H1 2025 marked a pivot from breadth to depth, fewer deals, but richer valuations and more strategic focus. Cancer remains the most active and competitive therapeutic area, with capital prioritizing late-stage partnerships, transformative acquisitions, and early-stage innovation in modalities such as RNA, cell therapies, and next-generation antibodies. H1 2025 dealmaking remained strong, with activity surpassing $35 billion across R&D partnerships, M&A, and venture financings. Cancer R&D partnerships drove the bulk of value in H1 2025, led by BioNTech-BMS ($11.1 billion, June), Pfizer-3SBio ($6.2 billion, May), and Philochem–RayzeBio ($350 million upfront, up to $1.4 billion, June), alongside Evopoint-Astellas ($130 million upfront, $1.1 billion milestones, May) and Abion (> $1 billion potential milestones, June).
M&A added momentum with Sanofi’s $9.5 billion acquisition of Blueprint (June), BioNTech’s $1.3 billion buyout of CureVac (March/June), and oncology-focused SPAC combinations including Veraxa-Voyager ($1.6 billion, April), Minovia-Launch One ($180 million + PIPE, June), and Vesicor-Black Hawk ($70 million + PIPE, April). Venture funding complemented these moves, with Antares ($177 million, June), CellCentric ($120 million, May), HanchorBio ($80 million, June), HAYA Therapeutics ($65 million, May), and Avidicure ($50 million, April) supporting novel platforms in immuno-oncology, radiopharmaceuticals, targeted protein degradation, and RNA therapies.
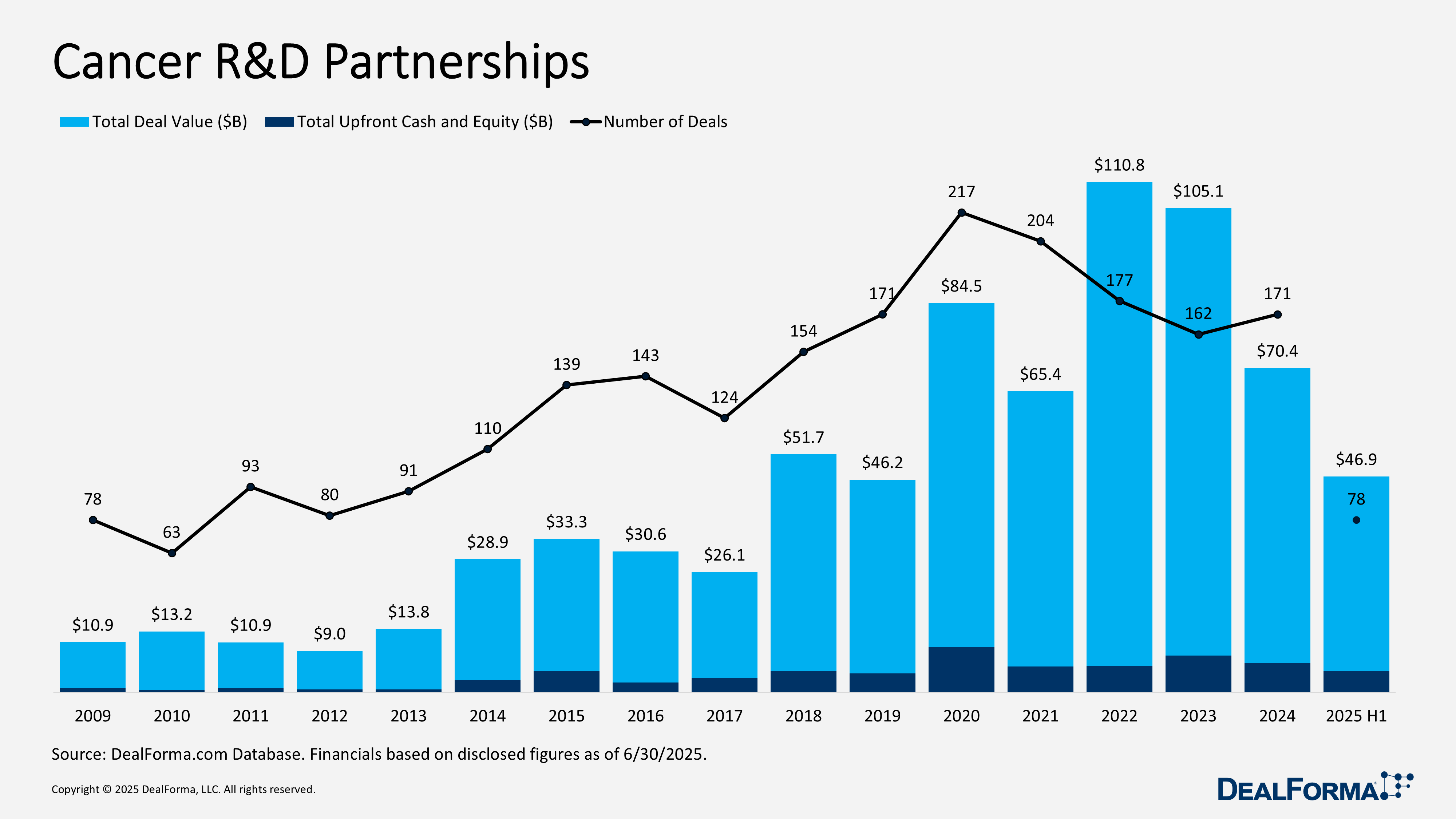
Cancer R&D Partnerships
During H1 2025, cancer R&D partnerships generated 78 deals worth $46.9 billion, including $4.7 billion upfront. While deal volume declined versus 2024, average upfronts rose sharply to $195 million nearly double last year’s $103 million indicating richer individual transactions. Already, H1 totals represent 67% of 2024’s full-year deal value and 75% of its upfronts. For context, 2024 recorded 171 deals valued at $70.4 billion with $6.3 billion upfront, bringing the combined 18-month tally to 249 deals, $117.3 billion in value, and $11 billion upfront.
Top Cancer R&D Partnerships in H1 2025
