Our Q2 2025 review of biopharma R&D partnerships highlights momentum in innovation, particularly across oncology, neurology, autoimmune, and cardiometabolic diseases. Despite a quarter-over-quarter decline in deal volume and total announced value, the period demonstrated strength in strategic collaborations, with a surge in diagnostics and manufacturing tech partnerships helping offset the pullback in therapeutic platform deals. The quarter featured high-value agreements, led by BioNTech and BMS’s landmark $11.1 billion deal in June for BNT-327, a Phase III bispecific antibody for solid tumors, which included $1.5 billion paid upfront.
In May, Pfizer partnered with 3SBio to acquire global rights (excluding China) to SSGJ-707, a Phase II oncology candidate, in a transaction valued at $6.2 billion, comprising $1.3 billion upfront and a $100 million equity investment. Vor Bio strengthened its autoimmune portfolio in June by licensing telitacicept from RemeGen, securing rights outside Greater China for $45 million upfront, $80 million in stock warrants, and over $4 billion in potential milestones. In April, GSK advanced its CNS pipeline with a $2.7 billion agreement with ABL Bio, anchored by a $49.6 million upfront for antibody and RNA therapies leveraging the Grabody-B BBB shuttle platform. Meanwhile, Novo Nordisk entered a May partnership with Septerna to co-develop four oral GPCR-targeting therapies for metabolic diseases, in a deal worth up to $2.7 billion, including $195 million upfront and $500 million in milestones.
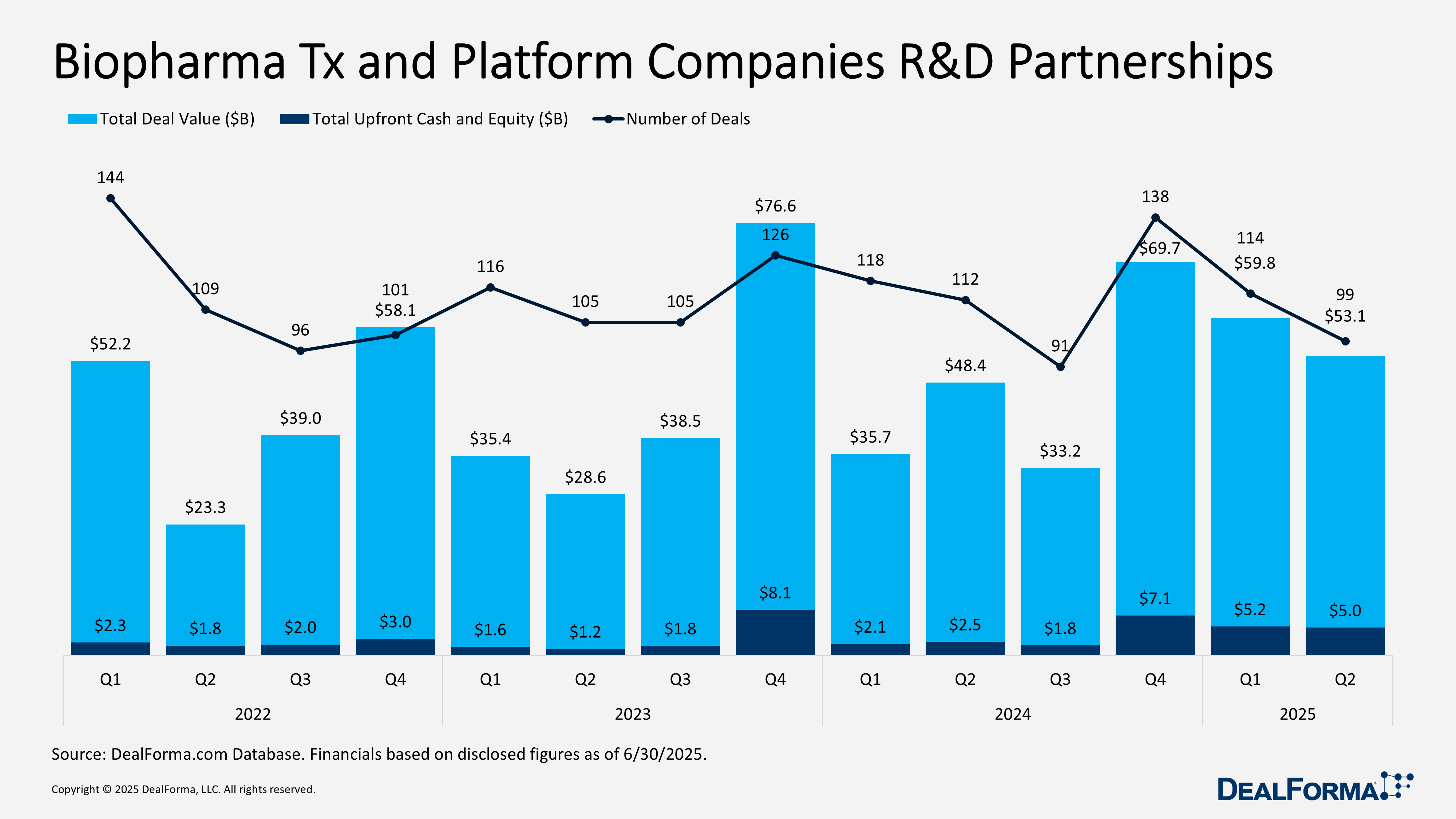
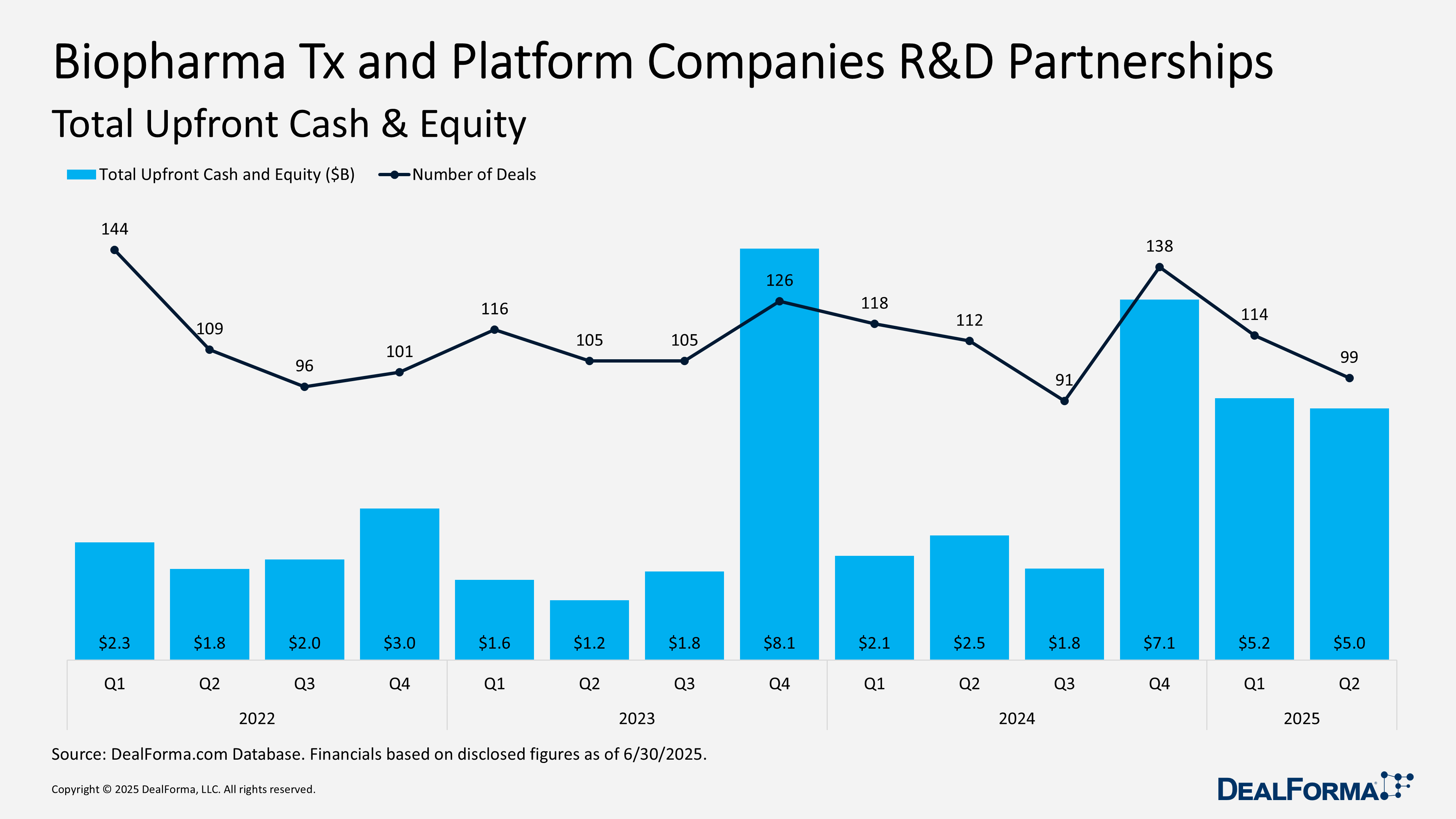
In Q2 2025, biopharma therapeutics and platform companies secured 99 R&D partnerships totaling $53.1 billion in announced deal value, including $5 billion in upfront payments and equity. This marks a slight decline from Q1, which saw 114 deals worth $59.8 billion and $5.2 billion upfront.
