DealForma review of Q1 2025 saw a surge in biopharma venture funding, with several companies securing $200 million+ rounds to advance differentiated therapies across metabolic, neurological, immunological, and ophthalmic diseases. Backed by top investors, Verdiva Bio launched with a $411 million series A, co-led by Forbion and General Atlantic, to develop oral and injectable therapies for obesity and cardiometabolic diseases, including a once-weekly oral GLP-1 and amylin treatment.
Aviceda Therapeutics raised $208 million in an upsized series C, co-led by Omega Funds and TCGX, to advance AVD-104 for geographic atrophy, with pivotal Phase 2b/3 trial data expected in H2 2025. Abcuro closed a $200 million series C led by NEA to fund the Phase 2/3 MUSCLE trial for its first-in-class anti-KLRG1 antibody, ulviprubart, targeting inclusion body myositis. Windward Bio secured $200 million in series A funding, led by OrbiMed and Novo Holdings, to advance WIN378, a long-acting anti-TSLP antibody for severe asthma, with initial data expected in 2026. Finally, Tenvie Therapeutics raised $200 million in seed funding, led by ARCH Venture Partners and F-Prime Capital, to develop brain-penetrant small-molecule therapies for neurological diseases, including NLRP3 and SARM1 inhibitors.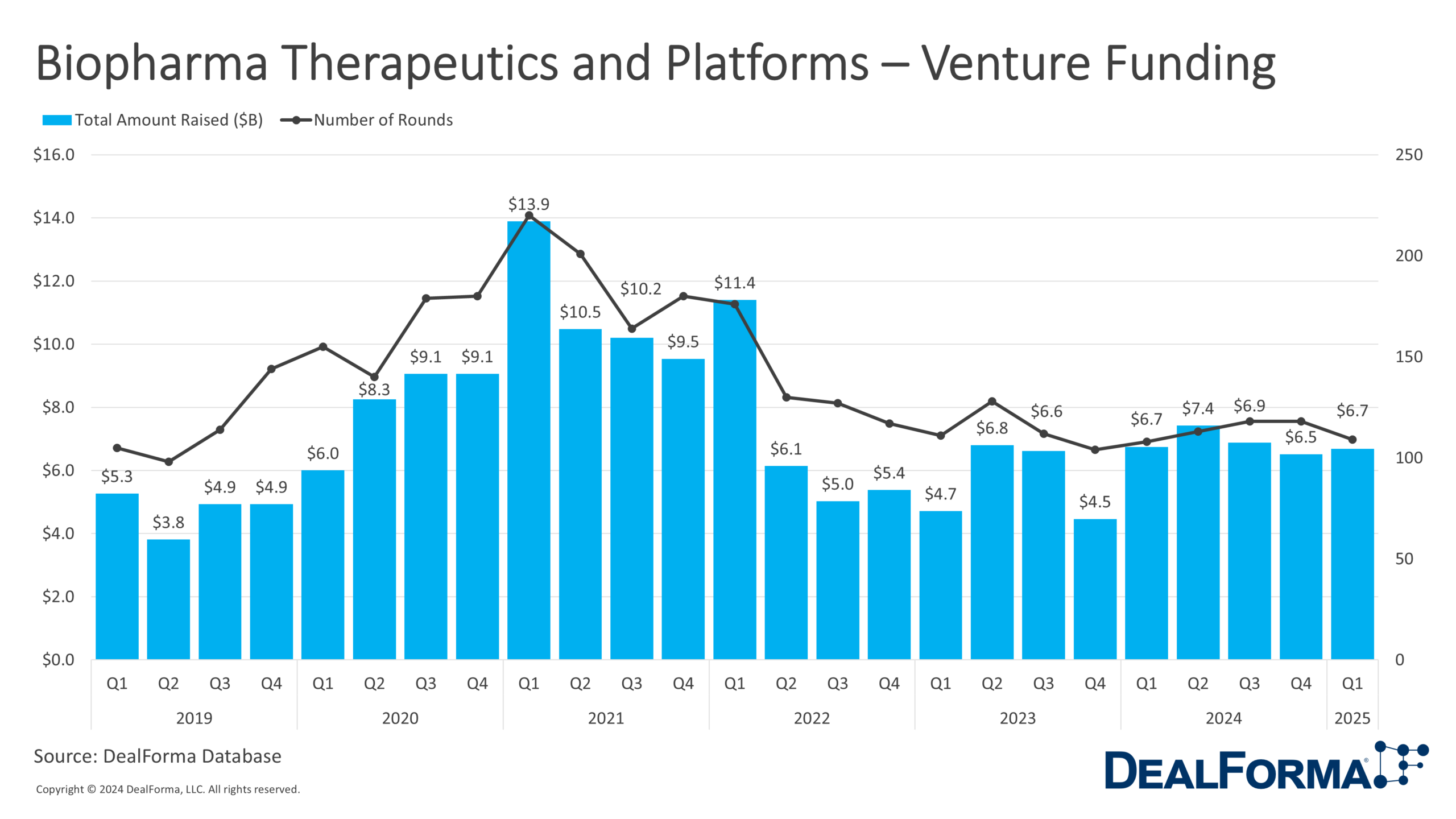
In Q1 2025, biopharma therapeutics and platform startups raised $6.7 billion across 109 venture rounds, with an average deal size of $70 million. While deal volume declined slightly from Q4 2024 (118 rounds), total funding rose from $6.5 billion, driven by larger individual investments. Combined, the last 2 quarters saw $13.2 billion across 227 rounds.
