During Q3 2025, the Endocrine/Metabolic sector remained active across R&D, M&A, and venture funding. R&D partnerships reached 11 deals but declined in total value, led by Superluminal and Eli Lilly ($1.3 billion), Replicate and Novo Nordisk ($550 million), and Visirna and Sanofi ($395 million).
M&A surged to six deals totaling $10.3 billion, including Pfizer and Metsera ($7.6 billion), CB Biotechnology and Theratechnologies ($254 million), and OSR Holdings and Woori IO ($11 million), reflecting consolidation in obesity, cardiometabolic, and diabetes-focused assets.
Venture funding slowed to 11 rounds raising $445 million, highlighted by Crystalys ($205 million), ViCentra ($85 million), and Twin Health ($53 million). Overall, the sector remains active, with M&A and R&D driving major investments and venture funding supporting high-potential, technology-driven cardiometabolic and metabolic solutions.
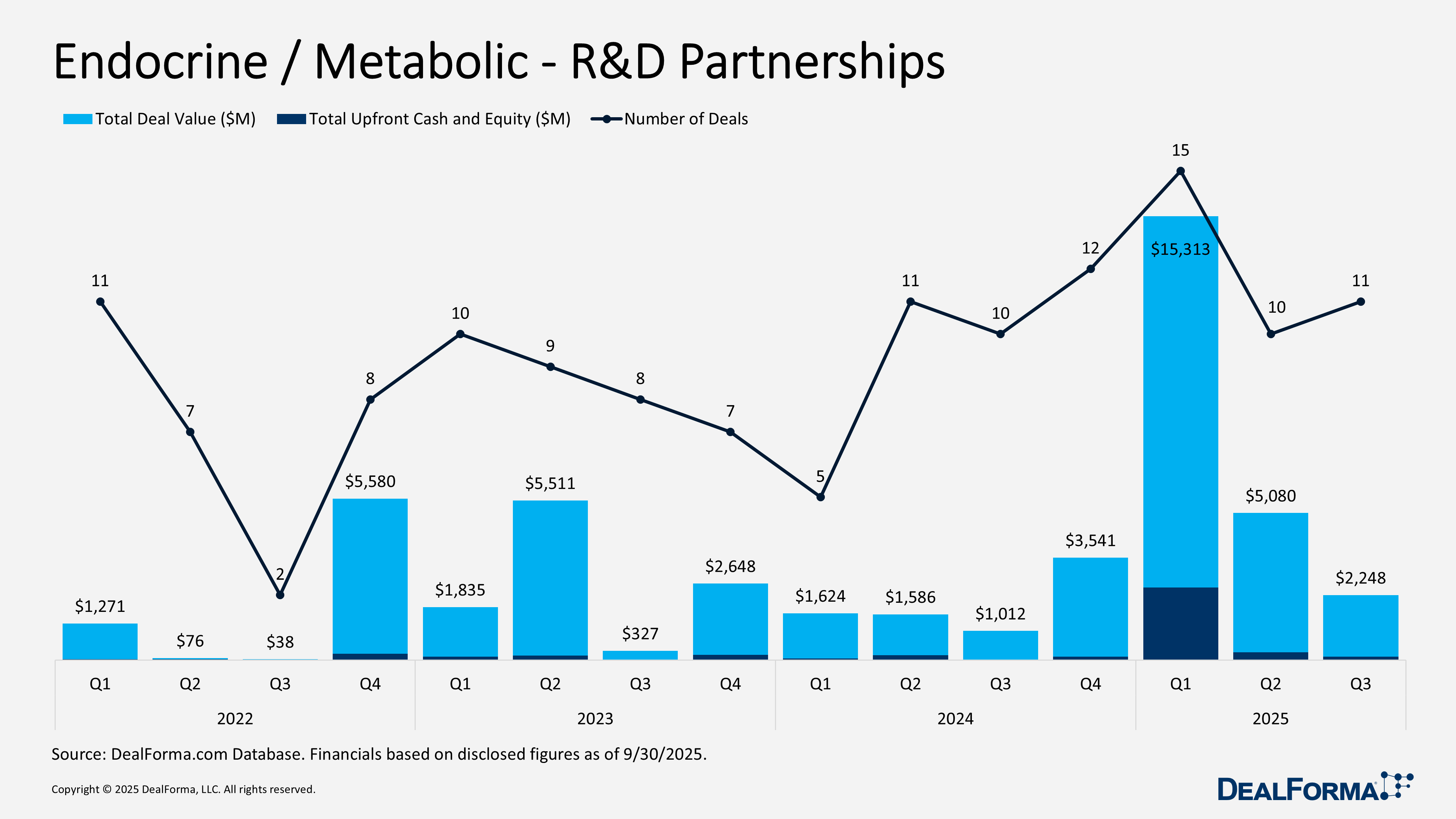
Endocrine / Metabolic – R&D Partnerships
In Q3 2025, endocrine and metabolic R&D partnerships comprised 11 deals with $2.2 billion in total announced value and $130 million in upfront cash and equity. This equates to an average deal size of approximately $204 million and upfront payments of about $12 million per deal, representing 5.8% of total value. Compared with Q2 2025, activity increased modestly in volume but declined sharply in scale. Q2 recorded 10 deals totaling $5 billion with $275 million upfront, or roughly $508 million per deal and $28 million upfront per transaction, equal to 5.4% of headline value. Quarter over quarter, deal count rose 10%, while total value and upfront payments fell by 56% and 53%, respectively. Across Q2 and Q3 2025 combined, the sector generated 21 R&D partnerships with an aggregate value of $7.3 billion and $405 million in upfront consideration.
Top Endocrine / Metabolic R&D Partnerships in Q3 2025
Superluminal Medicines development and commercialization deal with Eli Lilly – August 2025