Quarter-over-quarter, deal volume declined by 13% and total announced value fell 11%, while upfront payments remained relatively stable with only a $200 million dip. Despite the Q2 slowdown, the first-half 2025 activity reached $112.9 billion across 213 partnerships.
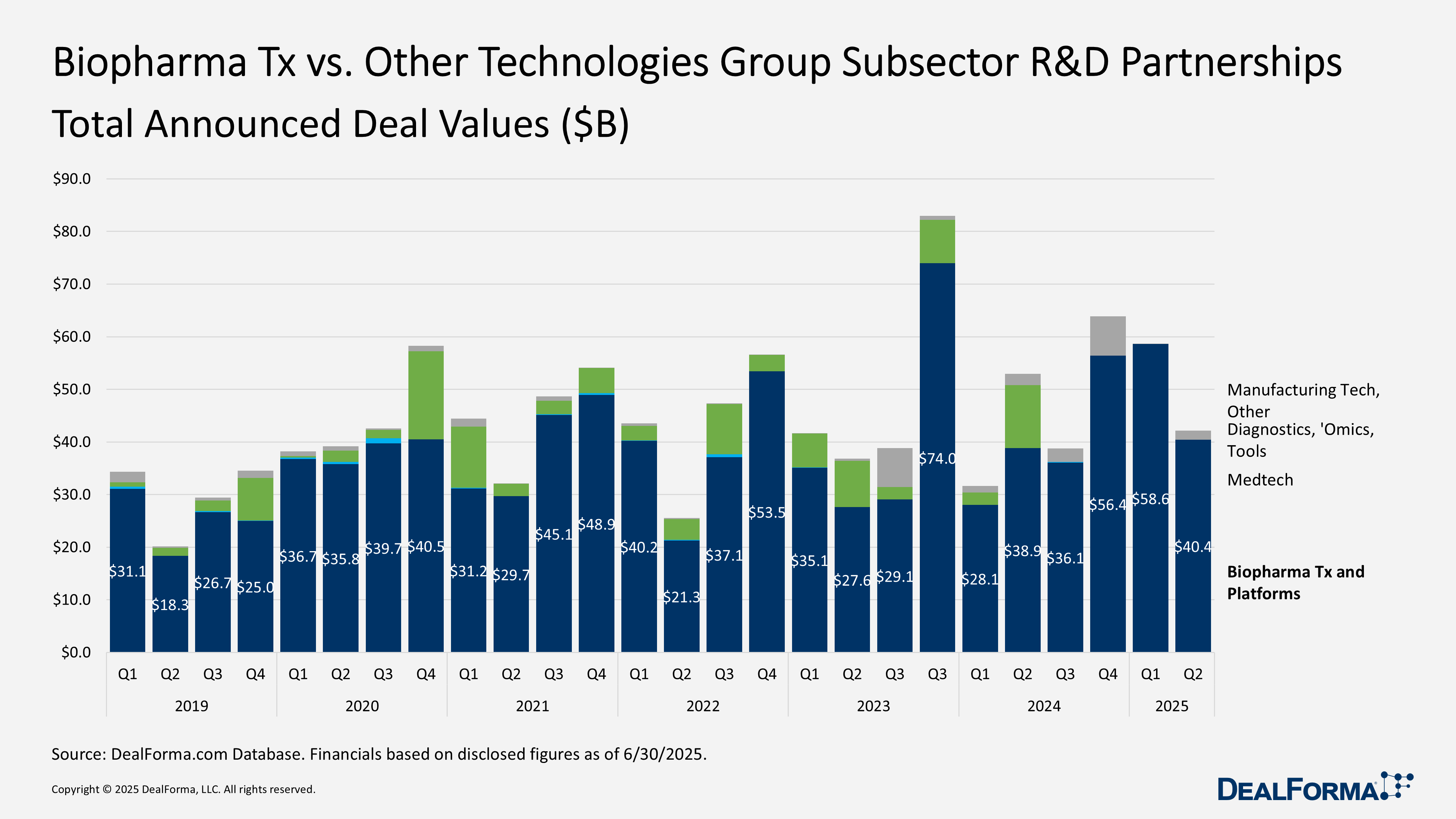
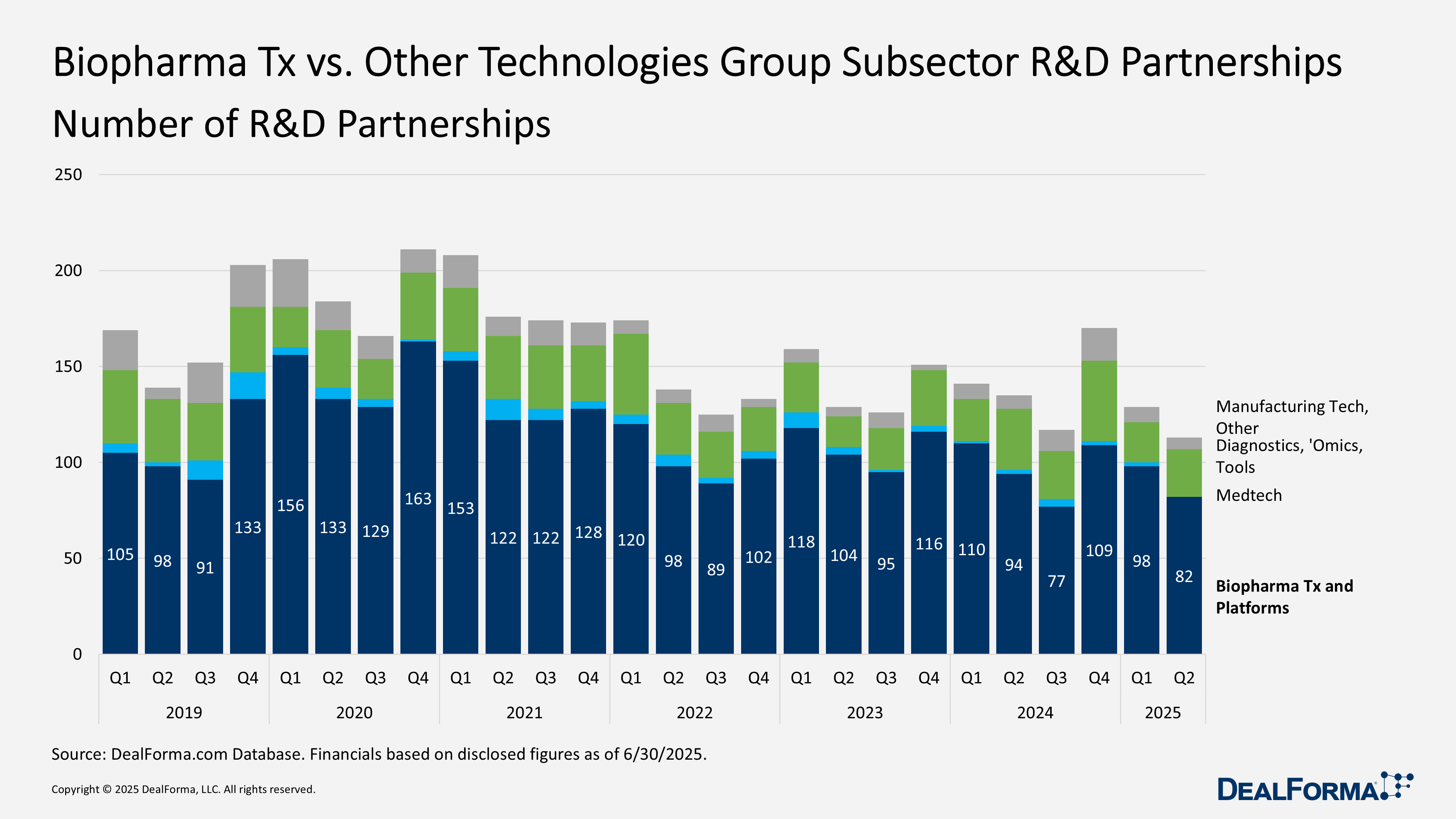
Biopharma Tx vs. Other Technologies Group Subsector
In Q2 2025, the composition of biopharma R&D partnerships shifted significantly. Therapeutic-platform deals remained the largest category, with 82 agreements totaling $40.4 billion, but this marked a sharp drop from Q1 98 deals valued at $58.6 billion. In contrast, diagnostics, sequencing, and omics partnerships surged, with deal value jumping over five-fold to $12 billion across 25 deals, up from $2.3 billion and 21 deals in Q1.
