Gene therapy in H1 2025 remained robust across hematology, neurology, rare diseases, oncology, and ophthalmology, with momentum led by late-stage assets, novel capsids, and LNP delivery platforms. On the R&D side, Vertex partnered with Orna (Jan) on gene-editing therapies for SCD and TDT, paying $65 million upfront with up to $635 million in milestones plus $365 million per product options. Eli Lilly (Apr) licensed Sangamo’s STAC-BBB AAV capsid for CNS gene therapies, for $18 million upfront and up to $1.4 billion in milestones. Nippon Shinyaku (Jan) secured rights to Regenxbio’s RGX-121/111 for Hunter and Hurler syndromes with $110 million upfront and up to $700 million in milestones/royalties.
On the M&A front, Carlyle and SK Capital (Feb) acquired Bluebird Bio, securing its three approved therapies (Lyfgenia, Zynteglo, Skysona) in a $29 million cash deal plus a CVR tied to $600 million in sales. AbbVie (Jun) agreed to buy Capstan for up to $2.1 billion, gaining its tLNP platform and pipeline in autoimmune, oncology, and fibrosis, including Phase I CAR-T candidate CPTX-2309. On the Venture side, appetite remained strong. Atsena raised $150 million (series C, Apr) to advance ATSN-201 for XLRS and its AAV.SPR platform. SpliceBio closed $135 million (series B, Jun) led by EQT/Sanofi for Stargardt disease (SB-007) and broader pipeline expansion. RhyGaze raised $86 million (series A, Jan) for its cone optogenetics vision-restoration platform, while Epicrispr secured $68 million (series B, Mar) to advance EPI-321, the first epigenetic therapy in trials for FSHD.
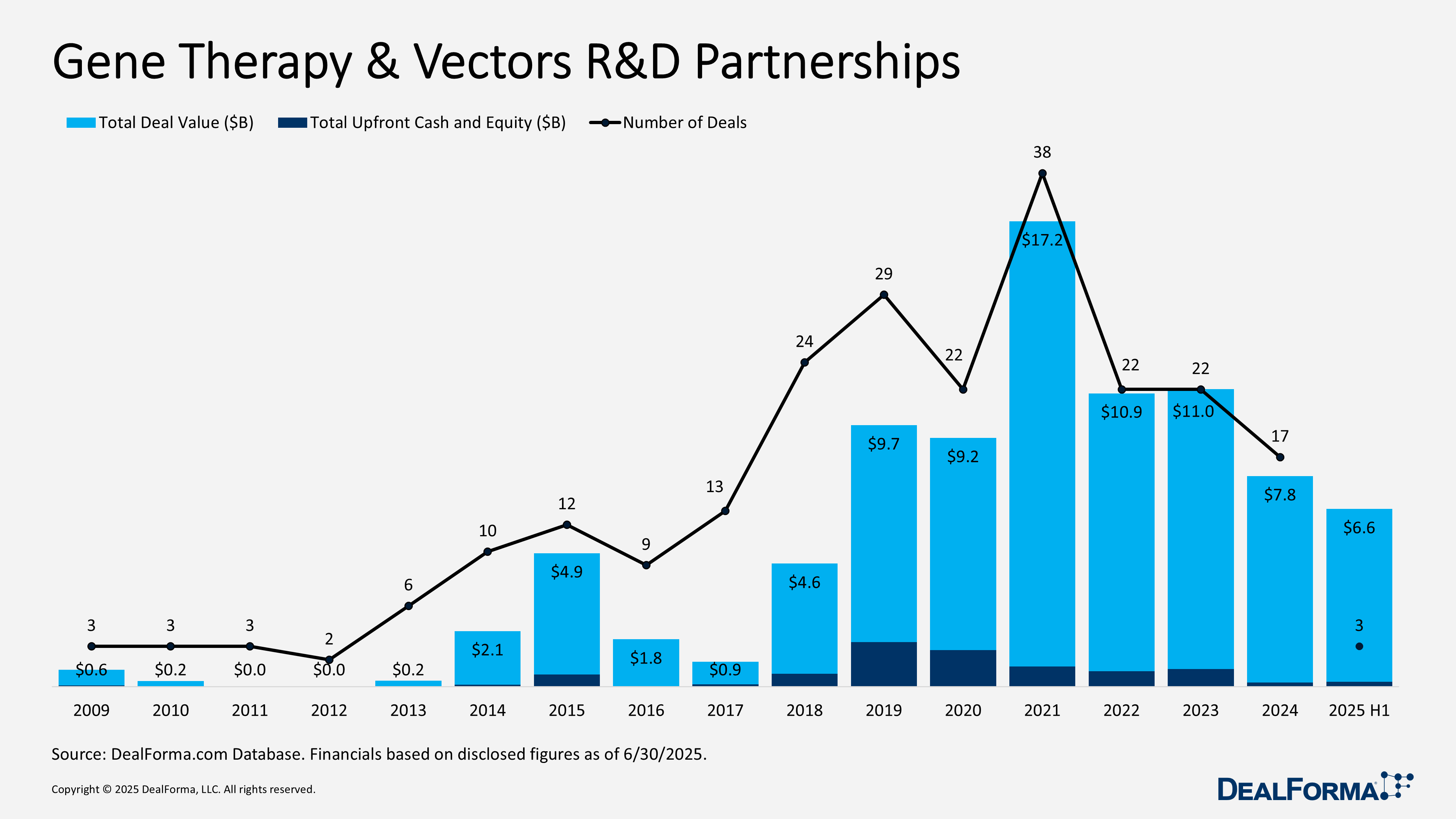
Gene Therapy & Vectors R&D Partnerships
In H1 2025, gene therapy and vector partnerships were limited in number but sizable in value. Only 3 signed partnerships, totaling $6.6 billion, with $200 million in upfront payments and an average of $64 million per deal, more than double the 2024 average. By comparison, 2024 recorded 17 partnerships worth $7.8 billion in total, with the same $200 million upfront but a lower $26 million per deal, reflecting earlier-stage assets and milestone-heavy structures. Together, 2024 through H1 2025 produced 20 transactions valued at $14.4 billion with about $400 million in upfront commitments.
Top Gene Therapy R&D Partnerships in H1 2025
