Cardiovascular activity in Q3 2025 remained strong across R&D partnerships, M&A, and venture funding, with 6 R&D deals totaling $8.1 billion, 9 M&A transactions totaling $2.4 billion, and 18 venture rounds raising $755 million, reflecting confidence in late-stage therapies, cardiometabolic innovation, and advanced devices. Key R&D deals included Argo Biopharma’s $5.2 billion potential licensing of its Phase II ANGPTL3 program and additional dyslipidemia/siRNA assets to Novartis, Hengrui’s $1 billion Phase III myosin inhibitor collaboration with Braveheart Bio for obstructive hypertrophic cardiomyopathy, and Mabwell’s $1 billion dual-target siRNA license to Kalexo Bio for dyslipidemia and ASCVD risk reduction.
Notable M&A included Novartis acquiring Tourmaline Bio for $1.4 billion (pacibekitug, Phase III anti-IL-6 for cardiovascular and autoimmune disease), Edwards Lifesciences’ $497 million acquisition of Vectorious Medical to expand preventive heart failure diagnostics, and MannKind’s $360 million acquisition of scPharmaceuticals (Furoscix and SCP-111 autoinjector for heart failure/CKD). Venture highlights included Kardium’s $250 million for its Globe system in atrial fibrillation (78% 12-month effectiveness, no device-related safety events), Baraya Extended Care’s $124 million series B to expand post-acute care in Saudi Arabia, and Reprieve Cardiovascular’s $61 million series B supporting its heart failure fluid management system and FASTR II pivotal trial (400-patient FDA-enabling study).
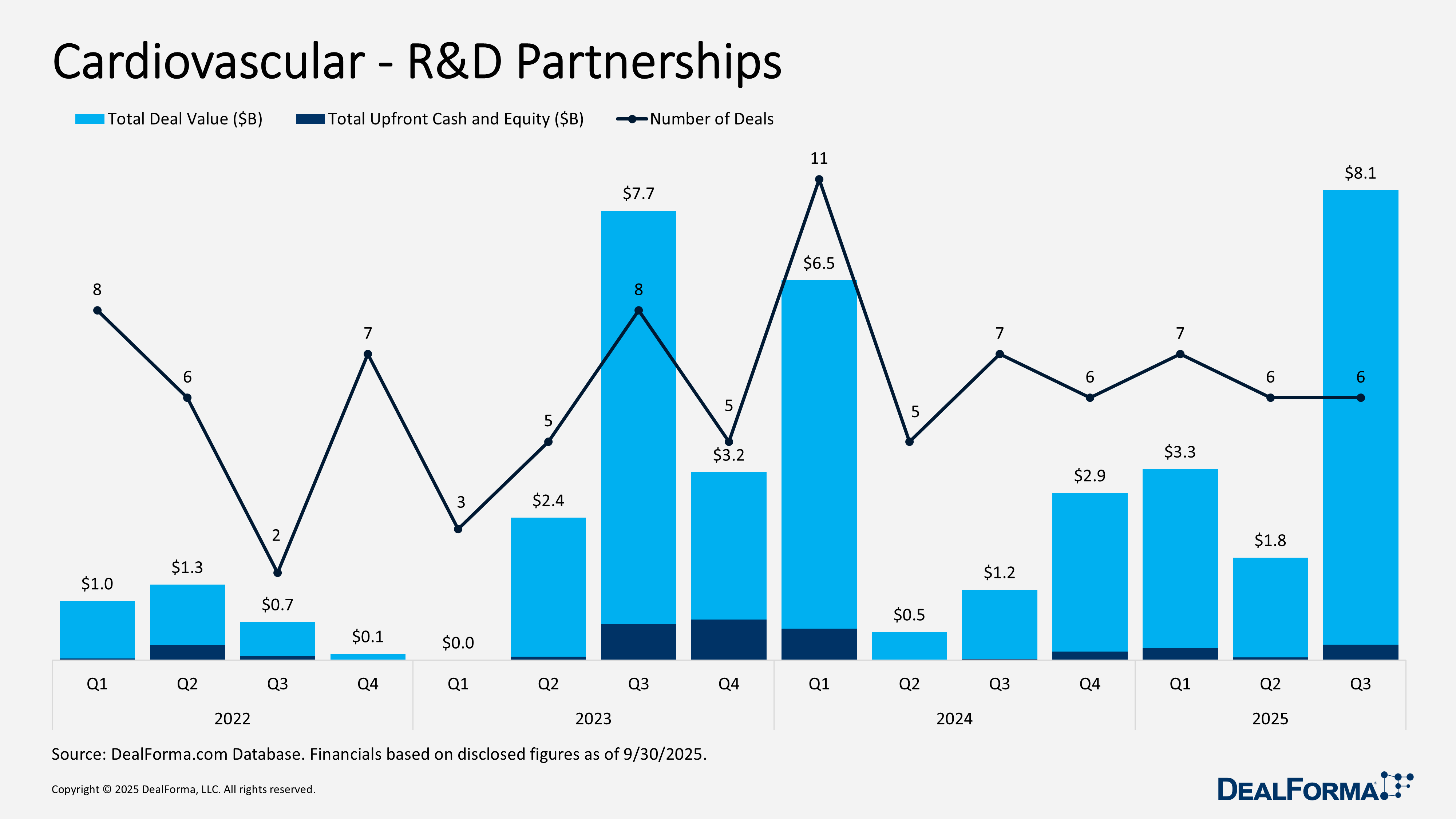
Cardiovascular – R&D Partnerships
Cardiovascular R&D partnership activity remained steady across Q2 and Q3 2025, but the financial profile shifted sharply. Q3 delivered 6 partnerships totaling $8.1 billion with $300 million in upfront cash and equity, indicating stronger investor and pharma commitment to late-stage and high-value cardiovascular innovation. In contrast, Q2’s 6 deals amounted to only $1.8 billion with $100 million upfront, reflecting more conservative terms earlier in the year. Across both quarters, the sector recorded 12 partnerships worth $9.8 billion and $300 million in upfronts.
