Neurology deal activity in 2025 was mixed, with weaker R&D partnerships offset by strong M&A growth, steady venture funding, and improved IPO activity, signaling confidence in later-stage and clinically validated assets. R&D partnerships declined in volume and value (56 deals; $25 billion; $900 million upfront) but held steady in median deal size, reflecting restrained capital deployment. M&A activity strengthened significantly (14 deals; $21.2 billion total value; $19.3 billion cash), driven by larger transactions. Venture funding grew modestly (70 rounds; $3.7 billion), with stable average round sizes ($57 million), indicating consistent early- and mid-stage investment. IPO activity improved (7 IPOs; $920 million), with more listings despite slightly smaller average deal sizes, signaling the reopening of public markets.
Key deals underscore this trend: Valo Health partnered with Merck KGaA (up to $3 billion); ABL Bio partnered with GSK (GBP 39 million upfront; up to GBP 2.1 billion); Arrowhead Pharmaceuticals partnered with Novartis ($200 million upfront; up to $2 billion); Johnson & Johnson acquired Intra-Cellular Therapies ($14.6 billion); Bain Capital acquired Mitsubishi Tanabe Pharma (JPY 510 billion; $3.3 billion); Alkermes acquired Avadel Pharmaceuticals ($2.2 billion revised); Oura raised $900 million (valuation $11 billion); Neuralink raised $650 million (valuation $9 billion); MapLight Therapeutics raised $373 million in series D; MapLight Therapeutics IPO ($296 million); LB Pharmaceuticals IPO ($285 million); and Saluda Medical IPO ($151 million), collectively underlining strong capital flows across partnerships, acquisitions, private funding, and public markets.
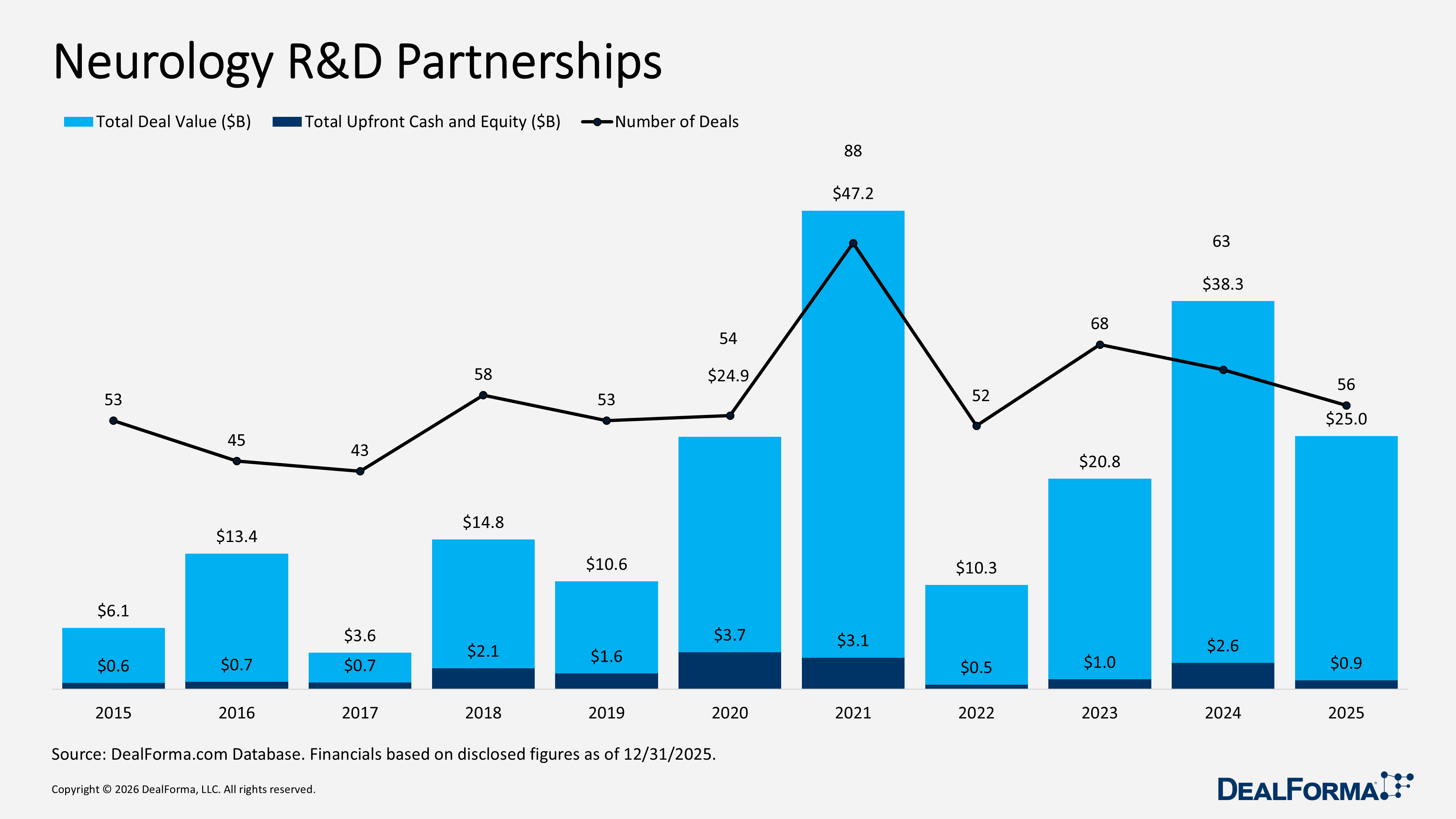
Neurology R&D Partnerships
Neurology R&D partnerships declined modestly in 2025, with 56 deals versus 63 in 2024. Total deal value fell to $25 billion from $38.3 billion, while upfront cash and equity dropped significantly to $900 million from $2.6 billion, indicating smaller transactions and cautious initial funding. Despite this, the median upfront payment rose to $43 million from $27 million. Across 2024 and 2025 combined, the sector recorded 119 deals totaling $63.2 billion, with $3.5 billion in upfront cash and equity,