BioNTech development and commercialization deal with BMS – June 2025
BMS secured exclusive global rights to BioNTech’s BNT-327, a bispecific antibody targeting PD-L1 and VEGF-A, currently in global Phase III trials for small cell and non-small cell lung cancer. A Phase III trial in triple-negative breast cancer is planned for late 2025, with broader development spanning 20+ studies and over 10 indications. BioNTech, which gained BNT-327 via its Biotheus acquisition, will supply drug material, while both companies will co-develop the program and share costs, profits, and losses equally. BioNTech retains market authorization and sales rights in the US, EU, UK, China, and Turkey; BMS holds rights elsewhere. Financial terms include $1.5 billion upfront, $2 billion in fixed anniversary payments through 2028, and up to $7.6 billion in milestones.
3SBio development and commercialization deal with Pfizer – May 2025
Pfizer obtained exclusive global rights (ex-China) to develop and commercialize SSGJ-707, a bispecific antibody targeting PD-1 and VEGF for NSCLC, metastatic colorectal cancer, and gynecological tumors. Pfizer also has the option to market the drug in China and will handle manufacturing in the US (North Carolina and Kansas facilities). 3SBio will initiate the first Phase III trial in China in 2025. Financial terms include $1.3 billion upfront, a $100 million equity investment, and eligibility for up to $4.8 billion in milestones, plus double-digit tiered royalties. Additionally, 3SBio will receive $150 million in option fees in July 2025.
Philochem development and commercialization deal with RayzeBio – June 2025
RayzeBio (BMS subsidiary) licensed exclusive global rights to Philochem’s OncoACP3, a Phase I radiopharmaceutical small molecule targeting ACP3 for prostate cancer diagnosis and therapy. Development plans include therapeutic applications with Actinium-225 alongside diagnostic use. Philochem will receive $350 million upfront, up to $1 billion in milestones, and mid-single to low double-digit royalties on both therapeutic and diagnostic sales. US antitrust clearance for the transaction was completed in August 2025.