Top Cardiovascular R&D Partnerships in Q3 2025
Argo Biopharma license option deal with Novartis – September 2025
Argo Biopharma licensed Novartis global rights outside China to its Phase Two ANGPTL3 program, along with two additional molecules for severe hypertriglyceridemia and mixed dyslipidemia, and one siRNA asset in IND-enabling studies. Argo retains the first negotiation right for BW 00112 and may elect profit and loss participation in several regions. Novartis receives an ex-China license and a profit and loss option in China, while Argo holds a profit and loss option in the United States. The deal includes $160 million upfront plus an equity investment, with total potential value reaching $5.2 billion through option fees, commercial milestones, and tiered royalties.
Hengrui Pharma development and commercialization deal with Braveheart – September 2025
Hengrui Pharma licensed Braveheart Bio exclusive global rights, excluding China, Hong Kong, Macau, and Taiwan, to develop, manufacture, and commercialize HRS 1893, a selective myosin inhibitor in Phase Three for obstructive hypertrophic cardiomyopathy. The agreement includes $65 million upfront, split evenly between cash and an equity investment in Braveheart Bio. Hengrui is also eligible for a $10 million near-term milestone tied to technology transfer and up to $1 billion in additional development and commercial milestones, along with undisclosed royalties.
Lipdro Therapeutics’ Dr. Justin Chun (Founder) is eligible for undisclosed royalties – September 2025
Mabwell Bioscience licensed Kalexo Bio exclusive global rights to develop, manufacture, and commercialize 2MW 7141, a dual-target siRNA therapy for dyslipidemia and for reducing the risk of atherosclerotic cardiovascular disease events. Mabwell will receive up to $1 billion in total value, including $12 million in upfront and near-term cash and up to $988 million in development and commercial milestones, plus tiered royalties. Mabwell will also receive an undisclosed equity stake in Kalexo Bio.