Prominent Neurology R&D Partnerships in 2025
Valo Health development and commercialization deal with Merck – November 2025
Valo Health granted Merck KGaA exclusive rights to develop and commercialize therapies for Parkinson’s disease and related neurologic disorders. The collaboration leverages Valo’s AI-driven human causal biology platform, which analyzes over 17M de-identified patient records and biobank samples to identify targets, alongside its closed-loop chemistry platform to accelerate preclinical compound generation. Valo Health is eligible to receive up to $3 billion in upfront and milestone payments (breakout not disclosed), in addition to undisclosed R&D funding and royalties.
ABL Bio development and commercialization deal with GSK – April 2025
ABL Bio granted GSK exclusive worldwide rights to develop, manufacture, and commercialize antibody and nucleic acid-based therapies, including siRNA and ASOs, for neurologic diseases. The collaboration utilizes ABL Bio’s Grabody-B blood-brain barrier (BBB) shuttle platform and related expertise. ABL Bio will receive GBP 39 million (approx. $50 million) upfront and is eligible for up to GBP 2.1 billion (approx. $2.7 billion) in near-term, research, development, regulatory, and commercialization milestones (breakout not disclosed), along with tiered royalties.
Arrowhead development and commercialization deal with Novartis – September 2025
Arrowhead Pharmaceuticals granted Novartis exclusive worldwide rights to develop, manufacture, and commercialize ARO-SNCA, a preclinical siRNA therapy targeting alpha-synuclein for synucleinopathies, including Parkinson’s disease. The collaboration leverages Arrowhead’s Targeted RNAi Molecule (TRiM) platform, with an option for Novartis to expand into additional TRiM-based targets. Arrowhead will lead preclinical work through Clinical Trial Application (CTA) readiness, after which Novartis will assume full responsibility for clinical development, manufacturing, medical affairs, and commercialization. Arrowhead will receive $200 million upfront and is eligible for up to $2 billion in development, regulatory, and sales milestones (breakout not disclosed), plus tiered royalties.
