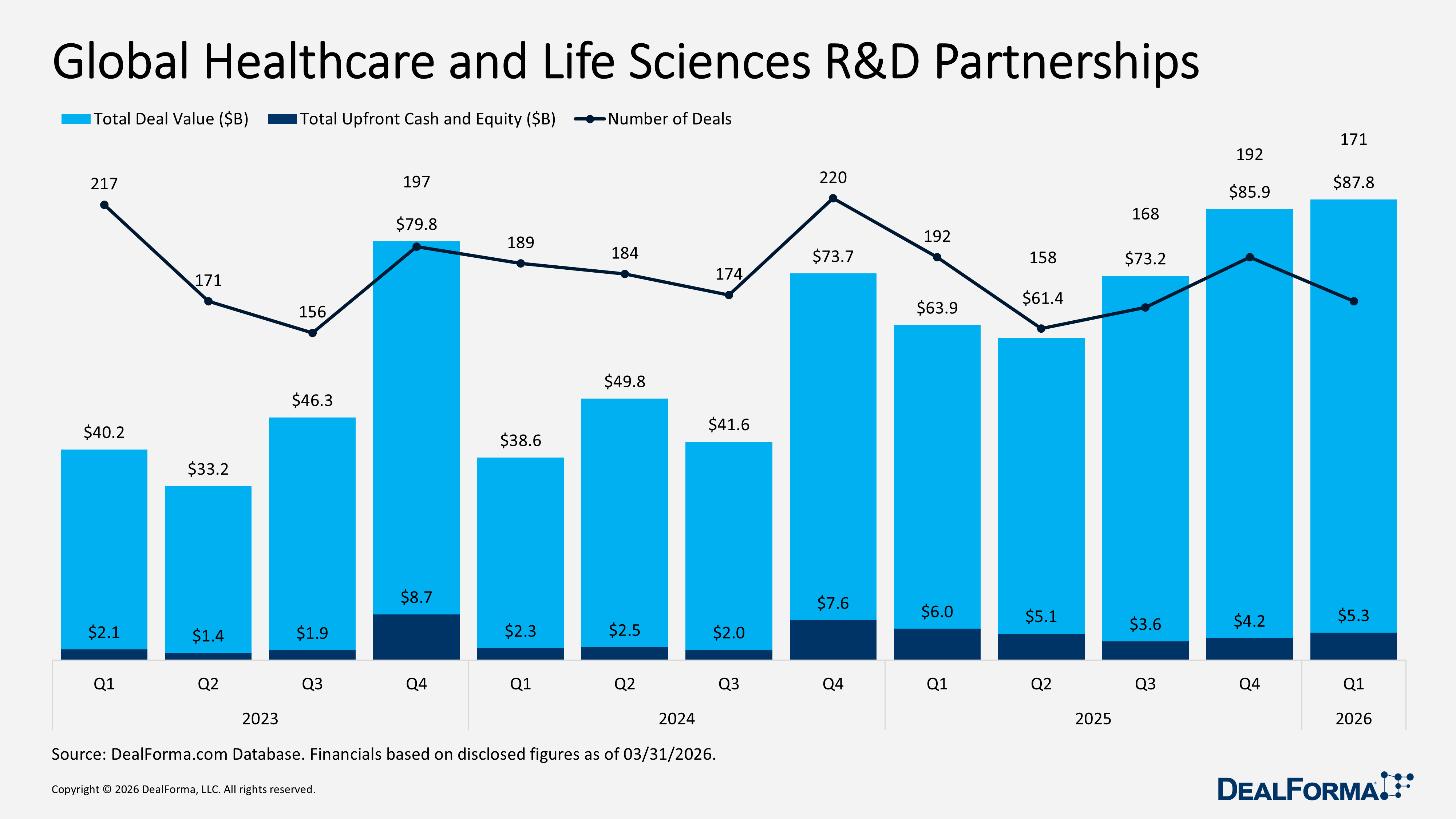
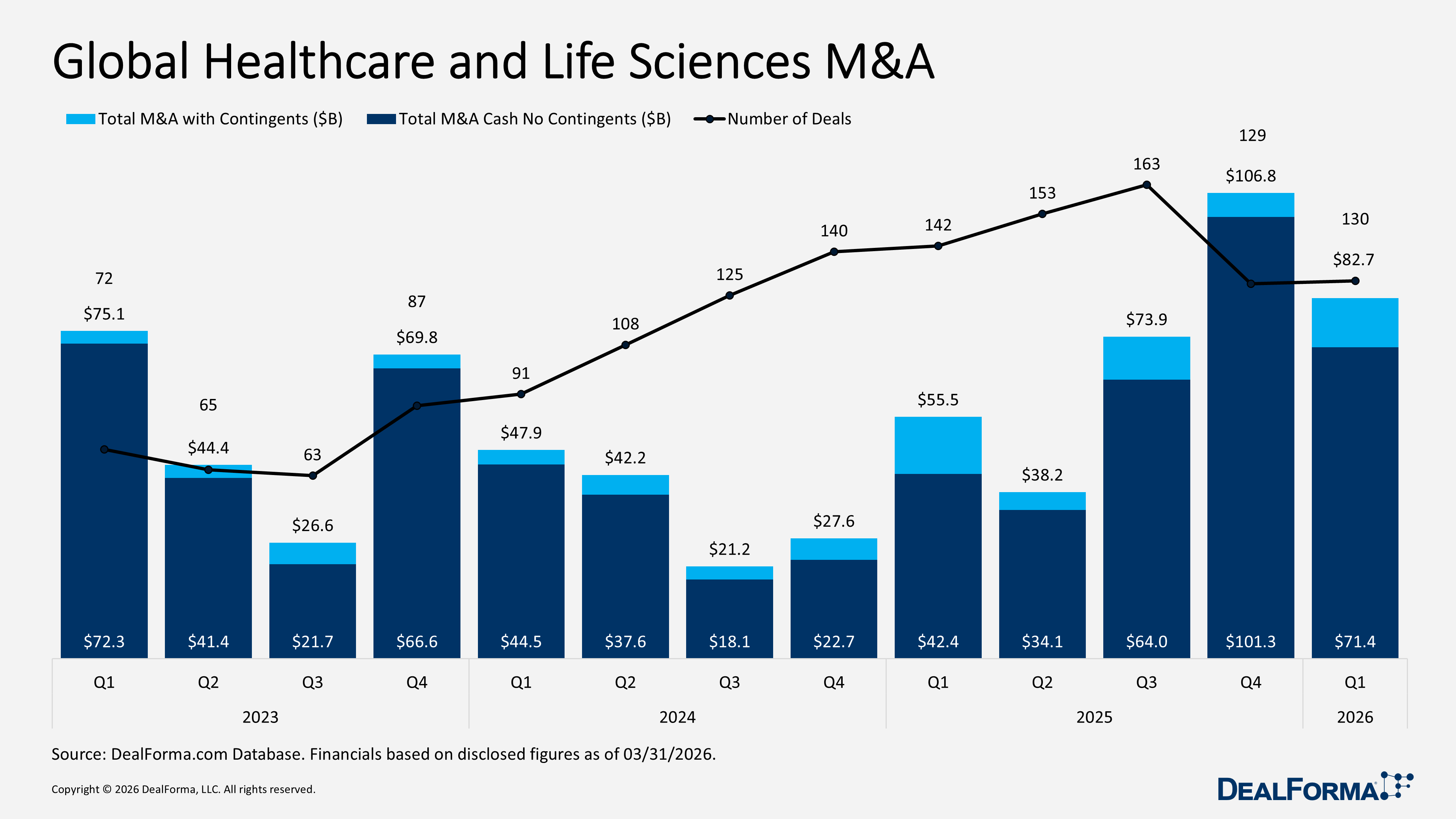
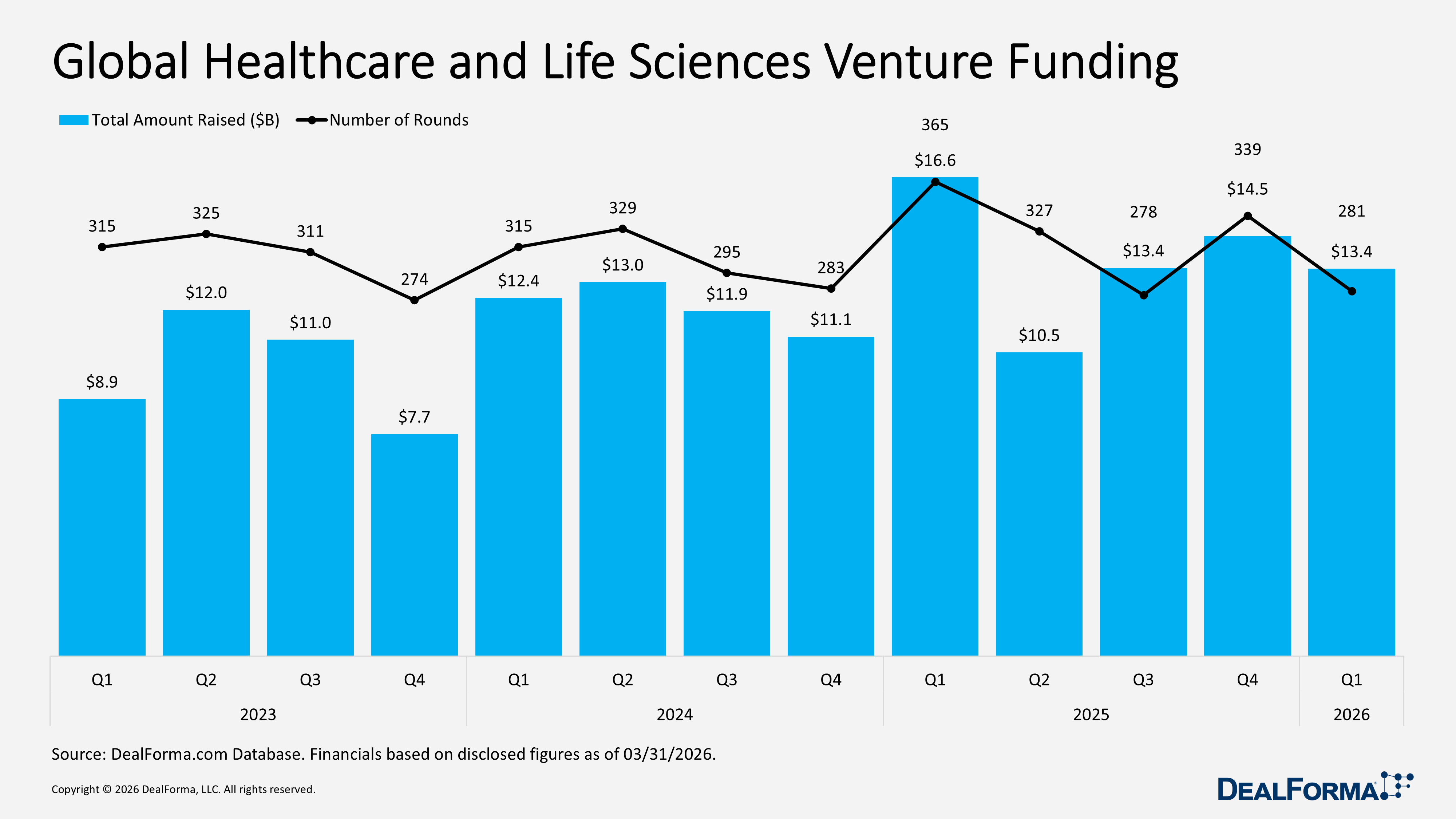
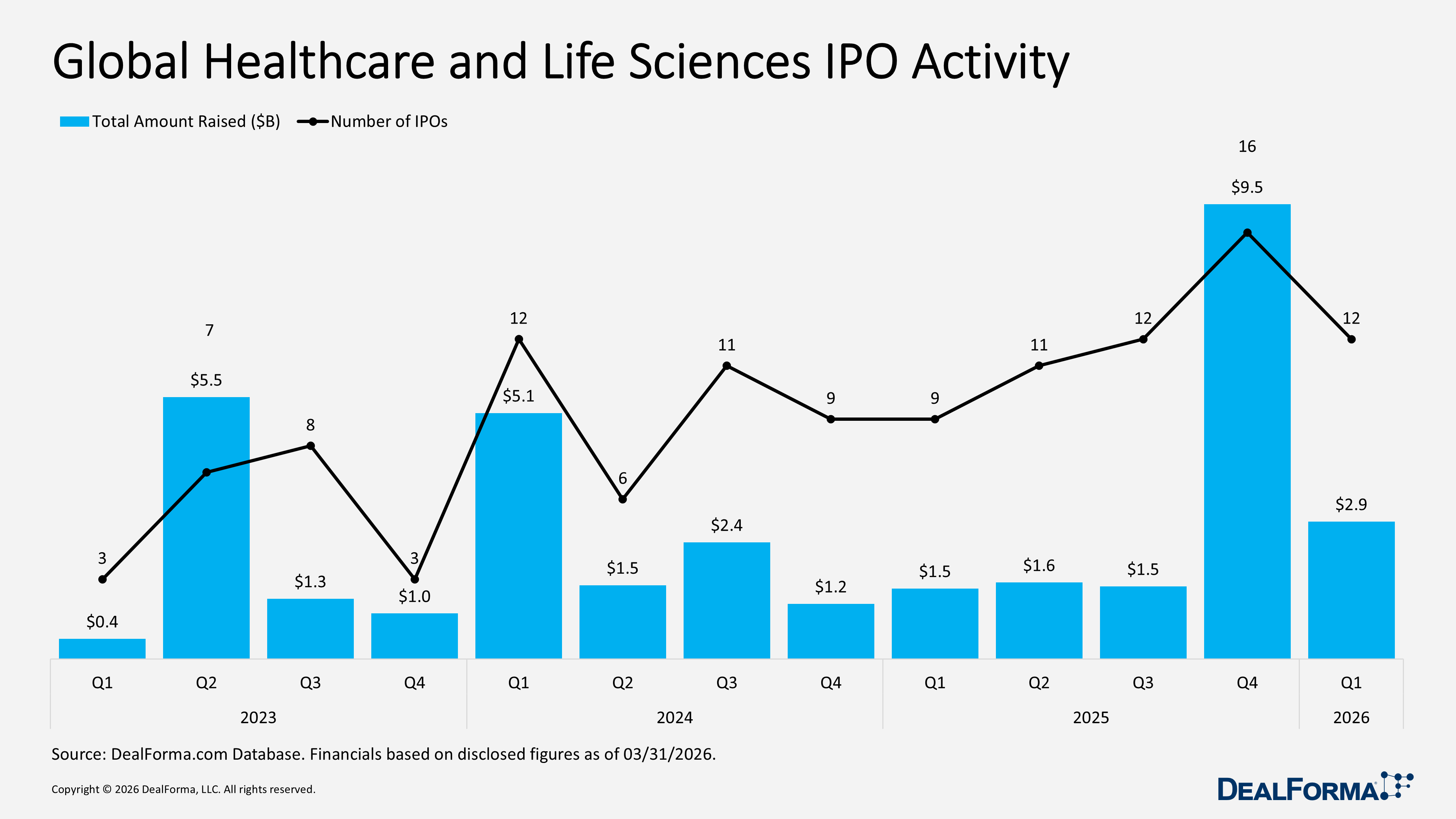
Our Q1 2026 review reflects a more selective and disciplined market, with capital concentrated in high-value partnerships and mid-sized M&A, while venture funding and IPO activity softened, suggesting a cautious yet active investment environment. R&D partnerships remained strong with 171 deals totaling $87.8 billion in value and $5.3 billion in upfront cash and equity, down from 192 deals and $85.9 billion in total value in Q4 2025, although the average upfront increased to $112 million (median $50 million). M&A activity stayed steady at 130 deals, but total value fell to $82.7 billion, including $71.4 billion in cash, with a median upfront of $625 million and an average upfront of $1.9 billion, compared to Q4 2025’s $106.8 billion total value, $101.3 billion in cash, $305 million median, and $2.2 billion average upfront. Venture funding slowed to 281 rounds raising $13.4 billion, down from 339 rounds and $14.2 billion in Q4 2025, while IPO activity dropped to 12 IPOs raising $2.9 billion, compared to 16 IPOs and $9.5 billion in Q4 2025.