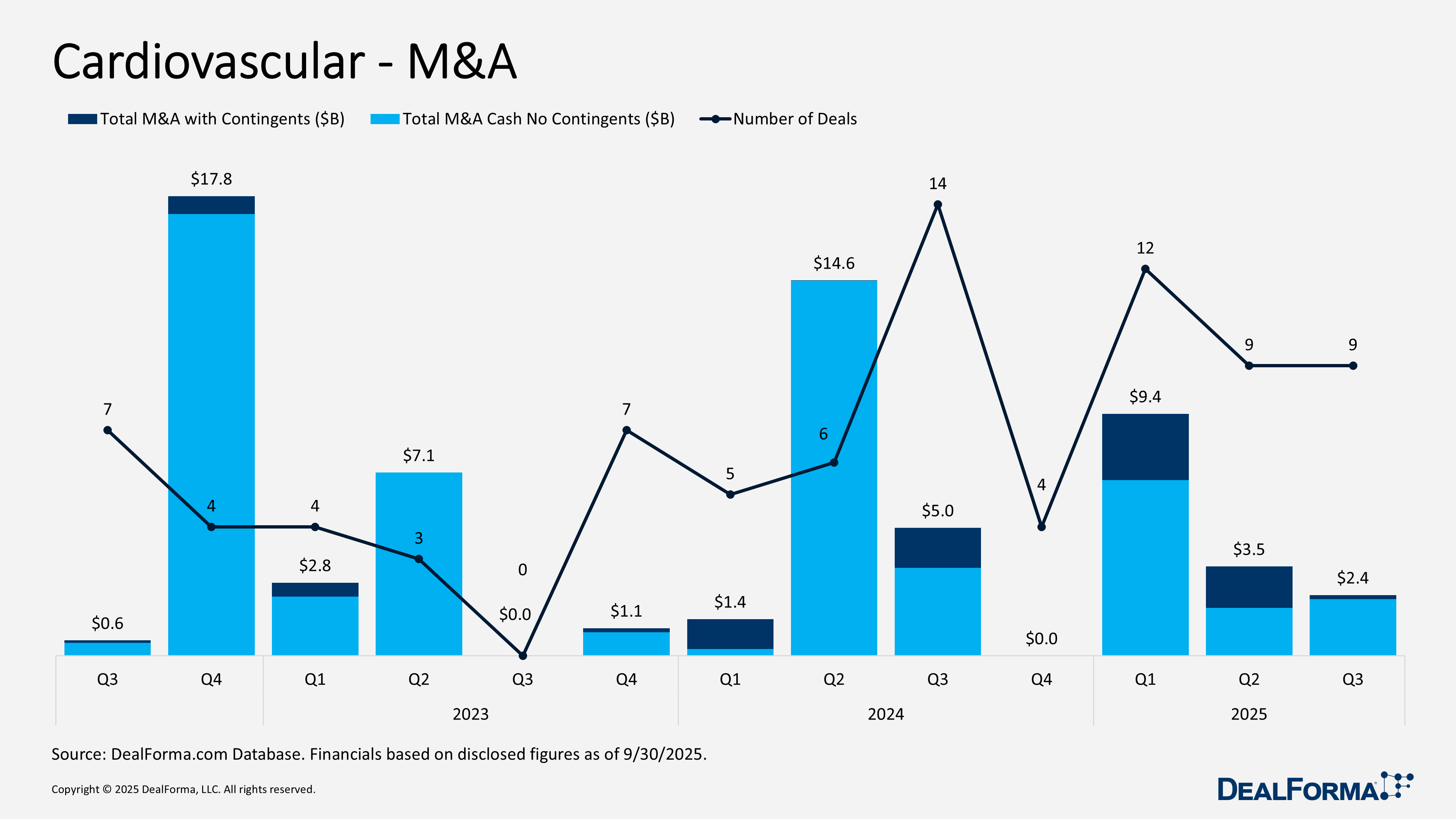
Cardiovascular – M&A
Cardiovascular M&A activity held steady in 2025, with 9 transactions in both the second and third quarters, although deal values shifted between periods. Q3 generated $2.4 billion in total value, including contingents, and $2.2 billion in upfront cash, reflecting a stronger quarter supported by more cash-focused transactions. Q2 produced the same number of deals but reached a higher $3.5 billion in total value with contingents, while upfront cash was lower at $1.9 billion, suggesting earlier transactions relied more on back-loaded structures. Across both quarters, buyers completed 18 deals worth $5.8 billion, including $4.1 billion in upfront cash.