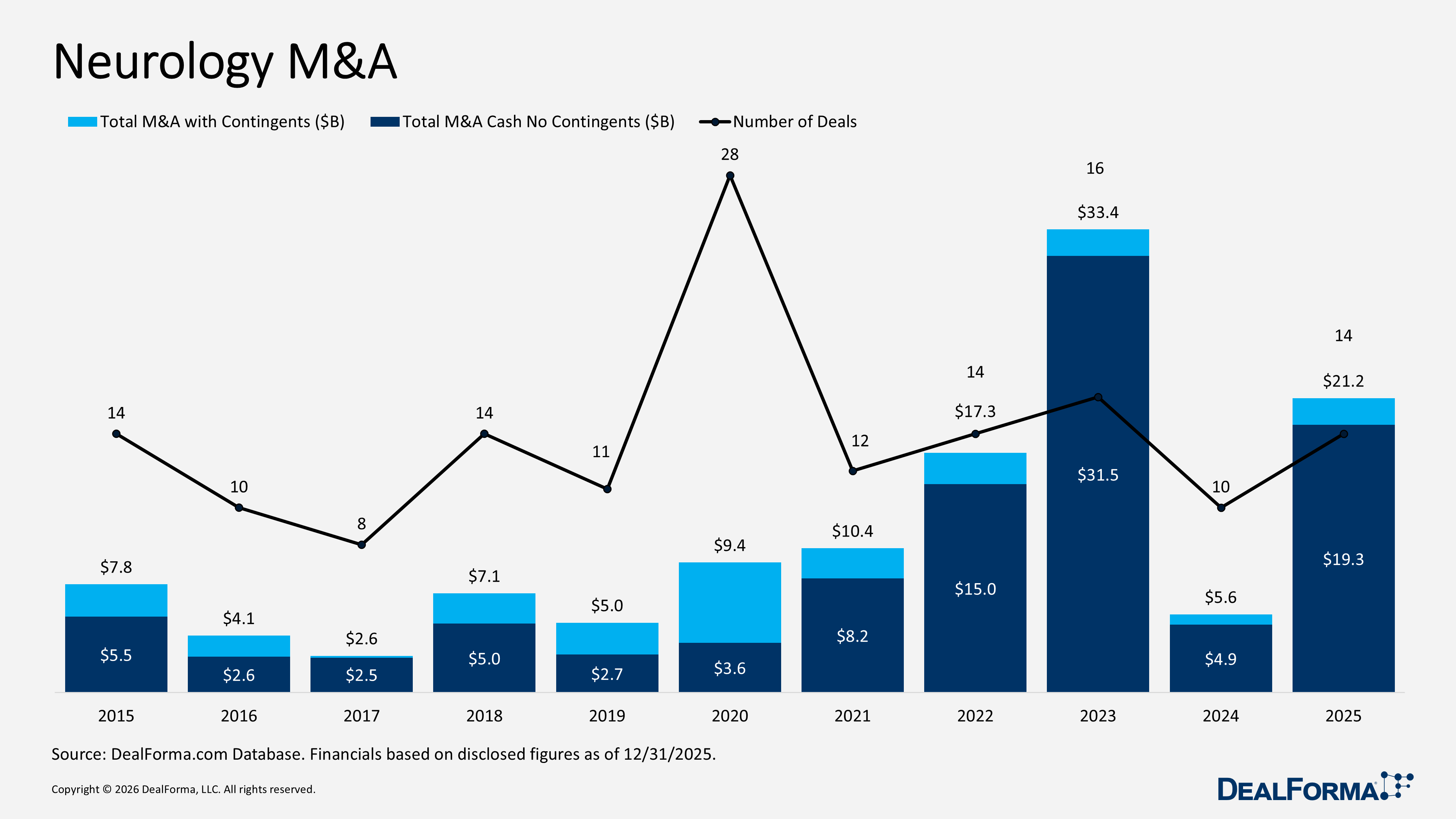
Neurology M&A
Neurology M&A activity strengthened in 2025, with deal volume increasing to 14 from 10 in 2024. Total deal value, including contingents, rose sharply to $21.2 billion from $5.6 billion. In comparison, cash value excluding contingents increased to $19.3 billion from $4.9 billion, reflecting both higher deal flow and significantly larger transaction sizes. Average upfront cash and equity climbed to $2.1 billion from $613 million, and the median upfront value increased to $470 million from $168 million. Across 2024 and 2025 combined, neurology M&A totaled 24 deals, with $26.9 billion in value, including contingents, and $24.2 billion in cash, excluding contingents.
