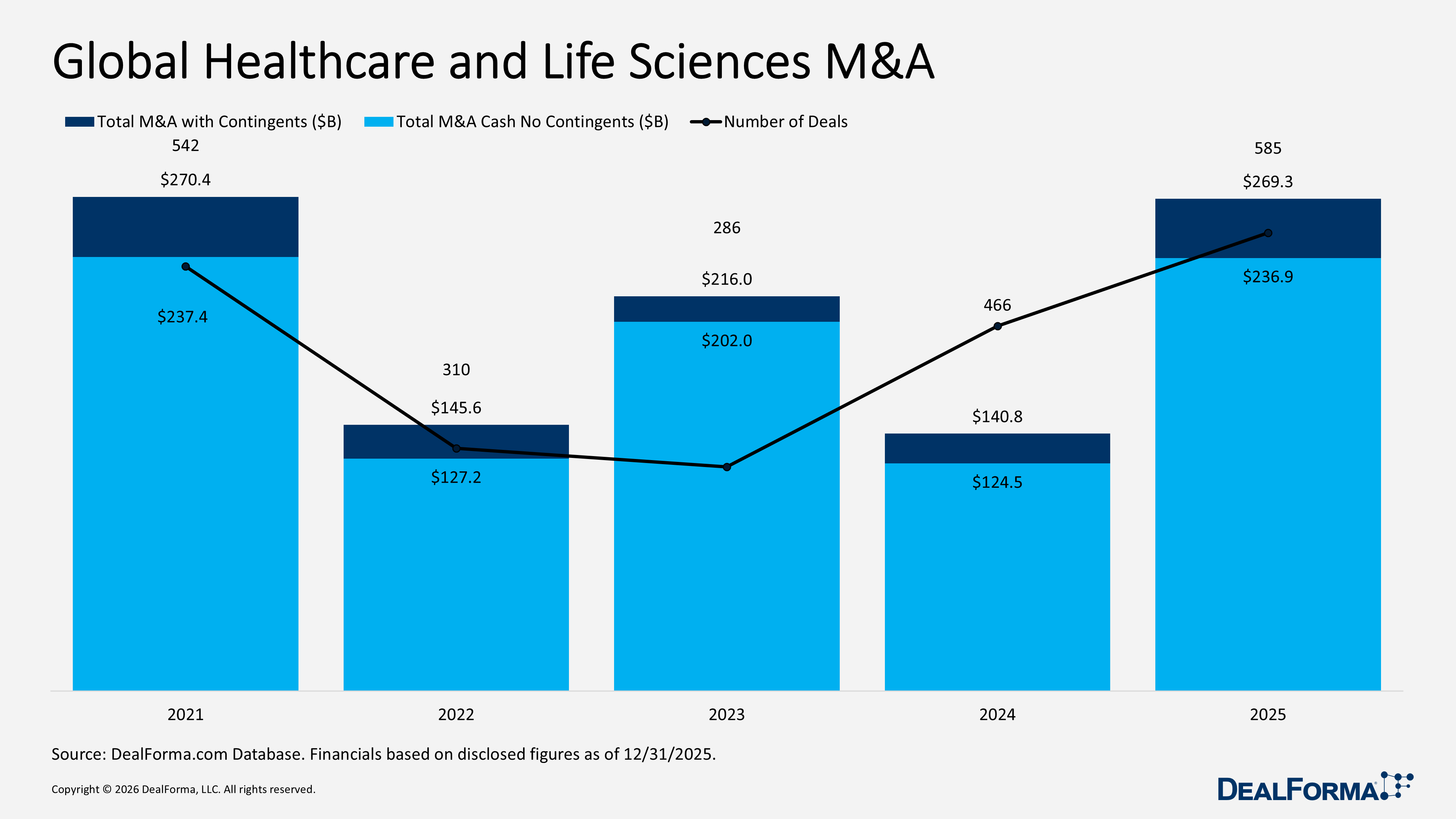
Global Healthcare and Life Sciences M&A
In 2025, global healthcare and life sciences M&A activity accelerated significantly, with 585 transactions totaling $269.3 billion, including contingents, or $236.9 billion on a cash and no-contingent basis. This reflects both higher deal volumes and materially larger transaction sizes, with average deal value rising to approximately $461 million, including contingents, or $406 million in cash terms. Earnouts and contingent considerations accounted for $32.3 billion, representing about 12% of the total announced value. By contrast, 2024 recorded 466 deals with $140.8 billion in total value, including contingents, or $124.5 billion on a cash basis, equating to roughly $302 million per deal, indicating a smaller and lower-priced M&A environment. Across 2024 and 2025 combined, the sector completed 1,049 M&A transactions valued at $409.8 billion, including contingents, or $361.2 billion in cash terms. The data highlight a clear inflection in 2025, which drove a 25% increase in deal volume and an approximately 91% increase in headline value compared with 2024.
