The first half of 2025 saw robust activity in the autoimmune space, with high-value R&D partnerships, strategic M&A deals, and strong venture funding, underscoring continued investor and industry interest despite shifts in deal structures and declining upfront payments. In R&D, Syneron Bio and AstraZeneca announced a March deal worth up to $3.4 billion, while Earendil Labs partnered with Sanofi in April on a bispecific antibody platform for IBD, with up to $1.7 billion in milestones. WuXi Biologics and Candid also signed a January agreement for tri-specific T-cell engagers, potentially worth $925 million.
On the M&A front, Alumis acquired Acelyrin in a $737 million all-stock transaction completed in May, expanding its pipeline in thyroid and neurologic autoimmune conditions. Paratek also completed its $330 million acquisition of Optinose, adding approved assets for CRS and migraine.
Venture funding remained active, with Abcuro raising $200 million in February to advance its Phase 2/3 IBM program, Timberlyne Therapeutics securing $180 million in January to develop a CD38-targeting antibody for autoimmune diseases, and GlycoEra closing a $130 million series B in May to progress its IgG4-targeted degrader and expand its immunology pipeline.
Autoimmune R&D Partnerships
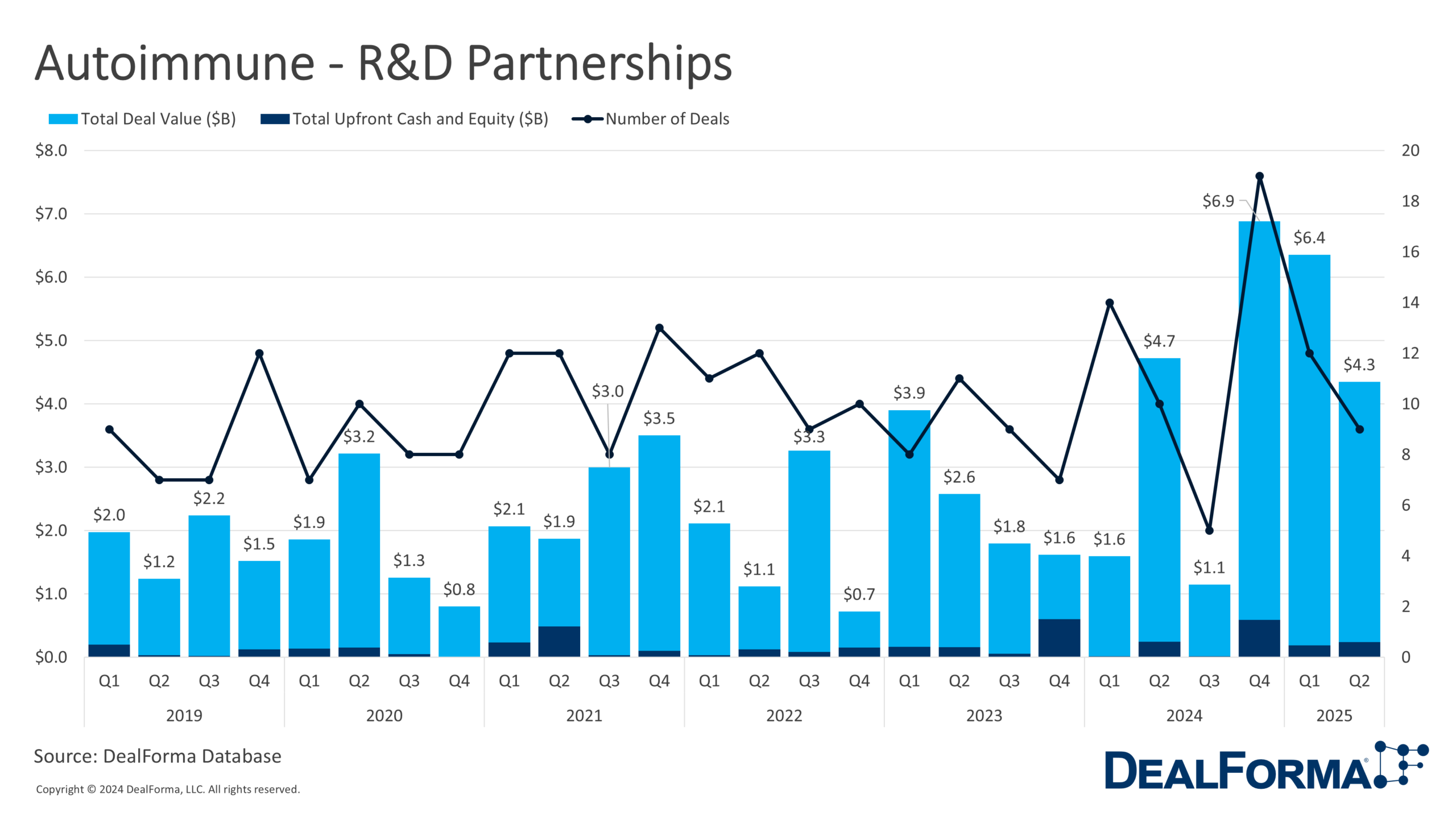
During H1 2025, 21 autoimmune partnerships were completed, slightly fewer than the 24 in H2 2024 but with a 34% increase in total disclosed value, rising to $10.7 billion from $8 billion. This boosted the average deal size from $330 million to $510 million. However, upfront payments declined to $400 million from $600 million, indicating a shift toward back-loaded deal structures with greater reliance on milestone payments.