Evopoint Biosciences development and commercialization deal with Astellas – May 2025
Astellas licensed exclusive global rights (ex-China, Hong Kong, Macau, Taiwan) to Evopoint’s XNW-27011, a CLDN18.2-targeting antibody-drug conjugate (ADC) in Phase I/II trials for gastric, gastroesophageal, and pancreatic cancers. The ADC uses a topoisomerase I inhibitor payload and linker technology, complementing Astellas’ existing CLDN18.2 programs such as VYLOY. Evopoint will receive $130 million upfront, up to $70 million in near-term milestones, and as much as $1.1 billion in development, regulatory, and commercialization milestones, in addition to royalties.
Abion development deal with an undisclosed company for ABN-501 – June 2025
Abion licensed exclusive global rights to ABN-501 and 4 additional preclinical antibodies targeting CLDN3 and other proteins for solid tumors, including NSCLC, breast, ovarian, and small cell lung cancers. Abion will complete preclinical work, while the partner holds first negotiation rights for future candidates against three additional proteins. Financials include $5 million upfront per antibody, up to $290 million in development milestones, and as much as $1 billion in sales milestones.
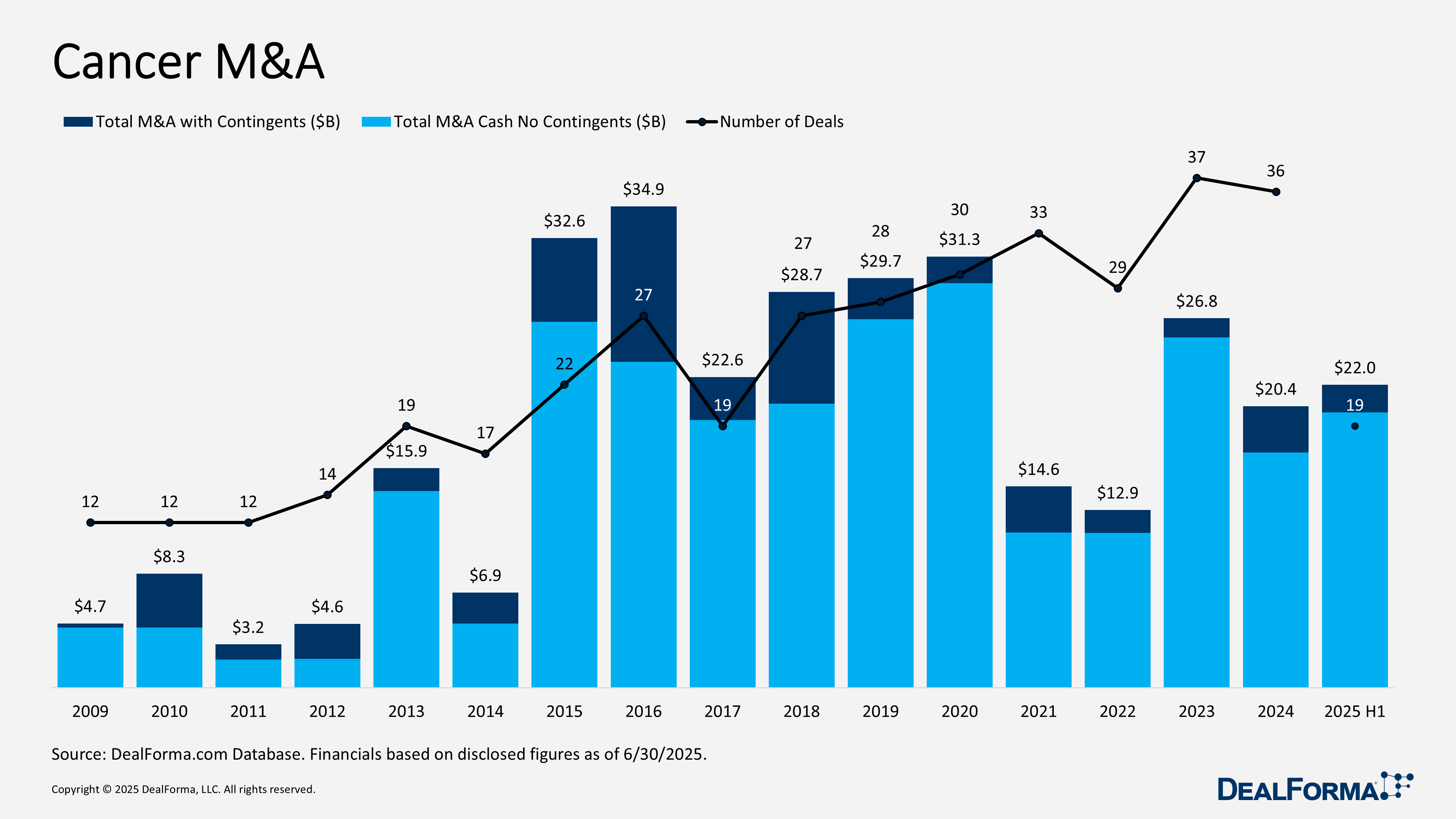
Cancer M&A
During H1 2025, cancer M&A delivered 19 deals totaling $22 billion (including contingents) and $20 billion in cash-only terms, already surpassing 2024’s $20.4 billion and $17.1billion, respectively. Average upfronts surged to $1.5 billion per deal, more than double 2024’s $743 million, underscoring a shift toward fewer but significantly larger, high-conviction acquisitions. For comparison, 2024 recorded 36 deals at $20.4 billion with contingents ($17.1 billion cash). Combined, the last 18 months saw 55 deals worth $42.4 billion ($37.1billion cash).
Top Cancer M&A in H1 2025
Sanofi acquiring Blueprint Medicines – June 2025
Sanofi acquired Blueprint Medicines for $9.5 billion, including $9.1 billion upfront and up to $400 million in milestones tied to the development and regulatory progress of BLU-808. Blueprint shareholders received $129 per share in cash, representing a 27 percent premium. Blueprint’s portfolio includes approved Ayvakit for gastrointestinal stromal tumor and systemic mastocytosis, late-stage elenestinib for mastocytosis, and mid-stage BLU-808 for chronic urticaria and allergic rhinoconjunctivitis, with additional proof-of-concept studies planned in asthma and mast cell activation syndrome.