Top 5 M&A Partnerships Global Healthcare and Life Sciences – 2025
Abbott acquiring Exact Sciences – November 2025
Abbott agreed to acquire Exact Sciences, a leader in cancer screening and diagnostics, in a cash transaction valuing the company at approximately $21 billion in equity and $23 billion in enterprise value. Exact Sciences’ portfolio includes Cologuard and Cologuard Plus for noninvasive colorectal cancer screening, blood based multi cancer detection tests, and advanced molecular diagnostics supporting early detection, treatment selection, and recurrence monitoring. Shareholders will receive $105 per share, representing a 51 percent one-day premium. The transaction is expected to close in Q2 2026.
Blackstone and TPG acquiring Hologic for $18.3B – October 2025
Blackstone and TPG agreed to acquire Hologic in an all-cash transaction valued at $18.3 billion, taking the diagnostics company private. The offer provides shareholders with $76 per share in cash, representing approximately $17.6 billion in upfront consideration, a 6 percent one-day premium, and a 46 percent premium to the last unaffected closing price. Shareholders are also eligible to receive up to $3.00 per share, or about $700 million in aggregate, through a contingent value right tied to the performance of Hologic’s breast health business. The CVR pays up to $1.50 per share in 2026 if revenues exceed $1.6 billion and up to $1.50 per share in 2027 if revenues exceed $1.7 billion.
BD’s Biosciences & Diagnostic Solutions Business merged with Waters – July 2025
BD agreed to merge its Biosciences and Diagnostic Solutions business with Waters Corporation through a Reverse Morris Trust transaction, creating a combined life sciences and diagnostics company listed on the NYSE under the Waters name. The deal values the transaction at $17.5 billion, including a $4 billion cash distribution to BD shareholders, approximately $4 billion of assumed debt, and a 39.2% equity stake in the combined entity valued at about $9.5 billion, with Waters shareholders retaining 60.8 percent ownership. On a pro forma basis, the combined company is expected to generate $6.5 billion in revenue and $2 billion in adjusted EBITDA in 2025, with $345 million in annualized EBITDA synergies by 2030 and a long-term target of $9 billion in revenue, $3.3 billion in adjusted EBITDA, and a 32% operating margin by 2030.
Johnson & Johnson acquiring Intra-Cellular Therapies – January 2025
Johnson & Johnson acquired Intra-Cellular Therapies for approximately $14.6 billion in cash, paying $132 per share, a 40 percent one-day premium. The deal adds Intra-Cellular’s approved Caplyta for schizophrenia, bipolar disorder, depression, sleep disorders, and other indications, along with a pipeline including Phase II lenrispodun for Parkinson’s disease and heart failure, lumateperone deuterated for anxiety and psychosis, Phase I ITI-1020 for cancer, ITI-333 for pain, and preclinical neurological programs. The acquisition, completed on April 2, 2025, establishes Intra-Cellular as a dedicated J&J business unit.