Prominent Neurology M&A in 2025
Johnson & Johnson acquiring Intra-Cellular Therapies – January 2025
Johnson & Johnson acquired Intra-Cellular Therapies to expand its neuroscience portfolio. The acquisition includes Caplyta, an approved therapy for schizophrenia with additional indications across bipolar disorder, depression, sleep disorders, and autism, alongside a broader pipeline spanning Parkinson’s disease, heart failure, anxiety, pain, cancer, and other CNS disorders across various development stages. Intra-Cellular shareholders received $132 per share in cash, representing a 40% one-day premium and a total deal value of approx. $14.6 billion. The transaction was completed on April 2, 2025, with Intra-Cellular Therapies continuing to operate as a business unit within Johnson & Johnson.
Bain Capital acquiring Mitsubishi Tanabe Pharma – February 2025
Bain Capital acquired Mitsubishi Tanabe Pharma, including approved therapies such as Uplizna (NMOSD), Zynlonta (R/R diffuse large B-cell lymphoma), Canalia (diabetes), Orkedia (secondary hyperparathyroidism), Invokana (diabetic nephropathy), and Dysval (tardive dyskinesia), alongside a broad pipeline across schizophrenia, Parkinson’s disease, hypertension, spinal cord injury, myasthenia gravis, erythropoietic protoporphyria, systemic sclerosis, HTLV-1-associated myelopathy, ANCA-associated vasculitis, and other autoimmune indications. Mitsubishi Tanabe received JPY 510 billion (approx. $3.3 billion) upfront. The acquisition closed on July 1, 2025, with Mitsubishi Tanabe Pharma integrated under Bain Capital.
Alkermes acquiring Avadel Pharmaceuticals for $2.1B – October 2025
Alkermes acquired Avadel Pharmaceuticals for $2.1 billion, gaining Lumryz, a once-daily oxybate for narcolepsy. The deal strengthens Alkermes’ entry into the sleep medicine market and expands its pipeline in hypersomnolence disorders. Initially, Avadel shareholders were to receive approximately $1.9 billion in cash ($18.5 per share), representing a 12% one-day premium and a 38% premium to the three-month VWAP, plus up to $1.50 per share (approx. $160 million) in contingent value rights (CVR) tied to FDA approval. Following a superior unsolicited proposal from Lundbeck, Alkermes revised the agreement on November 19, 2025, increasing the cash consideration to $2.2 billion ($21 per share) and maintaining up to $1.50 per share (approx. $158 million) in CVRs. The acquisition was approved by Avadel shareholders in January 2026 and completed in February 2026, with closure in Q1 2026.