Announced in August 2025, this platform stage collaboration grants Eli Lilly exclusive rights to develop and commercialize small molecule therapies against undisclosed GPCR targets in cardiometabolic disease and obesity. The partnership combines Superluminal Medicines’ AI and machine learning based structure driven discovery platform with Lilly’s global development and commercialization capabilities. Under the agreement, Superluminal is eligible to receive up to $1.3 billion in total consideration, spanning upfront, near-term, equity, development, and commercial milestones, along with tiered royalties.
Replicate development and commercialization deal with Novo Nordisk – August 2025
Announced August 2025, this platform stage partnership grants Novo Nordisk exclusive worldwide rights to develop and commercialize self-replicating RNA-based therapies using Replicate’s srRNA technology for cardiometabolic indications, including type 2 diabetes and obesity. The technology enables sustained and adjustable protein expression in vivo. Under the agreement, Replicate will receive undisclosed research funding and is eligible for up to $550 million in upfront payments and development and commercial milestones, in addition to tiered royalties.
Visirna development and commercialization deal with Sanofi – August 2025
Announced August 2025, this Phase III licensing agreement grants Sanofi exclusive rights in Greater China to develop and commercialize plozasiran, an RNA interference therapy from Arrowhead’s subsidiary Visirna. Plozasiran targets apolipoprotein C III and is intended for familial chylomicronemia syndrome and severe hypertriglyceridemia. The asset has completed Phase III development and holds breakthrough therapy and priority review designations, with a new drug application submitted to China’s regulator. Financially, Visirna will receive $130 million upfront and is eligible for up to $265 million in additional milestones, while Arrowhead will earn royalties on future sales.