Biopharma Venture Funding by Series
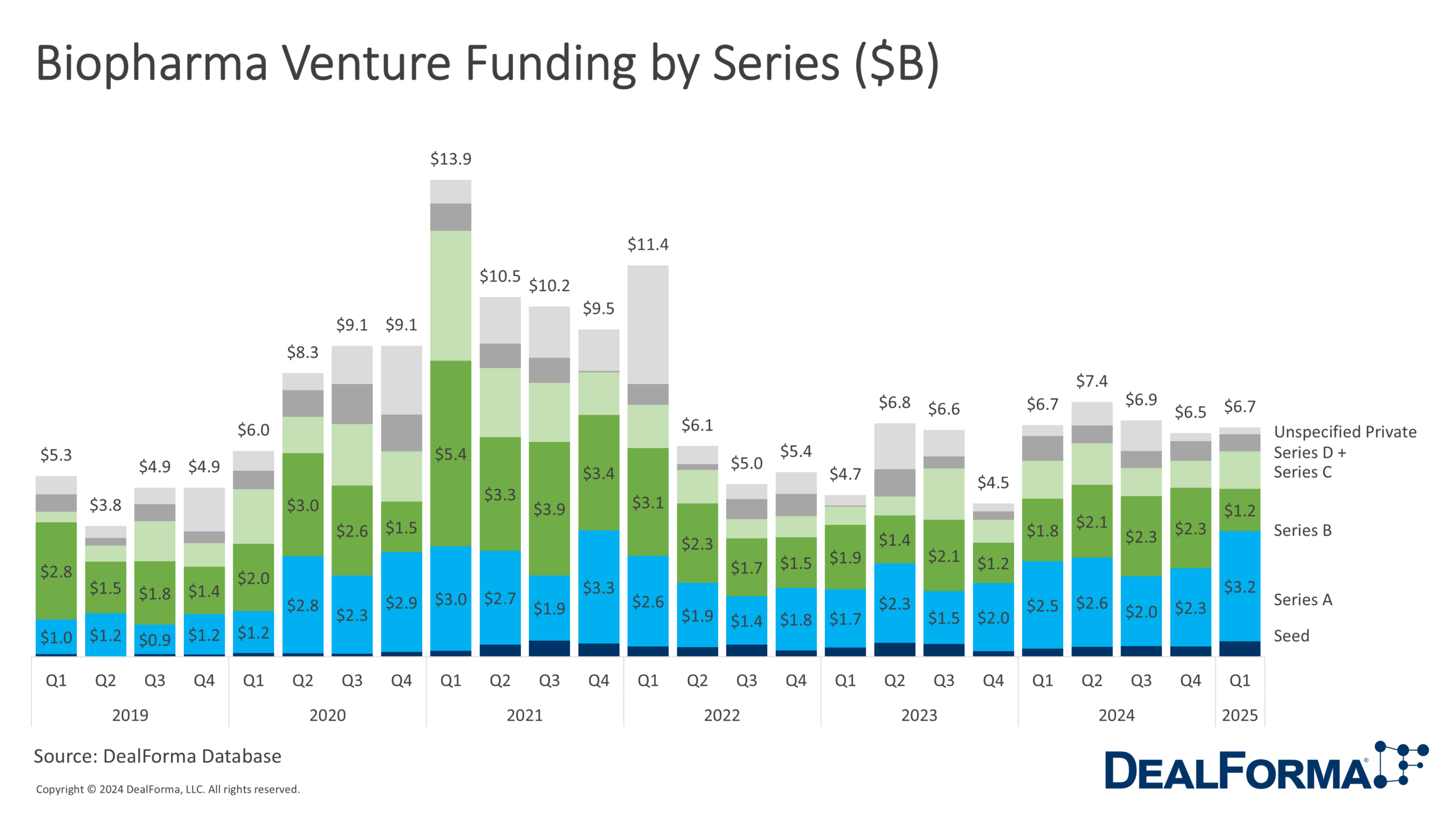
In Q1 2025, biopharma venture funding reached $6.7 billion across 109 rounds, slightly exceeding Q4 2024’s $6.5 billion despite a drop in deal count. Investment shifted toward earlier stages, with series A surging 40% to $3.2 billion and seed funding rising to $500 million. In contrast, series B fell to $1.2 billion. Later-stage remained stable, with weaker series C edged up to $1.1 billion, while series D and beyond declined to $500 million. Over the last 2 quarters, 227 rounds raised $13.2 billion, with more than 40% allocated to series A.