Top Cardiovascular M&A in Q3 2025
Novartis acquiring Tourmaline Bio – September 2025
Novartis moved to acquire Tourmaline Bio, whose lead asset is pacibekitug, a long-acting, fully human anti IL 6 antibody that is Phase Three ready and designed to address cardiovascular and autoimmune diseases by targeting inflammation. Pacibekitug was originally licensed from Pfizer. The acquisition valued Tourmaline at $1.4 billion, with shareholders receiving $48 per share in cash at a 59% one-day premium. Tourmaline will operate independently until the transaction closes in the fourth quarter of 2025, after which it will become a wholly owned subsidiary of Novartis. Novartis completed the acquisition on October 28, 2025.
Edwards Lifesciences acquiring Vectorious Medical – September 2025
Edwards Lifesciences is acquiring Vectorious Medical, a private cardiovascular diagnostics company. The acquisition builds on Edwards’ growing presence in interventional cardiology and follows its earlier purchase of Endotronix’s pulmonary artery pressure sensor and digital platform. It also aligns with Edwards’ strategy to strengthen its role in preventive heart failure care and expand in the fast-growing remote heart failure monitoring market. Edwards previously held a 52% stake in Vectorious and will now take full ownership. The transaction totals $497 million.
MannKind acquiring scPharmaceuticals – August 2025
MannKind agreed to acquire scPharmaceuticals, whose lead product is Furoscix, an FDA-approved subcutaneous furosemide therapy for outpatient management of fluid overload in chronic heart failure and chronic kidney disease. The ReadyFlow Autoinjector, SCP 111, designed to reduce administration time from hours to under ten seconds, is expected to file a supplemental NDA in Q3 2025. The acquisition expands MannKind’s cardiometabolic portfolio and complements its focus on endocrine and pulmonary diseases. scPharmaceuticals shareholders received $5.35 per share in cash ($303 million) plus contingent value rights worth up to $1 per share, bringing the total potential deal value to $360 million. CVRs provide up to $0.75 for FDA approval of SCP 111 and $0.25 if 2026 sales reach $110–120 million. MannKind also assumed approximately $81 million of debt. The acquisition was completed on October 7, 2025.
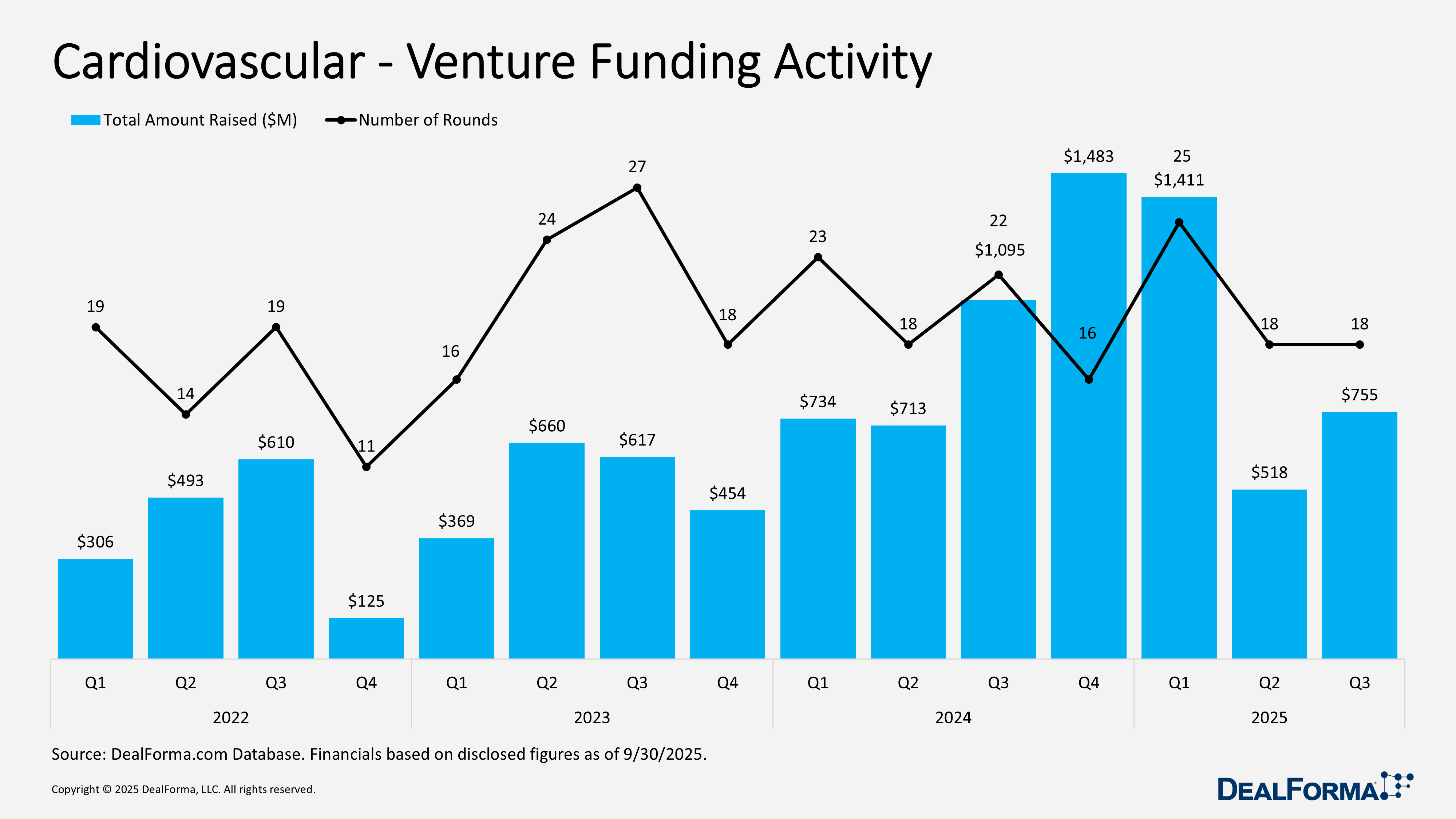
Cardiovascular – Venture Funding Activity