Orna development and commercialization deal with Vertex – January 2025
Vertex secured rights to develop and commercialize undisclosed gene editing therapies for sickle cell disease (SCD) and transfusion-dependent beta-thalassemia (TDT) using Orna’s lipid nanoparticle (LNP) delivery platform for hematopoietic stem cells. The agreement runs for 3 years with an option to expand to 10 additional targets. Orna received $65 million upfront (including a convertible note) and could earn up to $635 million in milestones tied to SCD/TDT. If Vertex exercises its option, Orna may receive up to $365 million in additional milestones and option fees per product, plus tiered royalties. A high-value collaboration that strengthens Vertex’s gene editing pipeline in hematologic diseases, while validating Orna’s LNP delivery platform for advanced stem cell therapies.
Sangamo development and commercialization deal with Eli Lilly – April 2025
Eli Lilly obtained exclusive global rights to develop and commercialize CNS gene therapies using Sangamo’s STAC-BBB AAV capsid. The deal covers one initial target, with the option to add up to four more. Sangamo will handle technology transfer, while Lilly oversees development through commercialization. Terms include $18 million upfront and up to $1.4 billion in target fees and milestones across 5 potential neurology programs, plus tiered royalties. The partnership highlights Lilly’s continued push into CNS gene therapy and provides meaningful non-dilutive capital for Sangamo, with potential multi-billion–dollar upside tied to milestones.
Regenxbio development and commercialization deal with Nippon Shinyaku – January 2025
Nippon Shinyaku acquired exclusive rights to commercialize Regenxbio’s RGX-121 and RGX-111 gene therapies for Hunter and Hurler syndromes in the US and Asia. Regenxbio will manage clinical development, manufacturing, and supply, while retaining rights outside the licensed regions and proceeds from any Priority Review Voucher tied to RGX-121. Financial terms include $110 million upfront, up to $40 million in development and regulatory milestones, $660 million in sales milestones, and double-digit royalties. A late-stage gene therapy deal expanding Nippon Shinyaku’s rare disease portfolio, while securing Regenxbio significant upfront capital and future milestone potential.