Veraxa Biotech reverse merging with Voyager Acquisition Corp. in a SPAC transaction – April 2025
Veraxa Biotech will merge with Voyager Acquisition Corp. in a $1.6 billion SPAC transaction, creating a NASDAQ-listed company (VERX) focused on ADCs and bispecific T-cell therapies via its BiTAC platform. The pipeline includes Phase I VX-A901 for acute myeloid leukemia, multiple myeloma candidates, and nine discovery programs in solid tumors, breast cancer, and other targets. Veraxa shareholders will own 79.2 percent of the combined company, Voyager shareholders 19.3 percent, and PIPE investors 1.5 percent. The deal includes up to $253 million from Voyager’s trust funds, with closing expected in Q4 of 2025.
BioNTech acquiring CureVac in an all-stock transaction – June 2025
BioNTech will acquire CureVac in an all-stock transaction valued at $1.3 billion, giving CureVac shareholders approximately 5-6 percent ownership of BioNTech. The deal represents a 34 percent premium, with a collar mechanism adjusting the exchange ratio based on BioNTech’s share price. CureVac’s pipeline includes two partnered programs with GSK (COVID-19 and influenza), a Phase I multi-epitope glioblastoma vaccine, a Cas9 gene-editing program, and an IND-cleared neoantigen therapeutic for NSCLC. Preclinical assets include additional cancer vaccines and an mRNA prophylactic vaccine for urinary tract infections.
Minovia Therapeutics reverse merging with Launch One in a SPAC transaction – June 2025
Minovia Therapeutics will merge with Launch One Acquisition in a SPAC transaction to form Mito US One Ltd., listed on NASDAQ. The company will advance mitochondrial cell therapies for rare diseases and longevity, centered on Minovia’s Mitochondrial Augmentation Technology (MAT). Lead asset MNV-201, in Phase II for Pearson syndrome and myelodysplastic syndrome, holds FDA Fast Track and Rare Pediatric Disease designations, with Phase III planned. Minovia equity holders will receive shares valued at $180 million, along with $5 million in bridge financing, $18 million in PIPE investment, and access to Launch One’s trust funds (subject to redemptions). They are also eligible for up to $58 million in earnout shares tied to share price and development milestones.