Manufacturing technology remained a small segment but saw a sharp rise in value, reaching $1.7 billion across 6 deals, up from just $100 million on 8 deals in Q1. Medtech and digital health recorded no activity after 2 minor deals last quarter. Overall, therapeutic platforms still dominated H1 2025 with $99 billion of the $115 billion disclosed, but momentum shifted toward diagnostics and manufacturing technologies.
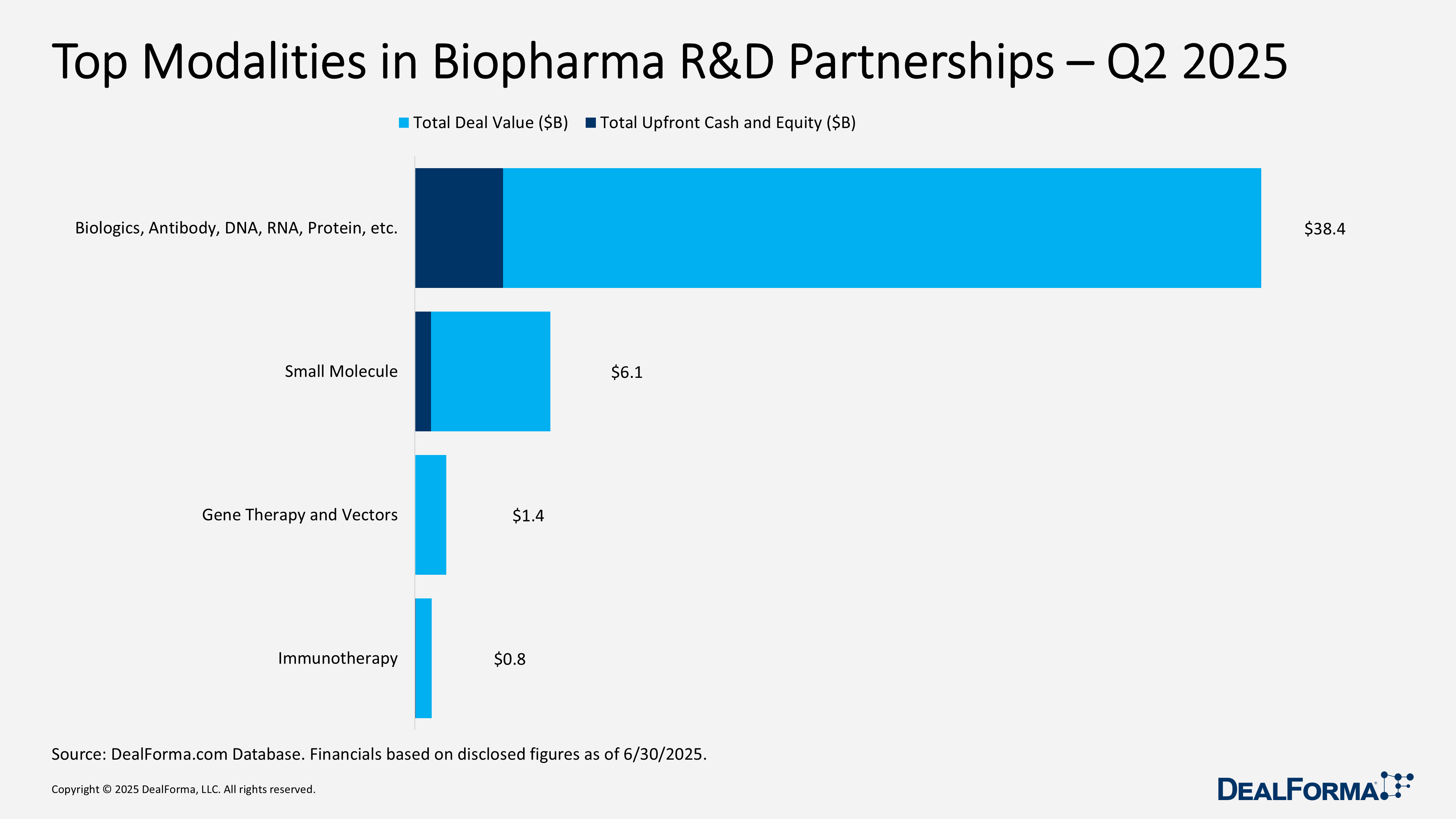
Top Modalities in Biopharma R&D Partnerships in Q2 2025
In Q2 2025, biologics-based partnerships dominated biopharma deal-making, representing 68 of 96 tracked alliances, 71% of total activity, and generating $38.4 billion in announced value, including $4 billion in upfront payments. Small-molecule deals followed with 24 agreements totaling $6.1 billion and $740 million upfront. Gene therapy and vector deals (2 deals, $1.4 billion) and immunotherapy collaborations (2 deals, $765 million) remained niche, attracting modest upfronts of $18 million and $35 million, respectively.
Top R&D Partnerships in Q2 2025
BioNTech development and commercialization deal with BMS – June 2025
BioNTech granted BMS exclusive global rights to develop and commercialize BNT-327, a Phase III bispecific antibody targeting PD-L1 and VEGF-A for multiple solid tumors, including small cell and non-small cell lung cancer. A Phase III trial in triple-negative breast cancer is planned for late 2025. BioNTech retains rights in the US, EU, UK, China, and Turkey, while BMS holds rights elsewhere. BioNTech receives $1.5 billion upfront, up to $2 billion in fixed payments through 2028, and is eligible for up to $7.6 billion in milestones.
3SBio development and commercialization deal with Pfizer – May 2025