Novartis acquiring Avidity Biosciences – October 2025
Novartis announced the acquisition of Avidity Biosciences for approximately $12 billion in cash, paying $72 per share, a 46 percent one-day premium, with an enterprise value of roughly $11 billion. Avidity’s pipeline includes Phase III Delpacibart etedesiran for myotonic dystrophy type 1, Phase II programs for FSHD and Duchenne muscular dystrophy, preclinical neuromuscular assets, and other rare disease candidates. The deal accelerates Novartis’ xRNA strategy by integrating Avidity’s Antibody Oligonucleotide Conjugates platform, while certain non-core programs are being spun off into a separate entity (SpinCo). The transaction is expected to close in H1 2026.
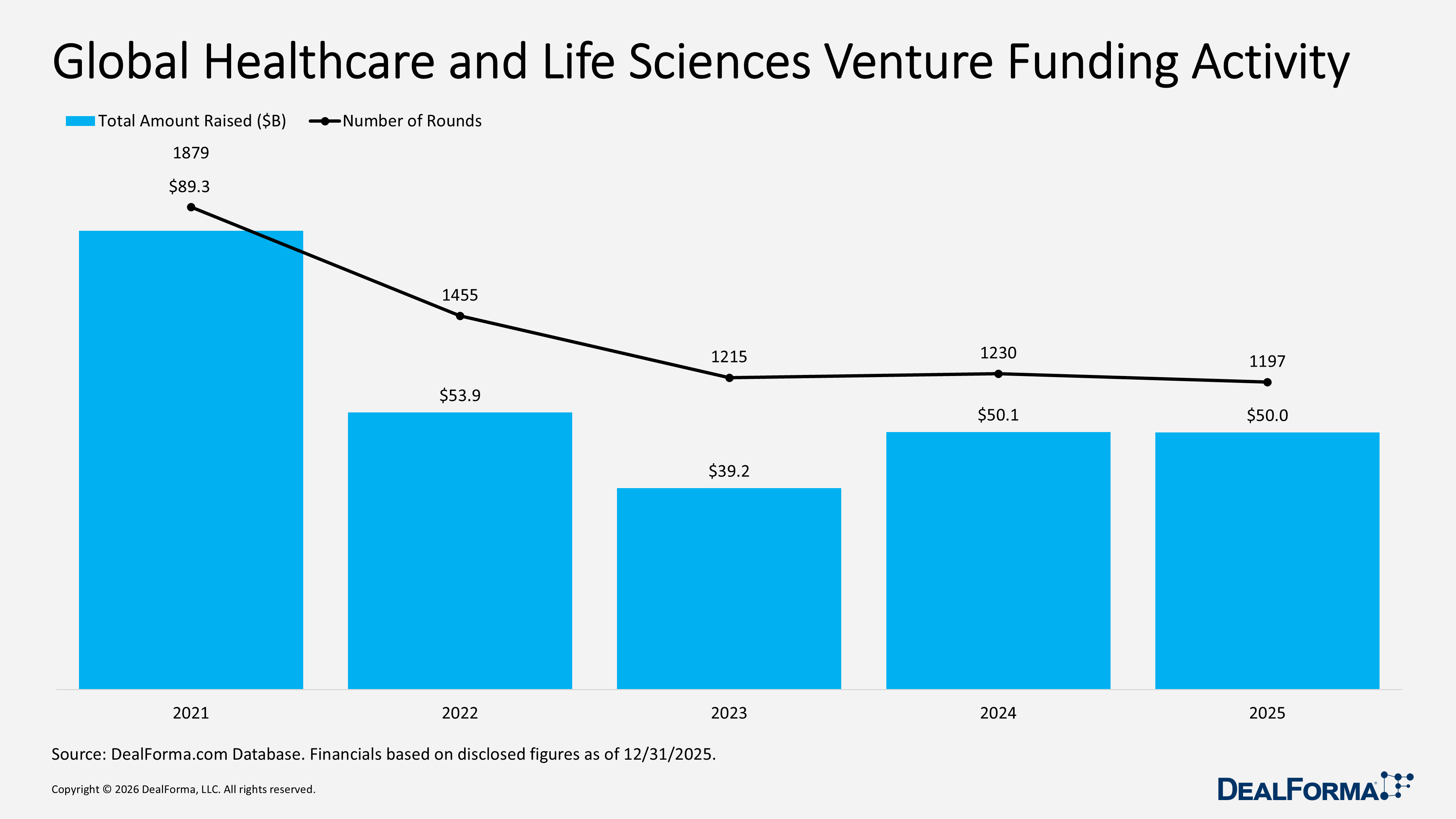
Global Healthcare and Life Sciences Venture Funding Activity
In 2025, global healthcare and life sciences venture funding increased substantially despite a slight decline in deal count. The market recorded 1,197 financings raising $50 billion, reflecting a sharp rise in average round size to approximately $42 million and signaling stronger capital deployment. By comparison, 2024 saw a marginally higher 1,230 rounds but raised only $50.1 billion, or about $41 million per round, indicating cautious funding with smaller check sizes. Across 2024 and 2025 combined, the sector completed 2,427 venture rounds totaling $100.1 billion. The key takeaway is that deal volume and total dollars remained very consistent, and the average check size in 2025 was marginally higher despite the small dip in total capital.