Vesicor reverse merging with Black Hawk Acquisition Corp. in a SPAC transaction – April 2025
Vesicor will merge with Black Hawk Acquisition Corp. in a SPAC transaction to form Vesicor Therapeutics Inc., listed on NASDAQ. The company will advance ecm-RV/p53, a preclinical non-viral RNA nanoparticle delivering p53 mRNA for breast, pancreatic, prostate, lung, and colorectal cancers. The transaction values Vesicor at $70 million pre-money, with an additional $10 million in PIPE financing. Equity ownership will be determined by Black Hawk’s vested equity shares.
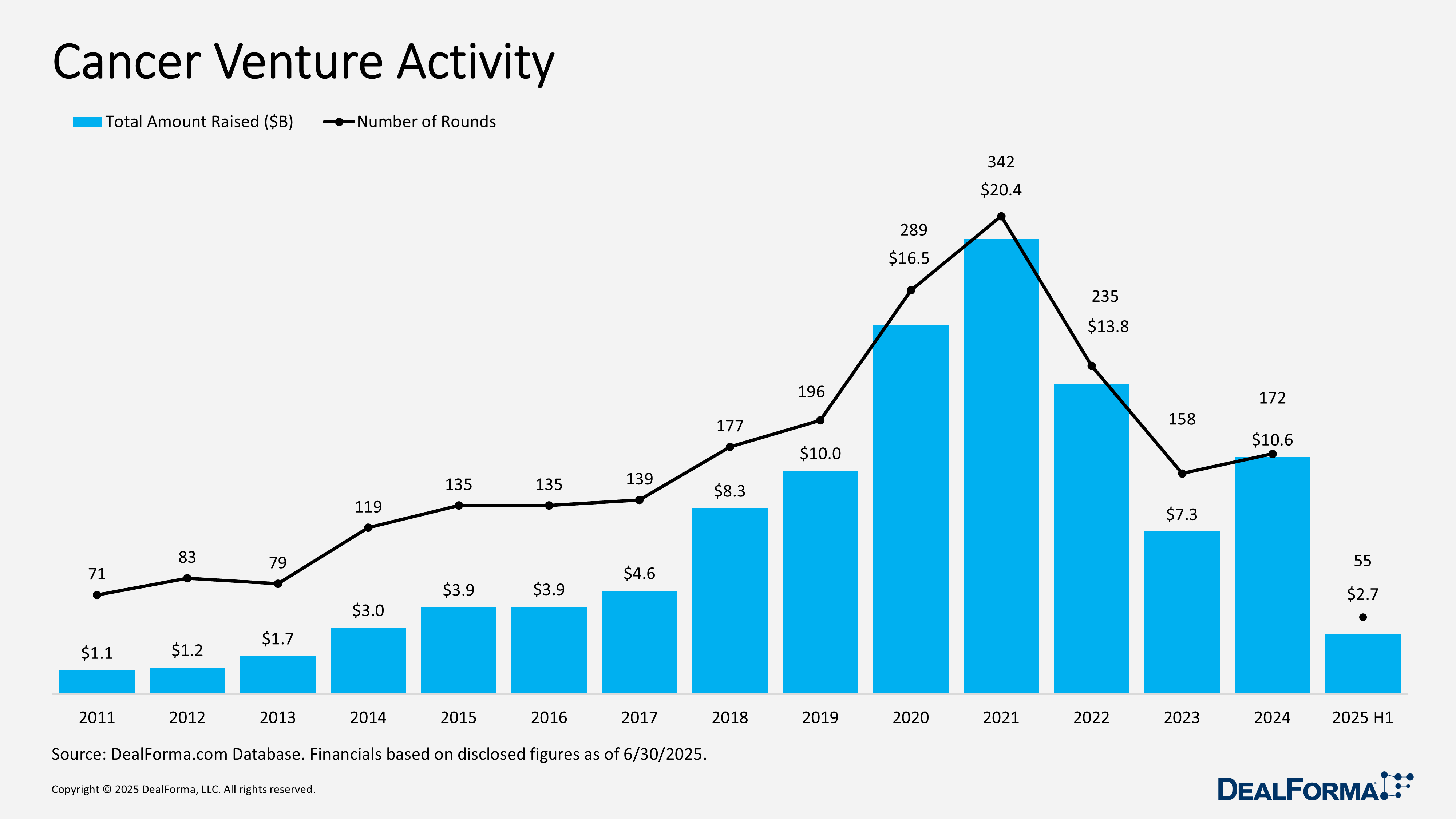
Cancer Venture Activity
During H1 2025, cancer venture funding totaled $2.7 billion across 55 rounds, averaging $55 million per raise. Activity slowed versus 2024, which saw 172 rounds worth $10.6 billion at a $66 million average. H1 funding represents roughly 25% of last year’s capital and 32% of its deal count, pointing to a cooler market with tighter check sizes. Over the past 18 months, cancer ventures raised $13.3 billion across 227 rounds.