Top Therapeutic Areas for Venture Funding – Q1 2025

In Q1 2025, cancer startups led biopharma venture funding with $1.6 billion across 29 rounds, averaging $63 million per deal. Neurology followed closely with 25 rounds totaling $1.2 billion ($57 million average). Infectious disease ventures raised $200 million across 7 rounds, while pulmonary startups stood out with $300 million from just 5 deals, matching oncology’s $63 million average. These 4 therapeutic areas accounted for 66 of the 109 total deals and $3.3 billion, nearly half of the quarterly funding.
Top Biopharma Technologies for Venture Funding – Q1 2025

In Q1 2025, platform biologics antibodies, nucleic acids, and protein-based technologies led biopharma venture funding with $2.1 billion across 31 rounds, averaging $69 million per deal. Small-molecule drug developers followed with $1 billion from 27 rounds, affirming ongoing support for chemistry-driven pipelines. Cell therapy startups secured $400 million across nine rounds ($47 million average), while gene therapy and vector platforms raised a similar $400 million from 10 deals ($40 million average). Though immunotherapy saw just 3 deals, they were sizable, averaging $137 million each, matching cell and gene therapies in total dollars. Combined, these 5 technology areas attracted 80 deals and $4.2 billion, over 60% of the quarter’s total.
Top Venture Funding in Q1 2025
Verdiva Bio – Series A – $410M – January 2025
Verdiva Bio launched with a $411 million oversubscribed series A co-led by Forbion and General Atlantic, alongside RA Capital, OrbiMed, Logos Capital, Lilly Asia Ventures, and LYFE Capital. Based in London and San Francisco, Verdiva is developing a pipeline of oral and injectable therapies targeting obesity and cardiometabolic diseases. Its lead candidates include once-weekly oral GLP-1 and amylin treatments for weight loss, supported by a proprietary oral delivery platform to improve patient convenience, scalability, and access.
Aviceda Therapeutics – Series C – $207.5M – January 2025
Aviceda Therapeutics, a Cambridge-based clinical-stage biotech developing next-generation immunomodulators through its proprietary HALOS™ nanotechnology platform, raised $208 million in an upsized series C. The financing was co-led by Omega Funds and TCGX, with participation from Enavate Sciences, Jeito Capital, Blue Owl Healthcare Opportunities, Longitude Capital, OrbiMed, Logos Capital, Marshall Wace, Catalio, abrdn, and Digitalis Ventures. Proceeds will advance pivotal trials of AVD-104—a first-in-class, dual-mechanism therapy for geographic atrophy (GA) secondary to age-related macular degeneration—and support pipeline expansion into other ophthalmic indications. 12-month data from the fully enrolled Phase 2b/3 trial is expected in H2- 2025.
Abcuro – Series C – $200M – February 2025
Abcuro closed a $200 million series C led by NEA, with participation from Foresite Capital and returning investors including RA Capital, Bain Capital Life Sciences, Sanofi Ventures, and BlackRock. The funds will advance ulviprubart (ABC008), a first-in-class anti-KLRG1 antibody, through its registrational Phase 2/3 MUSCLE trial for inclusion body myositis (IBM), support a potential BLA filing, and prepare for commercial launch. Abcuro is developing targeted immunotherapies for autoimmune diseases and cancer by modulating cytotoxic T cells.
Windward Bio – Series A – $200M – January 2025
Windward Bio launched with a $200 million series A round led by OrbiMed, Novo Holdings, and Blue Owl Healthcare Opportunities, with co-investments from SR One, Omega Funds, RTW, Qiming, Quan Capital, and Pivotal bioVenture Partners. The company licensed global rights (excluding China and select Asian markets) to WIN378, a long-acting anti-TSLP antibody with potential biannual dosing. A Phase 2 trial in severe asthma is slated to begin soon, with initial data expected in 2026 and broader plans targeting asthma and COPD across major markets.
Tenvie Therapeutics – Seed – $200M – January 2025
Tenvie Therapeutics launched with $200 million in funding led by ARCH Venture Partners, F-Prime Capital, and Mubadala Capital. Formed around a portfolio of small-molecule programs acquired from Denali Therapeutics, Tenvie is advancing brain-penetrant and peripherally restricted therapies targeting inflammation, metabolic dysfunction, and lysosomal restoration in neurological diseases. Lead assets include IND-enabling NLRP3 and SARM1 inhibitors, alongside a broader pipeline focused on neuroprotective pathways such as TRPML1 and TMEM175.
Also check out Biopharma Therapeutics and Platforms Venture Funding – Q3 2024