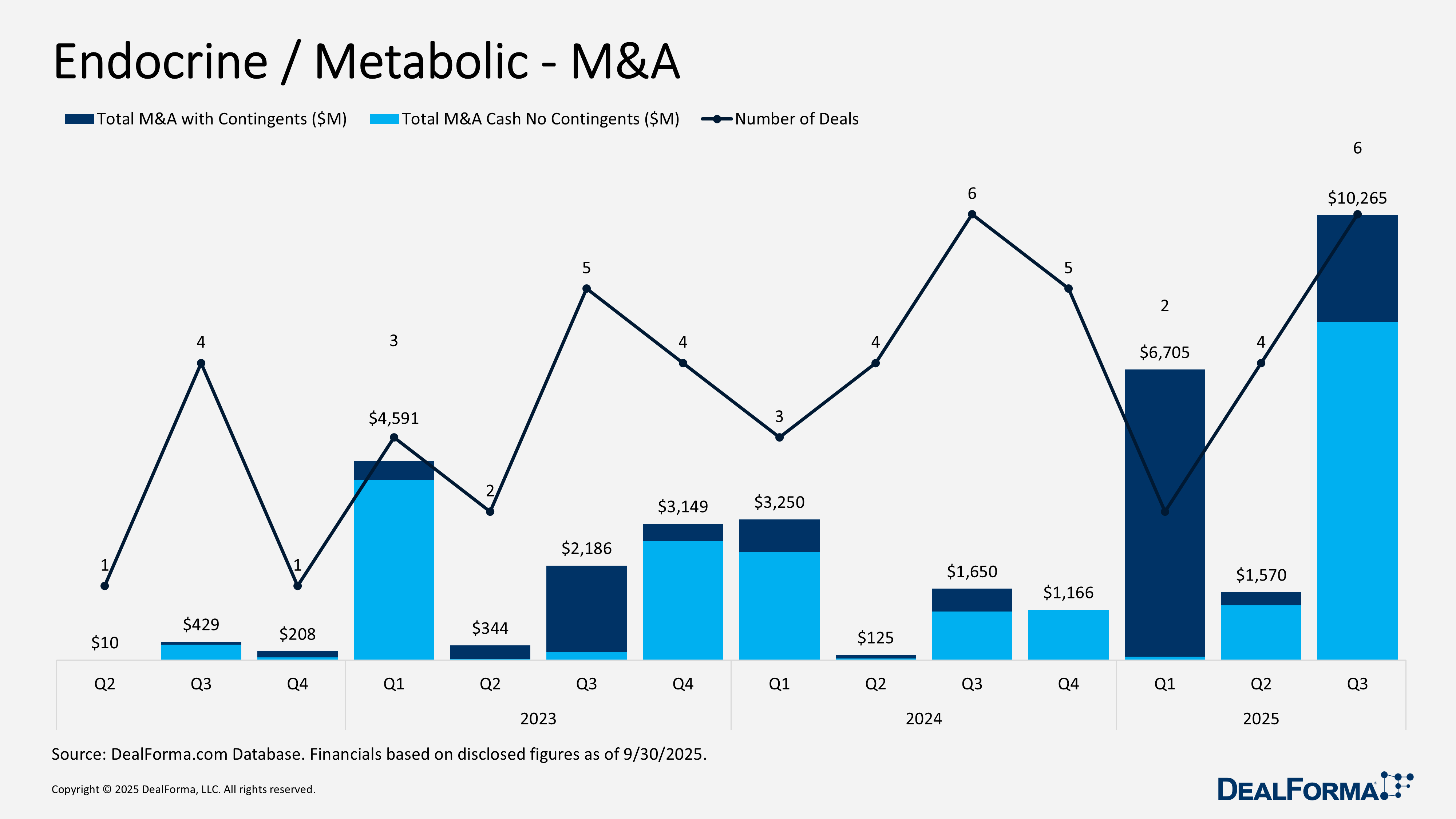
Endocrine / Metabolic – M&A
In Q3 2025, endocrine and metabolic M&A activity accelerated sharply, with 6 transactions totaling $10.3 billion in headline value. The cash consideration reached $7.8 billion, while approximately $2.5 billion, or 24% of the total value, was structured as contingent payments. The average deal size rose to roughly $1.7 billion. This marked a significant increase from Q2 2025, which recorded 4 deals worth $1.6 billion in headline value and $1.3 billion in cash, with $300 million, or 19%, in contingents and an average deal size of $393 million. Quarter over quarter, deal volume rose 50%, headline value increased 554%, and cash consideration climbed 514%. Across Q2 and Q3 2025 combined, the sector completed 10 M&A transactions totaling $11.8 billion in headline value and $9.1 billion in cash.
Top Endocrine / Metabolic M&A in Q3 2025
Pfizer acquiring Metsera – September 2025
Pfizer acquired Metsera, a Phase II-stage company focused on obesity and cardiometabolic disorders, with programs for injectable and oral GLP-1 and amylin. Lead assets include MET 097i, a weekly and monthly injectable GLP-1 receptor agonist in Phase II, MET 233i, a monthly amylin analog in Phase I, and multiple oral GLP-1 candidates approaching clinical entry, supported by next-generation combination therapies. Under the final agreement signed in November 2025, Pfizer paid $7.6 billion in cash, or $65.60 per share, with Metsera shareholders eligible for up to $2.4 billion in milestone-based contingent value rights. The acquisition closed on November 13, 2025, strengthening Pfizer’s position in obesity and cardiometabolic disease.