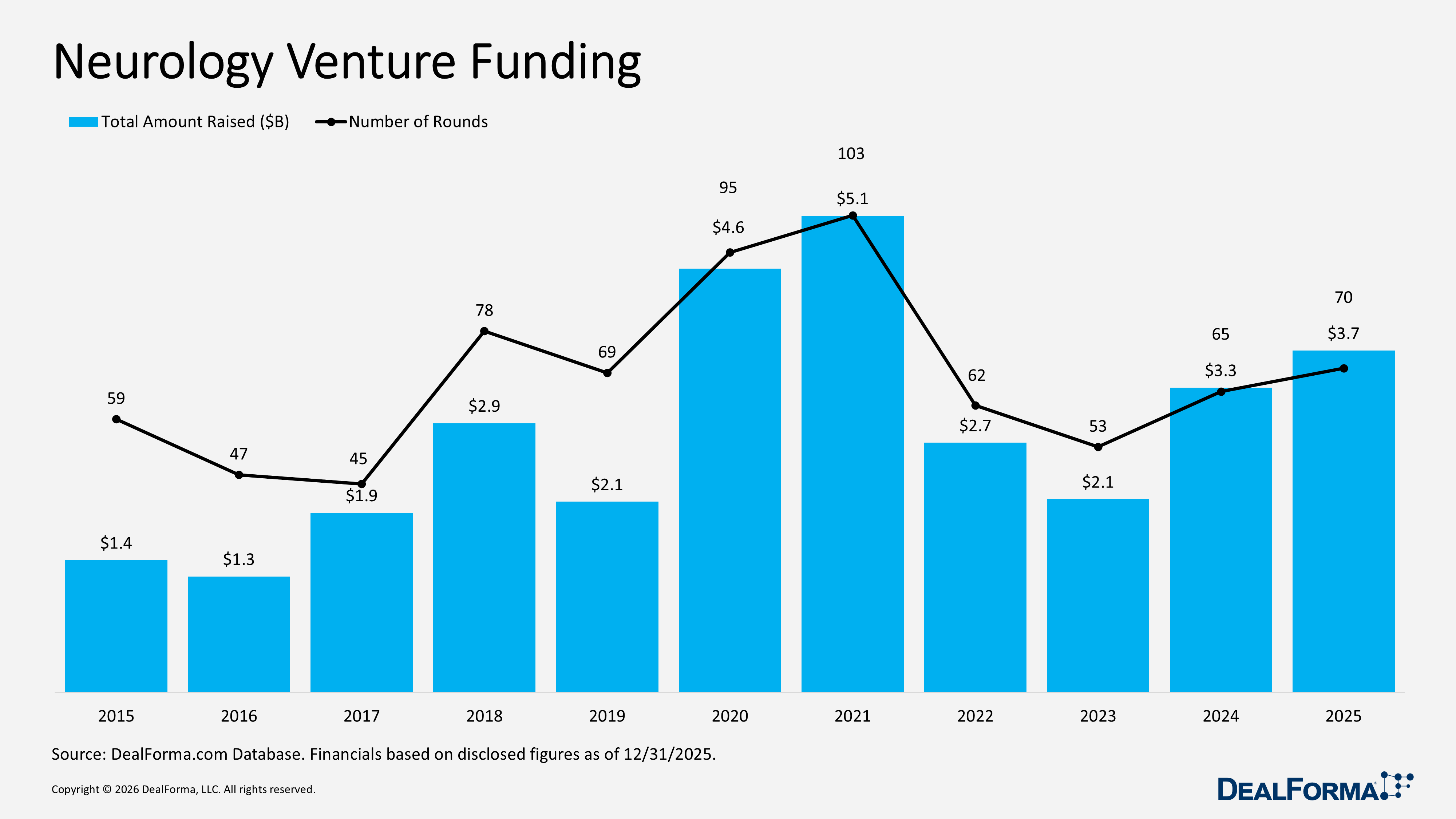
Neurology Venture Funding
Neurology venture funding showed modest growth in 2025, with rounds increasing to 70 from 65 and total capital rising to $3.7 billion from $3.3 billion. The average round size remained stable at $57 million. Across 2024 and 2025 combined, the sector raised $7 billion across 135 rounds,
Prominent Neurology Venture Funding in 2025
Oura – Series Unspecified – $900M – October 2025
Oura raised $900 million in a series E round, the largest neurology-adjacent digital health financing in 2025, increasing its valuation to approximately $11 billion from $5.2 billion in December 2024. Participation from crossover investors such as Fidelity Investments, ICONIQ Capital, and Atreides Management highlights strong IPO potential. The company’s multi-indication platform spans neurologic, pulmonary, and cardiovascular monitoring. Oura reported $500 million in 2024 revenue and projects $1 billion in 2025, with further upside linked to clinical validation of neurologic biomarkers and potential integration into the U.S. healthcare system.