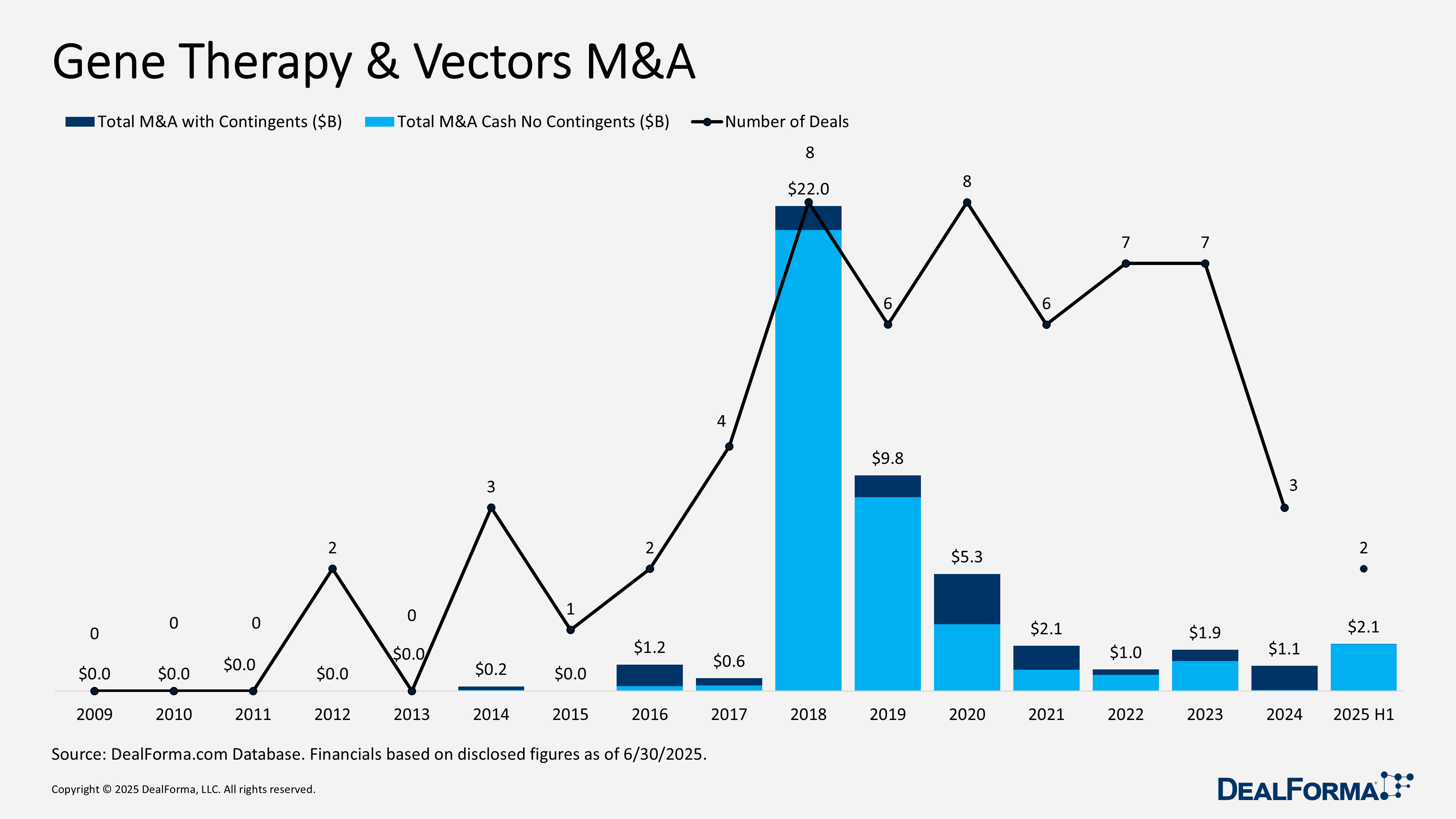
Gene Therapy & Vectors M&A
In H1 2025, gene therapy and vector M&A were limited in volume but striking in scale. Just two transactions reached $2.1 billion, entirely in upfront cash, averaging $1.1 billion per deal. In contrast, 2024 recorded 3 smaller deals worth a combined $1.1 billion, with virtually no upfront cash and an average of $37 million per deal, reflecting milestone-driven, early-stage structures. Together, 2024 through H1 2025 saw five transactions totaling $3.3 billion, of which $2.2 billion was paid upfront.