CB Biotechnology acquiring Theratechnologies – July 2025
CB Biotechnology acquired Theratechnologies, taking the company private. Key assets included approved Egrifta for lipodystrophy, Trogarzo for HIV, Phase I TH-1902 for solid tumors, preclinical TH-1904 for ovarian cancer, and SORT1+ technology. Shareholders received $3.01 per share plus contingent value rights (CVRs) worth up to $1.19 per share, tied to future gross profit milestones from Egrifta and Trogarzo, with total deal value up to $254 million. In-the-money options, SARs, and warrants received proportional cash and CVR allocations. The transaction, which included $12 million in termination fees if not completed under certain conditions, closed on September 25, 2025, and Theratechnologies is now privately held.
OSR Holdings acquiring Woori IO – July 2025
OSR Holdings agreed to acquire Woori IO, a medical device company specializing in noninvasive glucose monitoring using near-infrared spectroscopy. The technology offers pain-free, accurate diabetes management and potential integration into wearable devices. Woori shareholders will receive newly issued shares of OSR’s Korean affiliate (OSRK), convertible into OSR Holdings shares at a fixed rate if OSR’s stock reaches $10 within three years, representing approximately KRW 15 billion ($11 million). The acquisition, formalized on October 14, 2025, will make Woori IO a wholly owned subsidiary of OSRK and is expected to close within 6 months.
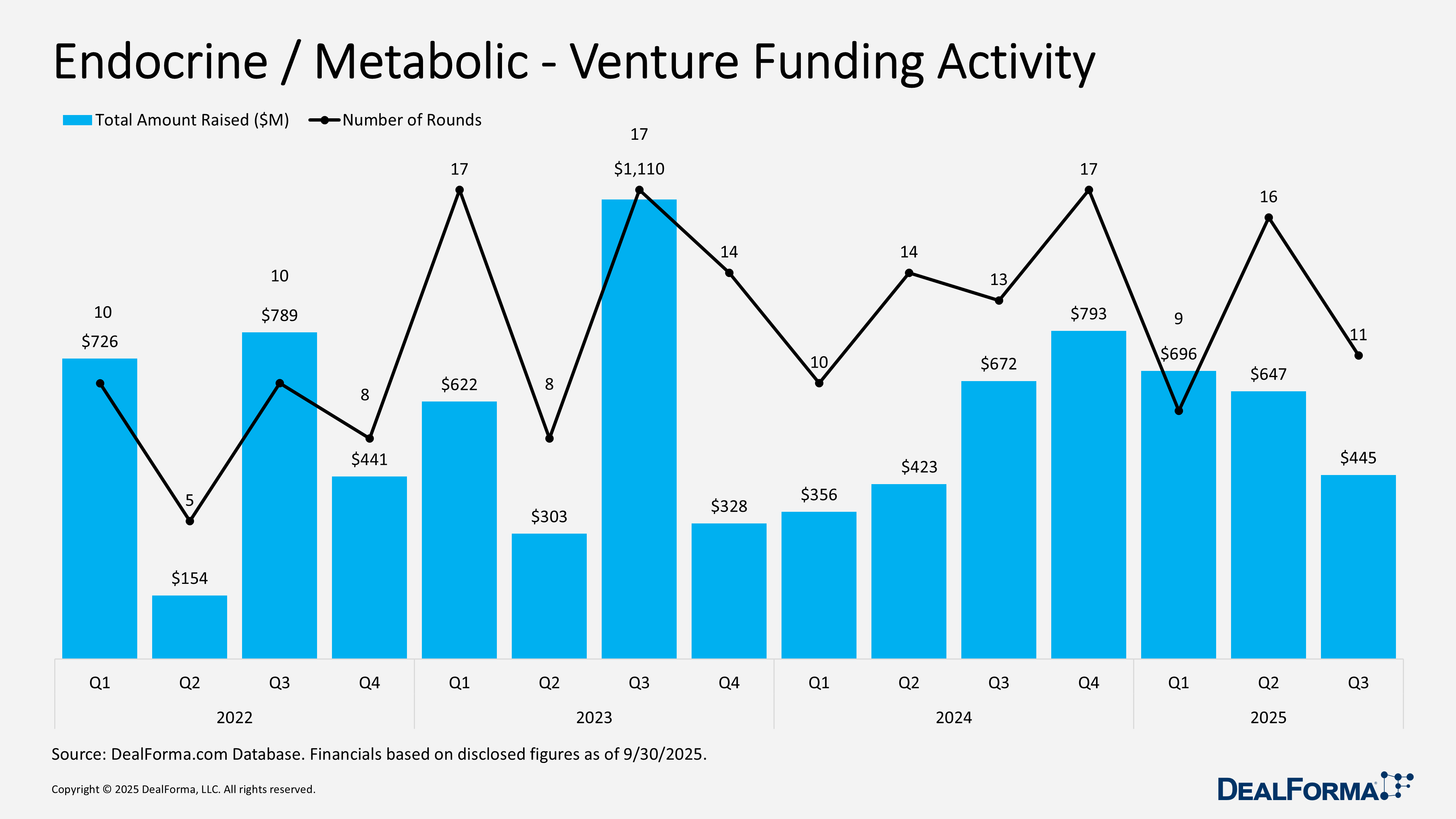
Endocrine / Metabolic – Venture Funding
In Q3 2025, endocrine and metabolic venture funding moderated to 11 rounds totaling $445 million, implying an average raise of about $40 million per round. This compares with Q2 2025, which saw 16 rounds raising $647 million, with a similar average. Quarter over quarter, both deal volume and total capital declined by 31%, while average round size remained stable. Across Q2 and Q3 2025 combined, the sector recorded 27 venture rounds raising a total of $1.1 billion.
Top Endocrine / Metabolic Venture Funding in Q3 2025
Crystalys Therapeutics – Series A – $205M – September 2025