Neuralink – Series E – $650M – June 2025
Neuralink raised $650 million in a series E round, one of the most prominent neurotechnology financings in 2025, backed by leading investors including Sequoia Capital, Founders Fund, and Lightspeed Venture Partners, alongside sovereign investors such as Qatar Investment Authority and G42. At the time of the raise, the company had enrolled five patients with severe paralysis and received FDA Breakthrough Device Designation in May 2025 for treating severe speech impairment. Total funding exceeded $1.2 billion, with a $9 billion pre-money valuation, more than double its 2023 level, reflecting strong clinical and regulatory momentum.
MapLight Therapeutics – Series D – $372.5M – July 2025
MapLight Therapeutics raised an oversubscribed $373 million series D round, co-led by Forbion and Goldman Sachs Alternatives, with strategic participation from Sanofi. The financing supports its differentiated CNS pipeline targeting schizophrenia and Alzheimer’s disease psychosis, with Phase II timing highlighting near-term data catalysts. The company subsequently completed an IPO later in 2025, marking a full private-to-public transition within the same year. Sanofi’s involvement indicates potential future partnership or acquisition interest. Its lead program, ML-007C-MA, is an oral M1/M4 muscarinic agonist addressing a significant unmet need in CNS disorders.
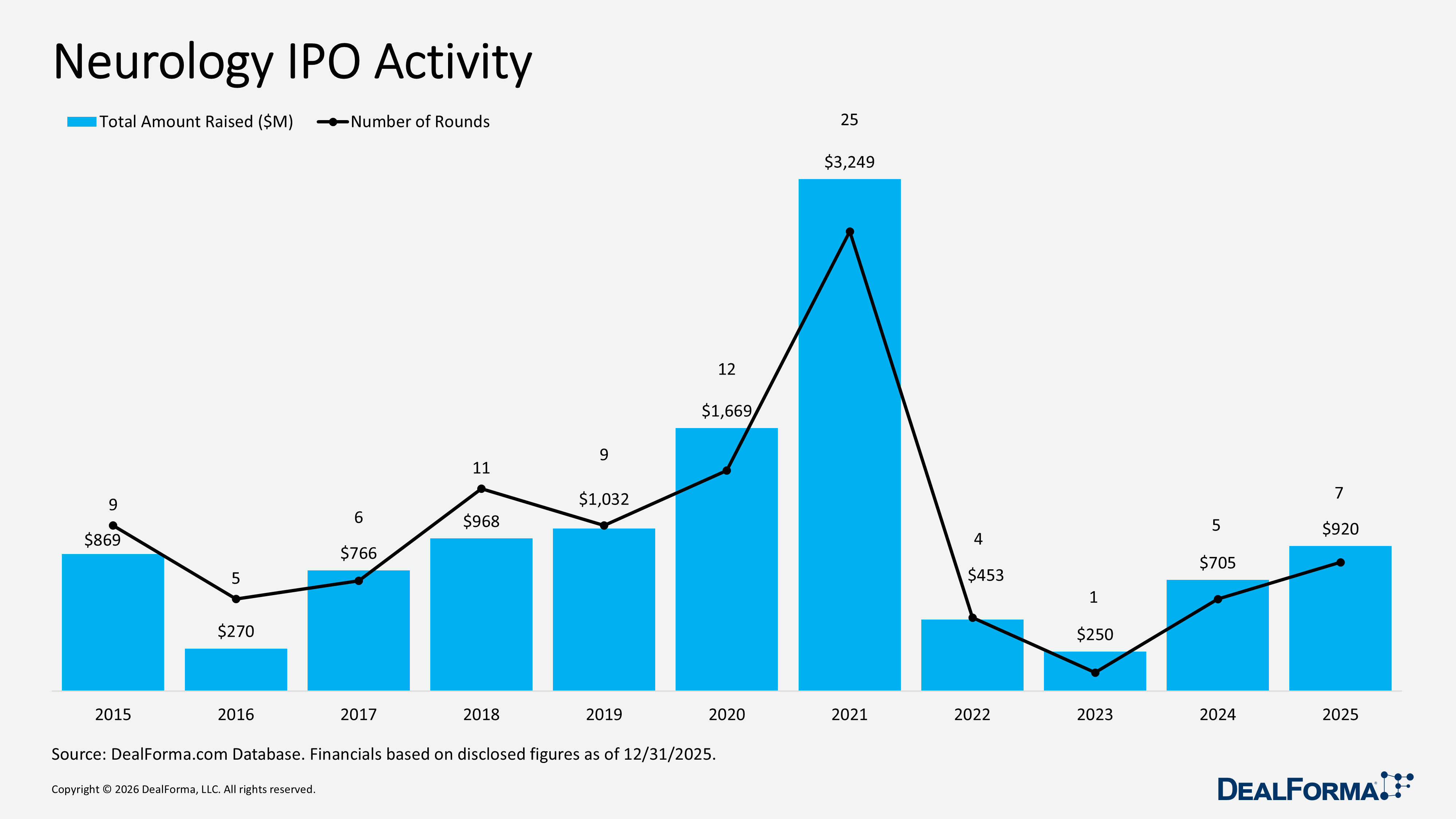
Neurology IPO Activity
Neurology IPO activity improved marginally in 2025, with listings increasing to 7 from 5 and total proceeds rising to $920 million from $705 million. However, the average IPO size declined to $131 million from $141 million, indicating smaller deal sizes despite higher market participation. Across 2024 and 2025 combined, the sector recorded 12 IPOs raising $1.6 billion.
Prominent Neurology IPO Activity in 2025
MapLight Therapeutics – IPO – $296M – October 2025