Top Gene Therapy M&A in H1 2025
AbbVie acquiring Capstan – June 2025
AbbVie agreed to acquire Capstan for up to $2.1 billion in upfront cash (subject to adjustments). Capstan’s pipeline includes CPTX-2309, a Phase I in vivo tLNP anti-CD19 CAR-T therapy for B–cell–mediated autoimmune diseases; CPTX-2506, a discovery-stage CAR-T targeting BCMA-expressing plasma cells for cancer and autoimmune indications; and additional undisclosed programs in immunology, oncology, and fibrosis. The acquisition also secures Capstan’s targeted lipid nanoparticle (tLNP) platform for delivering RNA payloads, such as mRNA, to engineer specific cell types in vivo.
Carlyle and SK Capital acquiring Bluebird Bio – February 2025
Carlyle and SK Capital acquired Bluebird Bio, taking the company private. Bluebird’s portfolio includes three approved gene therapies: Lyfgenia (sickle cell disease), Zynteglo (beta-thalassemia), and Skysona (adrenoleukodystrophy). Shareholders received $3 per share (~$29 million upfront cash) with the option of an additional $6.84 per share contingent value right (CVR) tied to $600 million in net sales by 2027, or $5 per share in cash with no CVR.
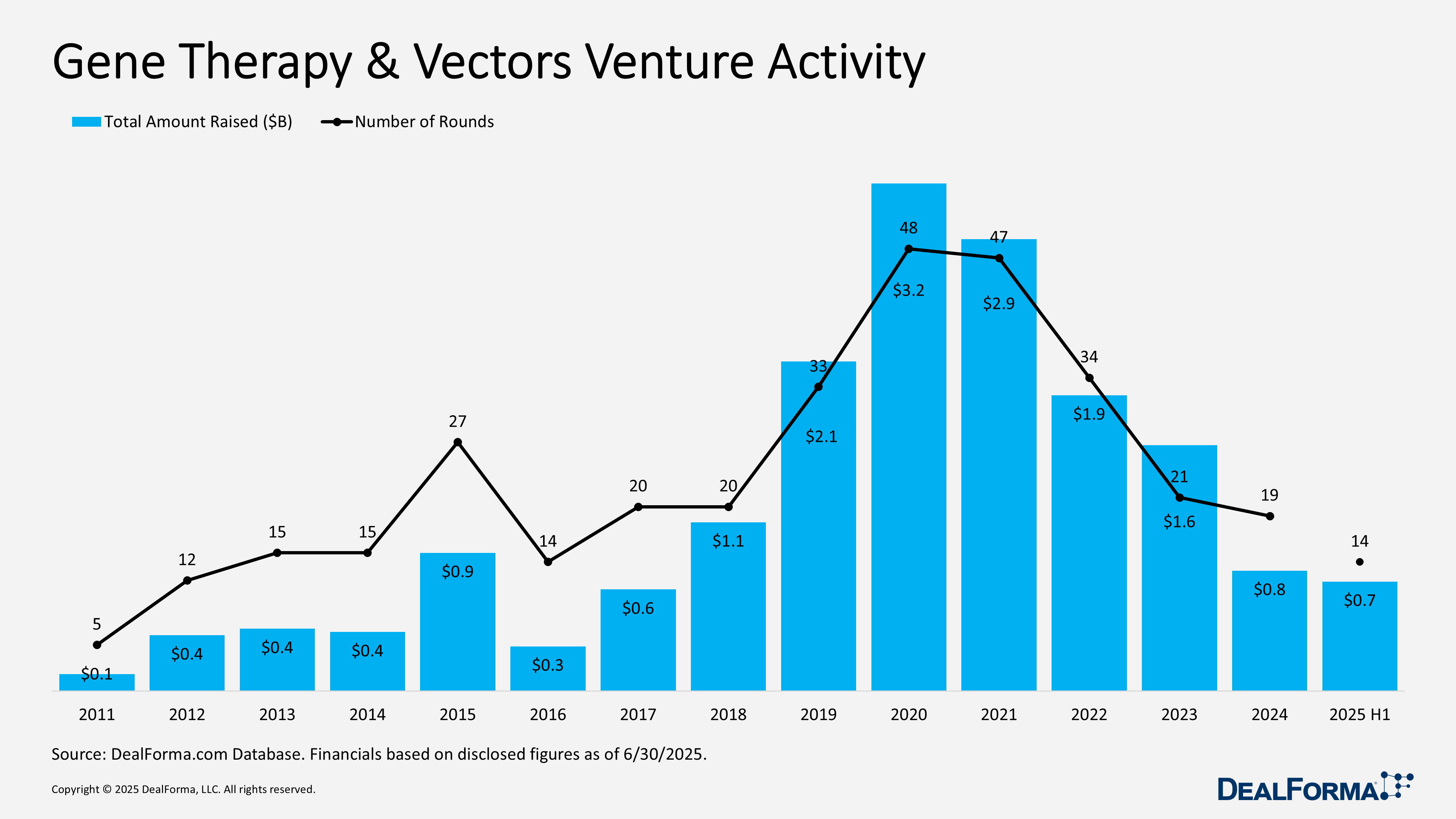
Gene Therapy & Vectors Venture Activity
In H1 2025, venture funding for gene therapy and vectors remained selective but substantial. 14 rounds raised $700 million, with an average of $53 million per round, signaling larger, later-stage, or more capital-intensive financings. By contrast, 2024 saw broader activity with 19 rounds totaling $800 million, averaging $45 million per round. Combined, 2024 through H1 2025 delivered 33 financings and approximately $1.5 billion raised.
Top Gene Therapy Venture Funding in H1 2025
Atsena Therapeutics – Series C – $150M – April 2025