Top Autoimmune R&D Partnerships in H1 2025
Syneron Bio development and commercialization deal with AstraZeneca – March 2025
AstraZeneca entered a discovery-stage deal with Syneron Bio to develop macrocyclic peptide therapies for autoimmune, rare, and metabolic diseases using Syneron’s Synova platform. Syneron will receive up to $75 million in upfront and near-term milestone payments, along with an undisclosed equity investment. The agreement includes up to $3.4 billion in potential development and commercial milestones, plus tiered royalties.
Earendil Labs development and commercialization deal with Sanofi – April 2025
Earendil Sanofi secured exclusive global rights to develop and commercialize Earendil Labs’ preclinical bispecific antibodies, HXN-1002 and HXN-1003, targeting Crohn’s disease and other inflammatory bowel diseases. The antibodies were discovered using Earendil’s AI-driven, high-throughput platform. The deal includes a $125 million upfront payment, up to $50 million in near-term milestones, and potential total milestones reaching $1.7 billion. Earendil will also receive tiered royalties in the high single to low double-digit range.
WuXi Biologics development and commercialization deal with Candid – January 2025
Candid acquired exclusive global rights from WuXi Biologics to develop and commercialize preclinical tri-specific T-cell engager antibodies targeting BCMA, CD20, and CD19 for autoimmune and inflammatory diseases. The candidates were developed using WuXi’s WuXiBody platform, with IND-enabling studies expected to start in H1 2026. WuXi Biologics is eligible for up to $925 million in total payments, including upfront, development, and commercial milestones, along with royalties.
Autoimmune M&A
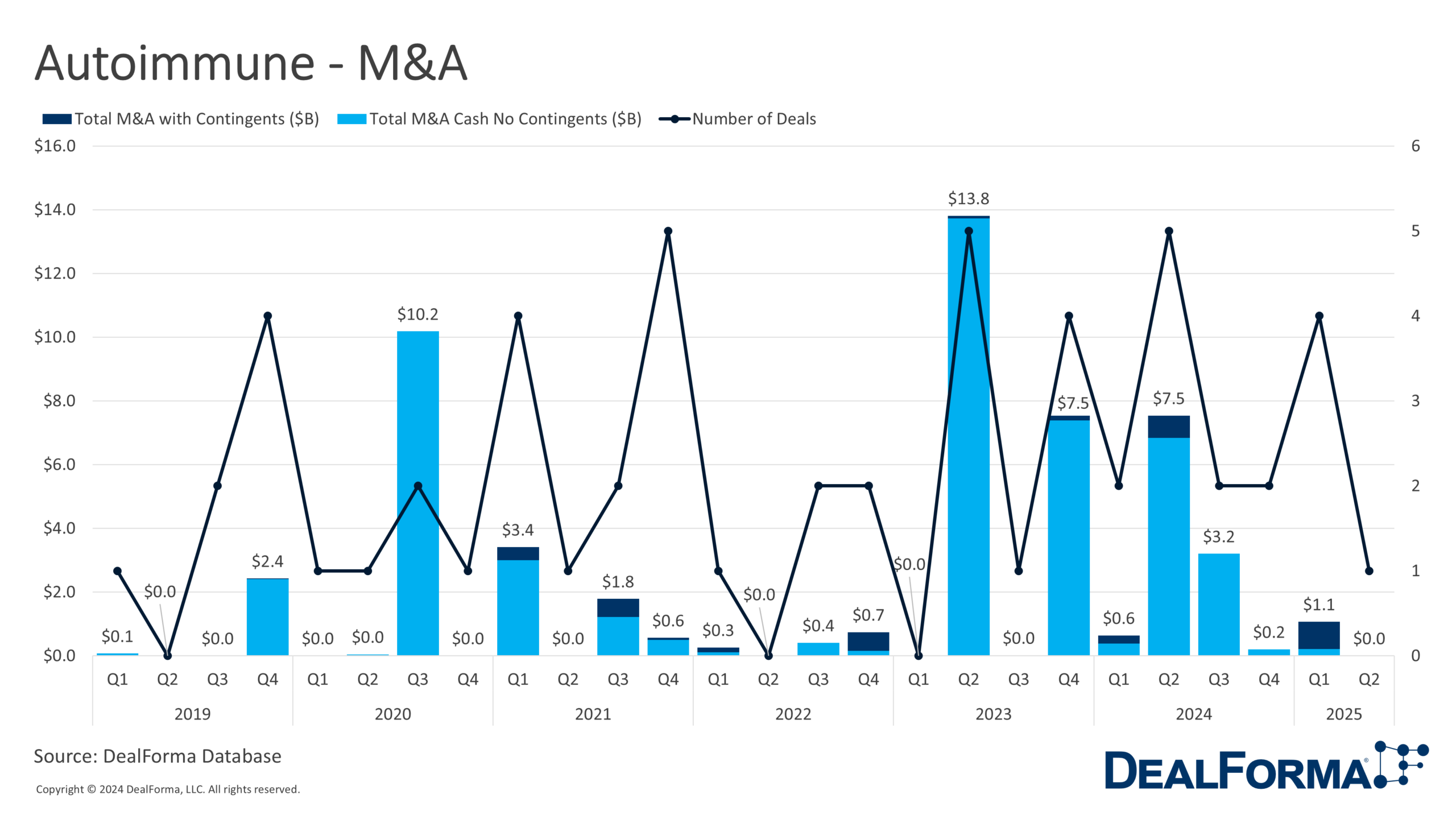