Key deals included, CSPC & AstraZeneca ($1.2 billion upfront, up to $17.3 billion milestones), Innovent & Lilly ($350 million upfront, up to $8.5 billion milestones), and RemeGen & AbbVie ($650 million upfront, up to $5 billion milestones) in R&D; Boston Scientific & Penumbra ($14.5 billion), Danaher & Masimo ($9.9 billion), and Gilead & Arcellx ($8.2 billion) in M&A; Earendil Labs ($787 million), Parabilis Medicines ($305 million), and Corxel Pharmaceuticals ($287 million) in venture funding; and Generate Biomedicines ($400 million), Eikon Therapeutics ($381 million), and Veradermics ($295 million) in IPOs.
Global Healthcare and Life Sciences R&D Partnerships
In Q1 2026, global healthcare and life sciences R&D partnerships remained robust, with 171 deals totaling $87.8 billion in value and $5.3 billion in upfront cash and equity. While deal volume declined from the previous quarter, the average upfront increased to $112 million, indicating a higher capital commitment per deal. In comparison, the median remained $50 million. In comparison, Q4 2025 recorded 192 deals with a similar total value of $85.9 billion, but lower upfront funding of $4.2 billion, an average upfront of $76 million, and the same $50 million median.
Prominent Global Healthcare and Life Science R&D Partnerships in Q1 2026
CSPC Pharmaceutical development and commercialization deal with AstraZeneca – January 2026
CSPC Pharmaceutical granted AstraZeneca exclusive worldwide rights, excluding Greater China, to co-develop and commercialize 8 metabolic programs, including 1 clinical-ready GLP-1R/GIPR agonist (SYH-2082), 3 preclinical assets, and 4 earlier-stage programs. The collaboration utilizes CSPC’s AI-driven peptide platform and once-monthly LiquidGel delivery technology. AstraZeneca will lead late-stage development and commercialization outside China. At the same time, CSPC retains rights across Mainland China, Hong Kong, Macau, and Taiwan, with AstraZeneca holding an option to co-commercialize in these markets upon approval. Financial terms include $1.2 billion upfront, up to $3.5 billion in development and regulatory milestones, and up to $13.8 billion in commercial milestones.
Innovent development and commercialization deal with Eli Lilly – February 2026
Innovent, through its subsidiaries, granted Lilly exclusive worldwide rights, excluding China, to develop and commercialize antibody programs. Innovent will lead early research and clinical development through Phase II in China, leveraging its antibody platforms and clinical capabilities, while Lilly will oversee global development and commercialization. Innovent retains rights in Greater China. Financial terms include $350 million upfront, with up to $8.5 billion in development, regulatory, and commercial milestones, plus tiered royalties.
RemeGen development and commercialization deal with AbbVie – January 2026
RemeGen granted AbbVie exclusive worldwide rights, excluding China, to develop, manufacture, and commercialize RC148, a PD-1/VEGF bispecific antibody for advanced solid tumors. The asset is being evaluated as both a monotherapy and in combination, including with antibody-drug conjugates, across multiple tumor types such as certain lung cancers. Financial terms include $650 million upfront, with up to $5 billion in development, regulatory, and commercial milestones, plus double-digit tiered royalties.
Global Healthcare and Life Sciences M&A
In Q1 2026, global healthcare and life sciences M&A activity remained stable at 130 deals, in line with the previous quarter, but total value declined to $82.7 billion, including $71.4 billion in cash without contingents. Despite the lower aggregate value, the median upfront increased to $625 million, indicating larger deal sizes, while the average upfront fell to $1.9 billion. In Q4 2025, deal volume was also 129, but total value was significantly higher at $106.8 billion, with $101.3 billion in cash without contingents, an average upfront of $2.2 billion, and a median of $305 million.