Atsena Therapeutics raised $150 million in an oversubscribed series C led by Bain Capital Life Sciences, with Wellington Management joining as a new investor and strong participation from existing backers. Funding will advance the lead program ATSN-201 for X-linked retinoschisis (XLRS), a childhood-onset condition leading to blindness, while also supporting preclinical pipeline development and broader application of its proprietary spreading AAV.SPR capsid.
SpliceBio – Series B – $135M – June 2025
SpliceBio raised $135 million in a series B co-led by EQT Life Sciences and Sanofi Ventures, with participation from Roche Venture Fund, NEA, UCB Ventures, Novartis Venture Fund, and others. Funds will advance SB-007, an AAV-based gene therapy in Phase 1/2 trials for Stargardt disease, a genetic cause of vision loss, and support the expansion of SpliceBio’s pipeline in ophthalmology, neurology, and additional indications.
RhyGaze – Series A – $86M – January 2025
RhyGaze AG, a spin-out from the Institute of Molecular and Clinical Ophthalmology Basel, raised $86M in a series A led by GV (Google Ventures), Arch Venture Partners, and F-Prime Capital, with support from BioGeneration Ventures and Novartis Venture Fund. The company previously secured a CHF 10M seed round in July 2024. Funds will advance its cone optogenetics platform, designed to restore light sensitivity in retinal cone cells, through preclinical studies, an observational trial to define clinical endpoints, and a first-in-human trial.
Epic Bio – Series B – $68M – March 2025
Epicrispr Biotechnologies raised $68 million in the first close of its series B, led by Ally Bridge Group with participation from SOLVE FSHD and other investors. Proceeds will advance EPI-321, a first-in-class epigenetic therapy for facioscapulohumeral muscular dystrophy (FSHD), into clinical trials. Epicrispr recently received CTA approval in New Zealand to launch the first-in-human study, marking the first epigenetic therapy to enter the clinic for a neuromuscular disease.
Also check out Gene Therapy R&D Partnerships, M&A and Ventures in 2024