MapLight Therapeutics completed its IPO, issuing 16,962,500 shares at $17.00 per share, including full exercise of the 2,212,500-share greenshoe option. The company also raised additional capital through a concurrent private placement of 476,707 shares to affiliates of Goldman Sachs & Co. LLC. Total gross proceeds from the IPO and private placement reached $296 million, before fees and expenses. The shares began trading on the Nasdaq Global Select Market on October 27, 2025, under the ticker “MPLT.”
LB Pharmaceuticals – IPO – $285M – September 2025
LB Pharmaceuticals raised $285 million in an upsized IPO, closing just 19 days after announcement and ending a 7-month gap in large biotech IPOs in 2025, following Aardvark Therapeutics’ $94 million offering in February. The deal was increased from 16.7M to 19M shares, with the stock closing approximately 15% higher on debut. Proceeds will support the development of lead candidate LB-102, an oral derivative of amisulpride, with $133 million allocated to Phase III in schizophrenia and $25 million to Phase II in bipolar depression. Phase II results met the primary endpoint in January 2025, strengthening the clinical profile. The company expects IPO proceeds to fund operations through Q1 2028.
Saluda Medical – IPO – $151M – December 2025
Saluda Medical completed its IPO on the Australian Securities Exchange, raising A$231 million (approx. $150 million) through the issuance of 87 million CDIs at A$2.65 per share. The stock began trading on December 5, 2025, under the ticker “SLD.” Saluda is a commercial-stage medical device company developing treatments for chronic neurological conditions using its proprietary neuromodulation platform. Its closed-loop, dose-control technology measures neural responses and automatically adjusts therapy in real time based on neurophysiological feedback, differentiating it from conventional systems. IPO proceeds will support commercial expansion, including sales, marketing, and product development. Existing investors include Wellington Management, Fidelity Management & Research Company, T. Rowe Price Associates, TPG Life Sciences Innovations, and Redmile Group.
Also check out Neurologic R&D Partnerships, M&A, Ventures, and IPOs in 2024