Prominent Global Healthcare and Life Sciences M&A in Q1 2026
Boston Scientific acquiring Penumbra – January 2026
Boston Scientific is acquiring Penumbra, a provider of FDA-approved neuro and peripheral vascular medical devices. Its portfolio includes neurovascular products such as ACCESS25, ARTEMIS, BMX81, and BMX96 systems, and ACE reperfusion catheters, as well as peripheral devices including the Indigo system, CAT RX, Lightning Flash 2.0, and Lightning Bolt 12. The transaction, expected to close in 2026, subject to regulatory approvals, will result in Penumbra being delisted from the NYSE. Penumbra shareholders can elect to receive $374 in cash or 3.87 shares of Boston Scientific stock, reflecting a 19.3% premium and an enterprise value of $14.5 billion, with consideration structured as 73.26% cash and 26.74% stock. Stock option holders and accelerated RSUs will convert into $274 in cash plus 1.03 Boston Scientific shares.
Danaher acquiring Masimo – February 2026
Danaher is acquiring Masimo in an all-cash transaction to strengthen its Diagnostics segment with Masimo’s leadership in pulse oximetry and advanced patient monitoring. Masimo’s portfolio includes Pulse oximetry (Masimo SET), advanced blood monitoring (Rainbow Pulse CO-Oximetry), modular and portable monitors (Root, Radical-7, Rad-97), wearable and handheld devices (Radius VSM, Radius PPG, Rad-67, MightySat Rx, Rad-G, Masimo W1), and hospital automation/connectivity platforms (Patient SafetyNet, Masimo SafetyNet, Iris Gateway/iSirona). Masimo will operate as a wholly owned subsidiary of Danaher and be delisted from Nasdaq. The company is projected to generate over $530 million EBITDA in 2027, with expected synergies of more than $125 million in annual cost savings and more than $50 million in annual revenue gains by year five post-acquisition. The deal values Masimo at approximately 18x estimated 2027 EBITDA (15x including synergies) and is expected to close in H2 2026. Shareholders will receive $180 per share in cash, a 38.3% one-day premium, for a total enterprise value of $9.9 billion, including assumed debt and net of acquired cash.
Gilead acquiring Arcellx – February 2026
Gilead is acquiring Arcellx, whose pipeline includes the preregistration BCMA-directed CAR T-cell therapy Anticabtagene autoleucel for relapsed/refractory multiple myeloma, multiple Phase I programs for AML, MDS, myasthenia gravis, and preclinical/discovery programs for AML, HCC, and SCLC. The acquisition builds on Gilead’s existing Kite collaboration and is expected to close by Q2 2026. Arcellx shareholders will receive $115 per share in cash, a 79% one-day premium (68% vs 30-day VWAP as of Feb 20, 2026), and $5 per CVR if cumulative Anticabtagene autoleucel sales reach $6 billion through 2029, valuing the deal at $8.2 billion. Remaining shares will be acquired via a second-step merger at the same terms. Gilead extended its tender offer on April 1, 2026, to April 24, 2026.
Global Healthcare and Life Sciences Venture Funding
In Q1 2026, global healthcare and life sciences venture activity declined with 281 funding rounds raising $13.4 billion, down from 339 rounds and $14.5 billion in Q4 2025, indicating a drop in both deal volume and capital raised. Despite this slowdown, overall activity remained solid, with 620 rounds and $27.9 billion raised across the two quarters combined.
Prominent Global Healthcare and Life Sciences Venture Funding in Q1 2026
Earendil Labs – Series Unspecified – $787M – March 2026
Earendil Labs, an AI-driven biotechnology company, raised $787 million from a syndicate of global investors, including Dimension Capital, DST Global, INCE Capital, Luminous Ventures, Miracle Capital, Sanofi, and the Biotech Development Fund (Hillhouse and Pfizer). The funding will scale its AI-enabled R&D platform, expand teams, and advance a pipeline of over 40 antibody and biologics programs. Lead asset HXN-1001, a half-life extended anti-TL1A antibody, is Phase 2-ready, with multiple IND filings planned in 2026 and 2027.