Top Cancer Venture Activity in H1 2025
CellCentric – Series C – $120M – May 2025
CellCentric raised $120 million in a series C round co-led by RA Capital Management and Forbion, with participation from Avego Bioscience Capital and BrightEdge, the investment arm of the American Cancer Society. The financing supports advancement of its lead program, with plans to initiate a Phase II/III trial in heavily pretreated multiple myeloma and pursue potential accelerated approval. A global Phase III program is expected in mid-2026.
Antares Therapeutics – Series A – $117M – June 2025
Antares Therapeutics launched with $177 million in series A financing co-led by Omega Funds, Atlas Venture, Lightspeed Venture Partners, BVF Partners, and Cormorant Asset Management, alongside several other healthcare investors. A spin-out of Scorpion Therapeutics, Antares is led by Scorpion’s former management team and is advancing a pipeline of small molecule precision oncology and transcription factor programs, with its first candidate expected to enter the clinic in 2026. Scorpion, founded in 2020, previously raised $420 million, generated six development candidates (three in clinical testing), and sold its PI3Kα inhibitor STX-478 to Eli Lilly in March 2025 for up to $2.5 billion.
HanchorBio – Series Unspecified – $80M – June 2025
HanchorBio (TPEx: 7827.TWO) is listed on the Taipei Exchange Emerging Stock Board under the name Hanchor-KY, marking its public market debut. The company has raised more than $80 million in equity financing, backed by investors including Vivo Capital, Panacea Venture, and Affinity Capital. The listing strengthens momentum behind its lead asset, HCB101, a best-in-class CD47-targeting immunotherapy. HanchorBio is a global clinical-stage biotechnology company focusing on immuno-oncology.
HAYA Therapeutics – Series A – $65M – May 2025
HAYA Therapeutics raised $65 million in series A financing to advance its precision RNA-guided therapeutics platform. The funding will support clinical development of its lead candidate, HTX-001, for heart failure, and expand its long non-coding RNA pipeline. The round was co-led by Sofinnova Partners and Earlybird Venture Capital, with participation from Eli Lilly and Company, ATHOS, +ND Capital, Alexandria Venture Investments, LifeLink Ventures, and additional support from existing backers including Apollo Health Ventures, Longview Ventures, 4see ventures, BERNINA Bioinvest, and Schroders Capital.
Avidicure – Seed – $50M – April 2025
Avidicure launched with a $50 million seed round to advance its multifunctional antibody platform, “AVC-Boosters,” designed to deliver targeted, potent cancer monotherapies by activating both innate and adaptive immune responses. Its lead program, AVC-S-101, targets TROP2 in non-small cell lung cancer and other solid tumors. The financing was led by EQT Life Sciences, with participation from Kurma Partners, BioGeneration Ventures, BOM, Curie Capital, and V-Bio Ventures.
See the full details on cancer funding in 2025 and cancer partnership activity in DealForma database.
Also check out Cancer R&D Partnerships, M&A and Venture Funding by Tech – Q1 2025 Review