Top 5 Venture Funding Activity Global Healthcare and Life Sciences – 2025
BVI Medical – Series Unspecified -$1B – March 2025
BVI Medical completed a $1 billion strategic capital raise with TPG, fully refinancing existing debt and adding new equity to support accelerated growth and global expansion. The funding positions the company for multiple upcoming product launches and operational scaling to meet rising demand. BVI has recently expanded its IOL manufacturing in Europe, launched new products including SERENITY and SERENITY Toric premium monofocal IOLs, and strengthened its market presence through strategic acquisitions, reinforcing its leadership in ophthalmic device innovation.
Oura – Series Unspecified – $900M – October 2025
OURA, maker of the Oura Ring, raised over $900 million in a funding round led by Fidelity Management & Research, with participation from ICONIQ, Whale Rock, and Atreides, valuing the company at approximately $11 billion. The capital will support AI-driven product innovation, global distribution expansion, and the development of new health features. Since its 2015 launch, OURA has sold over 5.5 million devices, with more than half sold in the past year. The company reported $500 million in revenue in 2024, doubling prior-year growth, and is on track to exceed $1 billion in 2025.
Neuralink – Series E – $650M – June 2025
Neuralink, Elon Musk’s brain-computer interface startup, raised $650 million in a series E funding round, valuing the company at approximately $9 billion pre-money. The round was backed by investors including ARK Invest, DFJ Growth, Founders Fund, Sequoia Capital, and others. The funding will support expanded patient access and the development of new devices. To date, Neuralink has raised over $1.2 billion, including a $280 million series D in August 2023. The company’s technology aims to enable paralyzed patients to control digital devices using only their minds.
Kailera Therapeutics – Series B – $600M – October 2025
Kailera Therapeutics, a clinical-stage biopharmaceutical company focused on next-generation obesity therapies, raised $600 million in a series B financing led by Bain Capital Private Equity. The fully funded round will support the global Phase 3 program for its lead candidate, KAI-9531, a dual GLP-1/GIP receptor agonist targeting best-in-class weight loss, with trials in adults with obesity or overweight, with and without type 2 diabetes, and in adults with BMI ≥35. The funding will also advance KAI-7535, an oral GLP-1 receptor agonist, to global trials, and support earlier-stage programs including KAI-4729, a GLP-1/GIP/glucagon tri-agonist, and a once-daily oral formulation of KAI-9531, positioning Kailera to expand its differentiated obesity portfolio.
Isomorphic Labs – Series A – $600M – March 2025
Isomorphic Labs, an AI-driven drug discovery company under Alphabet, raised $600 million in its first external funding round since its founding in 2021. The round was led by Thrive Capital, with participation from GV and additional Alphabet capital. The funding will support the development of Isomorphic’s next-generation AI drug design platform and advance its internal programs toward clinical development. The company, known for AlphaFold 3 in collaboration with DeepMind, previously secured deals with Eli Lilly and Novartis valued at nearly $3 billion combined, leveraging AI to discover small molecules for undisclosed targets.