Parabilis Medicines – Series F – $305M – January 2026
Parabilis Medicines, a clinical-stage oncology company, raised $305 million in a series F round led by RA Capital Management, Fidelity Management & Research Company, and Janus Henderson Investors, with participation from new and existing investors. The financing was completed at a higher valuation than its prior round. Proceeds will advance clinical development of FOG-001 (zolucatetide), including progression toward a registrational trial in desmoid tumors and broader evaluation across tumor types, while also supporting expansion of its discovery pipeline, including its prostate cancer program, and further development of its Helicon peptide platform targeting historically undruggable diseases.
Corxel Pharmaceuticals – Series D1 – $287M – January 2026
Corxel Pharmaceuticals, a clinical-stage cardiometabolic company, raised to $287 million in a series D1 financing with participation from new and existing investors, including SR One, TCGX, RA Capital Management, HBM Healthcare Investments, SymBiosis, Adage Capital Management, Invus, SilverArc Capital, RTW Investments, and Hengdian Group Capital. Proceeds will primarily advance CX11, an oral small molecule GLP-1 RA, currently in Phase 2 in the United States and Phase 3 in China, with plans to progress into global Phase 2 trials for Type 2 Diabetes Mellitus and initiate Phase 3 development. Funding will also support the advancement of additional cardiometabolic programs, including acute ischemic stroke and hypertension, and strengthen global clinical capabilities.
Global Healthcare and Life Sciences IPO Activity
In Q1 2026, global healthcare and life sciences IPO activity weakened, with just 12 IPOs raising $2.9 billion, down from 16 IPOs and $9.5 billion in Q4 2025, reflecting a sharp decline in both listings and capital raised and signaling a more cautious market. Across the two quarters, the sector recorded 28 IPOs, raising a total of $12.3 billion.
Prominent Global Healthcare and Life Sciences IPO Activity in Q1 2026
Generate Biomedicines – IPO – $400M – February 2026
Generate Biomedicines priced its IPO of 25,000,000 shares at $16.00 per share, raising expected gross proceeds of $400 million before underwriting discounts, commissions, and expenses. The company also granted underwriters a 30-day option to purchase up to an additional 3,750,000 shares at the IPO price. The shares are expected to begin trading on the Nasdaq Global Select Market under the ticker “GENB” on February 27, 2026, with the offering set to close on March 2, 2026, subject to customary conditions. All shares are being offered by the company. Goldman Sachs & Co. LLC and Morgan Stanley are acting as joint lead book-running managers, with Piper Sandler, Guggenheim Securities, and Cantor serving as additional book-running managers.
Eikon Therapeutics – IPO – $381.2M – January 2026
Eikon Therapeutics priced an upsized IPO of 21,177,600 shares at $18.00 per share, generating expected gross proceeds of approximately $381 million before underwriting discounts, commissions, and expenses. The company also granted underwriters a 30-day option to purchase up to 3,176,640 additional shares. The shares are expected to begin trading on the Nasdaq Global Select Market under the ticker “EIKN” on February 5, 2026, with closing anticipated on or about February 6, 2026, subject to customary conditions. J.P. Morgan, Morgan Stanley, BofA Securities, Cantor, and Mizuho are acting as joint book-running managers. Eikon is a late-stage clinical biopharmaceutical company focused on developing innovative therapies to address serious unmet medical needs.
Veradermics – IPO – $294.8M – January 2026
Veradermics completed an upsized IPO of 17,339,294 shares at $17.00 per share, including 2,261,647 shares issued through the full exercise of the underwriters’ option, generating gross proceeds of approximately $295 million before underwriting discounts, commissions, and expenses. All shares were offered by the company. The shares began trading on the New York Stock Exchange under the ticker “MANE” on February 4, 2026. Veradermics is a dermatologist-founded, late clinical-stage biopharmaceutical company focused on developing innovative therapies for highly prevalent aesthetic and dermatological conditions.
Also check out Global Healthcare and Life Sciences IPO Activity – 2025 Review