Crystalys Therapeutics, a clinical-stage biopharmaceutical company, launched with a $205 million series A financing to advance therapies for patients with gout. The round was co-led by Novo Holdings, SR One, and Catalys Pacific, with participation from a broad syndicate including Perceptive Xontogeny, Lightstone Ventures, AN Venture Partners, abrdn-managed funds, KB Investments, Pontifax, Longwood Fund, Alexandria Venture Investments, Wedbush Healthcare Partners, and Prebys Ventures Fund. Proceeds will support global Phase 3 studies of the lead asset, dotinurad, a once-daily oral URAT1 inhibitor with potential best-in-class safety and efficacy. Dotinurad has shown robust clinical performance and is already approved in Japan, China, the Philippines, and Thailand.
ViCentra – Series D – $85M – September 2025
ViCentra, a European medical device company, closed an upsized $85 million series D financing led by Innovation Industries, with participation from Partners in Equity, Invest-NL, EQT Life Sciences, and Health Innovations. The funding will accelerate manufacturing, support the European launch of the next-generation Kaleido 2 insulin patch pump, and prepare for U.S. market entry. Kaleido 2 is a compact, lightweight, and precise insulin patch pump designed for lifestyle integration. It features customizable aluminum shells, intuitive use, and integration with Diabeloop’s automated insulin delivery algorithm and Dexcom CGM sensors. Early commercial uptake in Germany, France, and the Netherlands has exceeded expectations. The new capital positions ViCentra to scale production and drive rapid revenue growth while advancing next-generation diabetes care.
Twin Health – Series E – $53M – August 2025
Twin Health, an AI digital twin company for metabolic health, raised $53 million in a funding round led by Maj Invest of Denmark. The technology creates a real-time, personalized model of each individual’s metabolism using smart device data, lab results, and meal logs, providing tailored guidance on nutrition, activity, and sleep, supported by a clinical care team. The new funding will accelerate expansion with health plans and Fortune 500 clients across retail, healthcare, financial services, technology, and manufacturing.
Also check out Endocrine / Metabolic R&D Partnerships, M&A, Ventures and IPOs by TA – H1 2024 Review