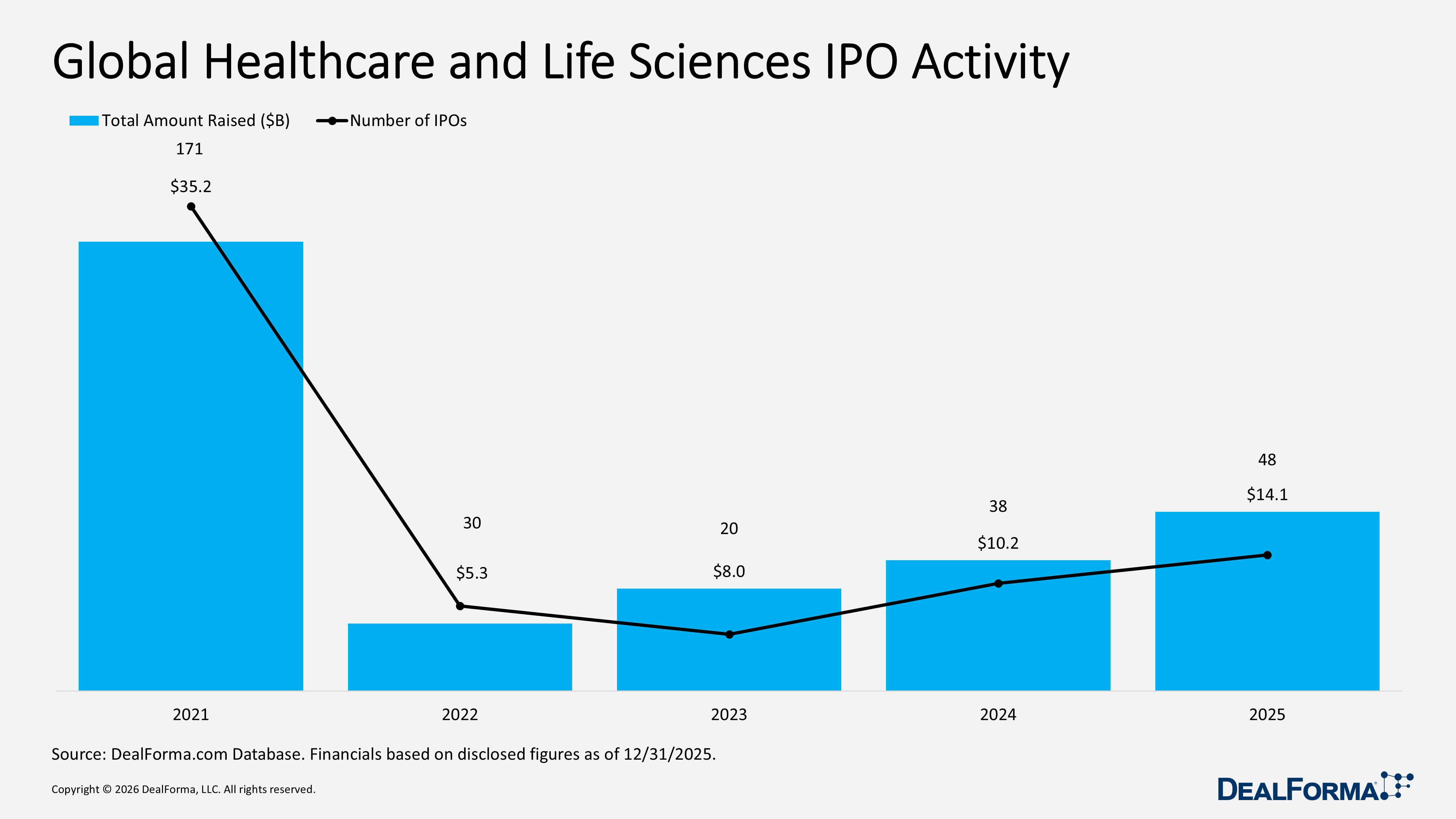
Global Healthcare and Life Sciences IPO Activity
In 2025, the global healthcare and life sciences IPO market experienced strengthening, with 48 offerings raising $14.1 billion, reflecting an improved public market environment and a higher average raise of approximately $294 million per IPO. By comparison, 2024 recorded 38 IPOs raising $10.2 billion, or about $268 million per offering, indicating fewer listings and more modest capital raises. Across 2024 and 2025 combined, the sector delivered 86 IPOs, generating $24.3 billion in proceeds. The recovery was led by 2025, with IPO volume up roughly 26% and total capital raised increasing by 38% year-over-year.