Autoimmune M&A activity rose slightly to 5 takeovers in H1 2025, but deal sizes declined sharply. The total deal value decreased by 68% to $1.1 billion, with upfront payments dropping to just $200 million, down from $3.4 billion and $850 million, respectively, across 4 deals in H2 2024. The average deal size contracted to $220 million, with only $40 million paid upfront.
Top Autoimmune M&A in H1 2025
Alumis acquiring Acelyrin – February 2025
Alumis acquired Acelyrin in an all-stock transaction, completed in May 2025, forming a clinical-stage biopharma company focused on immune-mediated diseases. The deal brings Acelyrin’s Phase II lead asset, lonigutamab, an IGF-1 receptor antagonist for thyroid eye disease, into Alumis’ pipeline, alongside ESK-001 and A-005. The final terms gave Acelyrin shareholders 0.4814 shares of Alumis stock per share held, resulting in a post-merger ownership split of 52% Alumis and 48% Acelyrin shareholders. The combined company, now operating under the Alumis name with Acelyrin as a wholly owned subsidiary, has a pro forma valuation of approximately $737 million. The acquisition significantly strengthens Alumis’ autoimmune and neurologic pipelines.
Paratek acquiring Optinose – March 2025
Paratek announced the acquisition of Optinose in a take-private deal valued at up to $330 million. The transaction included $9 per share in cash, a 50% premium, and up to $5 per share in contingent value rights (CVRs) tied to future sales of Xhance, Optinose’s approved drug-device therapy for chronic rhinosinusitis (CRS). CVRs include $1 per share if Xhance generates $150 million in annual sales before 2028, and an additional $4 per share if it reaches $225 million by 2029. Optinose also brings Onzetra Xsail, a nasal powder treatment for migraines. The acquisition, completed in May 2025, expands Paratek’s commercial portfolio in autoimmune, neurologic, and inflammatory diseases.