Cardiovascular venture funding strengthened in Q3 of 2025, with 18 rounds raising $755 million. Q2 recorded the same number of rounds but generated a lower $518 million, reflecting smaller average investment sizes earlier in the year. Across both quarters, the sector completed 36 financings totaling $1.3 billion.
Top Cardiovascular – Venture Funding Activity
Kardium – Series Unspecified – $250M – July 2025
Kardium, a Canada-based developer of pulsed field ablation therapy for atrial fibrillation, raised $250 million to support the launch of its Globe system, following a $104 million round in June 2024 that enabled completion of its pivotal trial. The funds will expand manufacturing and build clinical and commercial teams. Pulsed field ablation is gaining rapid adoption due to the lower risk of collateral injury versus traditional methods. One-year data from the Pulsar trial showed 78% effectiveness in patients with intermittent AFib and no device-related safety events.
Baraya Extended Care – Series B – $125M – July 2025
TVM Capital Healthcare closed a $124 million series B round for Baraya Extended Care, exceeding its target. The funds will expand Baraya’s inpatient and outpatient network in Saudi Arabia, addressing rising demand for post-acute care amid an aging population. The round was led by TVM Healthcare Afiyah Fund with co-investors including Olayan Financing Company, SEDCO, ANB Capital, and SVC. Since 2023, Baraya has opened two clinics in Riyadh and Jeddah with over 9,000 monthly sessions and plans to launch a 216-bed long-term care hospital by early 2026. The investment leverages TVM Capital Healthcare’s regional expertise in scaling post-acute care providers.
Reprieve Cardiovascular – Series B – $61M – August 2025
Reprieve Cardiovascular raised $61 million in a series B round, led by Deerfield Management with participation from Rex Health, Arboretum Ventures, Lightstone Ventures, Sante Ventures, Genesis Capital, Cadence Capital, and an undisclosed strategic investor. The funds will support its heart failure fluid management system and the pivotal FASTR II trial, which will enroll up to 400 patients in the U.S. and Europe to compare the device against standard diuretic therapy and support FDA premarket approval. The first patient has been enrolled at Washington University School of Medicine. FASTR II follows a successful 2024 FASTR pilot study that met primary efficacy and safety endpoints. Reprieve’s series A raised $42 million in 2024.
Also check out Cardiovascular R&D Partnerships, M&A, and Ventures – H1 2024 Review