Pfizer obtained exclusive rights (ex-China) to develop and commercialize SSGJ-707, a Phase II bispecific antibody targeting PD-1 and VEGF for NSCLC, metastatic colorectal cancer, and gynecological tumors. Pfizer may also commercialize the therapy in China and will manufacture it in the US. 3SBio will begin a Phase III trial in China in 2025. The deal includes $1.3 billion upfront, $100 million in equity, and up to $4.8 billion in milestones.
RemeGen development and commercialization deal with Vor Bio – June 2025
Vor Bio licensed global rights (excluding China, Hong Kong, Macau, and Taiwan) to telitacicept, a dual-target fusion protein approved in China for generalized myasthenia gravis (gMG), SLE, and RA. A global Phase III trial in gMG is ongoing. RemeGen will receive $45 million upfront, $80 million in stock warrants, and is eligible for over $4 billion in milestones, plus tiered royalties. The deal adds a late-stage autoimmune asset to Vor Bio’s pipeline.
ABL Bio development and commercialization deal with GSK – April 2025
GSK acquired global rights from ABL Bio to develop antibody and RNA-based therapies, including siRNA and ASOs, for neurological diseases. The partnership leverages ABL’s Grabody-B platform for enhanced blood-brain barrier delivery. ABL will receive £38.5 million ($49.6 million) upfront and is eligible for up to £2.1 billion ($2.7 billion) in milestones, plus tiered royalties. The deal supports GSK’s expansion in CNS innovation.
Septerna development and commercialization deal with Novo Nordisk – May 2025
Novo Nordisk secured global rights from Septerna to develop four oral small-molecule therapies targeting GPCRs, including GLP-1, GIP, and glucagon receptors, for obesity, type 2 diabetes, and other cardiometabolic diseases. The partnership spans early discovery through candidate selection, with Novo leading IND-stage development. Septerna may opt into profit-sharing for one program. The deal includes up to $195 million in upfront, $500 million in near-term milestones, $2 billion in future milestones, and tiered royalties.
Also check out Biopharma Therapeutics and Platforms R&D Partnerships Q1 2025