Top 5 IPO Activity Global Healthcare and Life Sciences – 2025
Medline – IPO – $7.2B – October 2025
Medline, a leading provider of medical-surgical products and supply chain solutions, completed an upsized IPO of 248.4 million Class A shares at $29 per share, raising approximately $7.2 billion. The offering included the full exercise of the underwriters’ option for 32.4 million shares. Proceeds will repay $179 million in outstanding senior secured term loans, with the remainder used for general corporate purposes and to redeem pre-IPO equity from certain shareholders. Medline, backed by Blackstone, Carlyle, and Hellman & Friedman, employs over 43,000 people and operates in more than 100 countries, delivering products and solutions that enhance clinical, financial, and operational outcomes for healthcare providers.
Caris Life Sciences – IPO – $494.1M – May 2025
Caris Life Sciences priced its IPO at $21 per share, offering 23.5 million shares with a 30-day underwriter option for an additional 3.5 million shares, raising over $494 million and valuing the company at approximately $5.9 billion. Shares began trading on Nasdaq under the ticker “CAI,” opening at $27 and closing at $28, marking a 29-33% increase over the IPO price and signaling strong demand. Caris, a next-generation AI TechBio and precision medicine company, recently launched MI Cancer Seek, the first FDA-approved companion diagnostic using simultaneous whole exome and transcriptome sequencing for solid tumors, and Caris Assure, a blood-based assay for therapy selection. The company leverages advanced AI and a large multimodal molecular database to analyze disease complexity, positioning it as a leader in molecular profiling and precision oncology.
Lumexa Imaging – IPO – $462.5M – November 2025
Lumexa Imaging, a US diagnostic imaging provider backed by Welsh, Carson, Anderson & Stowe, raised $463 million in its IPO, selling 25 million shares at $18.50 each and valuing the company at $1.8 billion. Formerly US Radiology Specialists, Lumexa operates 184 imaging centers across 13 states, having grown through more than a dozen acquisitions since its 2018 formation. For the nine months ending September 30, the company reported a net loss of $18 million on $755 million in revenue, improving from a $69 million loss on $701 million a year earlier. Lumexa will trade on Nasdaq under the ticker LMRI, with Barclays, JP Morgan, and Jefferies as joint bookrunners.
Hinge Health – IPO – $437.3M – March 2025
Hinge Health, a digital musculoskeletal care company, raised approximately $437 million in its NYSE IPO, selling 9.14 million shares under the ticker HNGE. Founded in 2014, the San Francisco-based company provides comprehensive musculoskeletal care through health coaches, orthopedic surgeons, physical therapists, and digital tools, including its FDA-cleared Enso wearable for pain relief and a specialized pelvic health program. Shares opened at $39.25, a 23% premium over the $32 IPO price, and closed at $37.56, reflecting strong confidence in Hinge Health’s growth and diversified care. Hinge also offers its FDA-cleared wearable Enso, which provides electrical pulses to relieve everyday pain, and a women’s pelvic health program.
Heartflow – IPO – $364.2M – July 2025
Heartflow, a leader in AI-driven coronary artery disease (CAD) technology, raised approximately $364 million in an upsized Nasdaq IPO, selling 19.17 million shares at $19 each, including 2.5 million shares from the underwriters’ full option exercise. Trading under the ticker HTFL since August 8, 2025, Heartflow offers its non-invasive precision coronary care platform, Heartflow One, which integrates Roadmap™, FFRCT, and Plaque Analyses to support guideline-directed CCTA pathways. Backed by over 600 peer-reviewed publications, the platform has helped clinicians manage more than 400,000 patients globally, transforming CAD into a screenable, diagnosable, and manageable condition.
Also check out Global Healthcare and Life Sciences – Q1 2025 Review