Autoimmune Venture Funding Activity
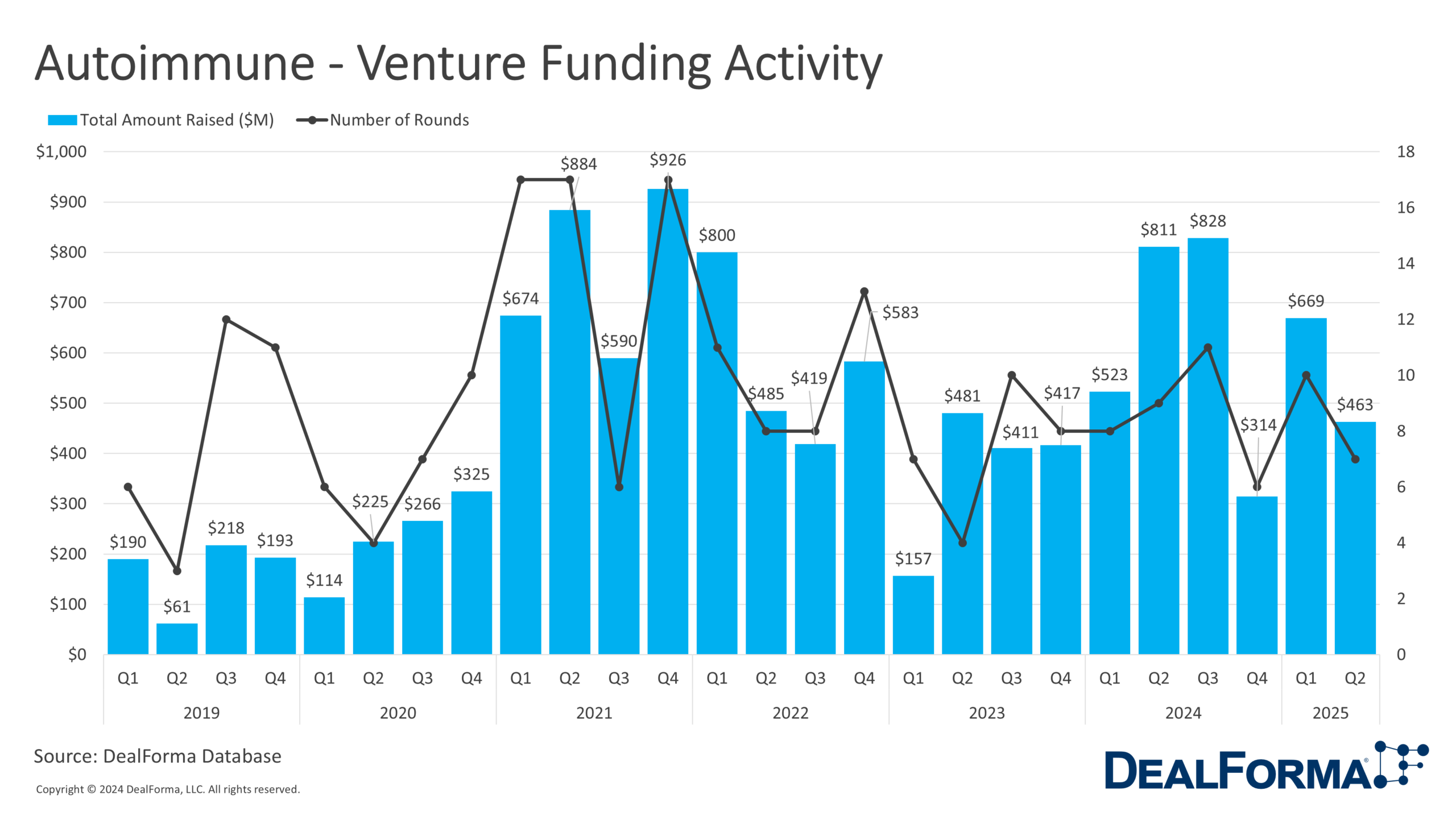
In Q2 2025, autoimmune-focused ventures secured $463 million across 7 financings, down from $669 million across 10 deals in Q1. Combined, H1 2025 totaled 17 financings worth $1.1 billion, matching the volume and value seen in H2 2024. Despite the dip in Q2, average deal size remained steady at approximately $66 million.
Top Autoimmune Venture Funding in H1 2025
Abcuro – Series C – $200M – February 2025
Abcuro, a clinical-stage biotech focused on autoimmune diseases and cancer, raised $200 million in a series C round led by New Enterprise Associates (NEA), with participation from Foresite Capital and existing investors including RA Capital, Bain Capital Life Sciences, Sanofi Ventures, and BlackRock. The funding will advance the registrational Phase 2/3 MUSCLE trial of ulviprubart (ABC008), a first-in-class monoclonal antibody targeting KLRG1, for the treatment of inclusion body myositis (IBM).
Timberlyne Therapeutics – Series A – $180M – January 2025
Timberlyne Therapeutics, a clinical-stage biopharma company developing treatments for autoimmune disorders, raised $180 million in a series A round led by Abingworth, Bain Capital Life Sciences, and Venrock Healthcare Capital Partners. Additional participants included Boyu Capital, Lilly Asia Ventures, Braidwell LP, and 3H Health Investment. The funding will advance CM313, a monoclonal antibody targeting CD38, for autoimmune diseases with high unmet need. Timberlyne holds global rights to CM313 outside greater China under a licensing agreement with Keymed Biosciences, which will receive upfront and milestone payments.
GlycoEra – Series B – $130M – May 2025
GlycoEra AG, a biotech company developing precision extracellular protein degraders for autoimmune diseases, closed an oversubscribed $130 million series B round. The financing was led by Novo Holdings, with participation from Catalio Capital Management, LifeArc Ventures, QIA, and existing backers including Sofinnova Partners, 5 AM Ventures, Roche Ventures, and Bristol Myers Squibb. Proceeds will advance GlycoEra’s lead IgG4-targeted degrader into clinical trials, initiate a second program, and further expand its immunology-focused pipeline.
Also check out Autoimmune R&D Partnerships, M&A, Ventures, and IPOs in 2024



